Quick Look
Grade Level: 7 (6-8)
Time Required: 45 minutes
Expendable Cost/Group: US $1.00
Group Size: 2
Activity Dependency: None
Subject Areas: Physical Science
NGSS Performance Expectations:

| MS-ESS2-1 |
Summary
Through discussion and hands-on experimentation, students learn about the geological (ancient) carbon cycle. They investigate the role of dinosaurs in the carbon cycle and the eventual storage of carbon in the form of chalk. Students discover how the carbon cycle has been occurring for millions of years and is necessary for life on Earth. Finally, they may extend their knowledge to the concept of global warming and how engineers are working to understand the carbon cycle and reduce harmful CO2 emissions.
Engineering Connection
Fossil fuels have enabled humans to achieve rapid industrial development, especially over the past 100 years. Millions of years ago, carbon-containing vegetation and animals (including dinosaurs) decayed at the bottom of ancient swamps and oceans, eventually forming fossil fuels such as coal, oil and natural gas. When we burn fossil fuels today to produce electricity, we are re-releasing the carbon (as carbon dioxide or CO2) contained in these ancient fuels. Today, people are concerned about the rising CO2 concentrations in the atmosphere due to the dangers of global warming. Engineers and scientists are working to reduce the accumulation of CO2 in the atmosphere.
Learning Objectives
After this activity, students should be able to:
- Define carbon as a necessary component of the Earth that supports all forms of life.
- Describe how it is possible that the carbon contained in chalk could possibly have originated from the respiration of a dinosaur living during the Jurassic Period.
- Explain why engineers are working to understand and rebalance the carbon cycle.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ESS2-1. Develop a model to describe the cycling of Earth's materials and the flow of energy that drives this process. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Develop and use a model to describe phenomena. Alignment agreement: | All Earth processes are the result of energy flowing and matter cycling within and among the planet's systems. This energy is derived from the sun and Earth's hot interior. The energy that flows and matter that cycles produce chemical and physical changes in Earth's materials and living organisms. Alignment agreement: | Explanations of stability and change in natural or designed systems can be constructed by examining the changes over time and processes at different scales, including the atomic scale. Alignment agreement: |
Common Core State Standards - Math
-
Use appropriate tools strategically.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Fluently add, subtract, multiply, and divide multi-digit decimals using the standard algorithm for each operation.
(Grade
6)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
The management of waste produced by technological systems is an important societal issue.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Analyze how the creation and use of technologies consumes renewable and non-renewable resources and creates waste.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
State Standards
Colorado - Math
-
Fluently add, subtract, multiply, and divide multidigit decimals using standard algorithms for each operation.
(Grade
6)
More Details
Do you agree with this alignment?
Colorado - Science
-
Compare and contrast the flow of energy with the cycling of matter in ecosystems
(Grade
6)
More Details
Do you agree with this alignment?
-
Identify and describe the impact of major geologic events on life on Earth
(Grade
7)
More Details
Do you agree with this alignment?
Materials List
Each student needs:
- splash-proof goggles (if conducting Alternative Procedure; see Troubleshooting Tips)
- Student Background Reading: Life of a Carbon Atom
- Dinosaur Breath Worksheet
Each group needs:
- 3-4 pieces of chalk (NOT dustless chalk)
- rolling pin and hard surface to crush chalk
- 1 small sandwich bag in which to crush chalk
- ¼ cup vinegar (either red or white)
- 2 small beakers, graduated cylinders, or small glass jars
- (optional) 1 small balloon
- (optional) 1 tsp. baking soda (see Troubleshooting Tips)
For the entire class to share:
- a few scales, to measure mass
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/cub_carbon_lesson01_activity1] to print or download.Pre-Req Knowledge
Students should be familiar with the main processes of the geological carbon cycle, including weathering, subduction, erosion and volcanic activity. It is also helpful if students have already been introduced to the concept of global warming and that human activity during the past 100 years has contributed to the increased emissions of carbon dioxide into the atmosphere.
Introduction/Motivation
Does anyone know what is contained in the piece of chalk I am holding in front of you? (Answer: Calcium carbonate.) Where would you guess this chalk is from (in terms of how it came to be "chalk")? Well, today we are going to find out! We are also going to learn why carbon is so important for life on Earth.
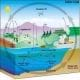
All living organisms, including plants and trees, fish in the ocean, and our own bodies, are made from the carbon atom. In fact, 18% of our bodies are made of carbon! Carbon atoms continually move through living organisms, the oceans, the atmosphere and the Earth's crust. This movement is known as the carbon cycle, which can take millions of years to complete.
Some carbon cycles, such as the carbon cycle that happens within our bodies when we eat, take less time to come full cycle. What did some of you eat today for breakfast? So, when you ate breakfast, you ingested carbon into your bodies in the form of carbohydrates and proteins. In your cells, oxygen combined with the food to give you energy for your day's activity. Carbon is a waste product of this process, and leaves your body when you exhale. Can anybody guess how carbon leaves our bodies when we exhale? I'll give you a hint – it's a gas, and plants and trees need it to produce energy. (Answer: Carbon dioxide, or CO2.)
Just as we are part of the carbon cycle, other animals are, too! Even dinosaurs that lived millions of years ago are part of the carbon cycle. Our experiment today will help us understand how the chalk that I am holding in front of you could contain carbon that was exhaled by a dinosaur that lived long ago. Can you imagine!?
First, can anyone tell me once more what chalk is made of? That's right – calcium carbonate. Can you guess where calcium carbonate/natural chalk comes from? A lot of natural chalk is mined from large rock formations such as the White Cliffs of Dover that form part of the British coastline. The Dover Cliffs are composed of calcium carbonate sediment. This sediment was created by the shells of ancient sea creatures that died and sank to the bottom of the ocean. These ancient sea creatures constructed their sturdy shells using some of the carbon that is found in the Earth's oceans. Oceans soak up a tremendous volume of carbon from the atmosphere. A long time ago, when dinosaurs walked the Earth, the Earth's oceans absorbed some of the carbon (as carbon dioxide) that was released when dinosaurs exhaled. So, it is very possible that the ancient carbon contained in a dinosaur's breath is here, in this piece of chalk today. Amazing!
You see, carbon atoms are never destroyed, they just move around and change form. Because the Earth can only tolerate so much carbon in one place at one time, it is important for scientists and engineers to understand how carbon cycles through the Earth. Today, many people are worried that human activities, particularly fossil fuel burning and deforestation are releasing unnatural amounts of carbon dioxide into the atmosphere, leading to global climate change. Humans burn fossil fuels such as coal, oil and natural gas, to produce energy to power our cars and light our houses and buildings. We practice harmful deforestation techniques when we clear cut forests for lumber. Deforestation emits carbon into the atmosphere as well.
Many engineers are working on creating beneficial technologies to reduce CO2 emissions. For example, environmental engineers are studying carbon sequestration, which is a term used to describe processes that remove carbon from the atmosphere. Mechanical and electrical engineers design buildings, homes, cars and appliances that use less energy. They are also engineering devices such as solar panels and wind turbines that generate electricity from renewable natural sources (the sun and the wind) and do not emit CO2 into the atmosphere.
Procedure
Before the Activity (Teacher Preparation)
- Gather materials and make copies of the Student Background Reading: Life of a Carbon Atom, and the Dinosaur Breath Worksheet.
- Read "Life of a Carbon Atom" and answer the investigating questions at the end of the reading.
- Prepare materials for the assessment activities, if desired.
With the Students
- Show students several carbon-containing objects to grab their interest.
- Review the activity tasks and learning objectives before starting the hands-on experiment. Write the objectives on the board or have students write them in their notebooks.
- Have students read the attached background information, "Life of a Carbon Atom" and then work individually or in pairs to discuss the investigating questions at the end of the reading before beginning the hands-on experiment. Allow about 20 minutes.
- After discussing the investigating questions in small groups or as a class, prepare students for the hands-on activity by explaining the activity set-up and procedure. The teacher may choose to demonstrate the activity for the students before they begin.
- Have groups label their two beakers or small glass containers as: Container #1 and Container #2.
- Next, have students seal their sticks of chalk into plastic bags. Using the rolling pin and a hard surface, crush the chalk into as fine a powder as possible.
- Have students fill Container #1 with about four tablespoons of the crushed chalk OR the crushed chalk/baking soda mixture (see Troubleshooting Tips). Have them fill Container #2 with ¼ cup of vinegar.

- Using the scale(*), have students measure the mass of Container #1 and Container #2 and record on their worksheets.
- After students record the mass of the two containers, have them pour the vinegar onto the crushed chalk and observe the chemical reaction. (Note: chalk + vinegar = CO2 [exhaled dinosaur breath] + water + calcium compound.)
- Have students record the mass of the reaction products (water + calcium carbonate in Container #1). (Note: the mass of the reaction products should be less than the original combined mass of Container #1 and Container #2. This is because the chemical reaction between the calcium carbonate and the vinegar released some of the carbon that was stored in the chalk into the atmosphere.)
*Alternative Procedure: If scales are not available, the release of carbon from the crushed chalk can be observed by placing a balloon securely around the mouth of Container #1, very quickly after the vinegar has been added (see Figure 2).The balloon will fill with carbon dioxide; however, for the balloon to fill a noticeable amount, use a crushed chalk/baking soda mixture (see Troubleshooting Tips).

- Have students describe the chemical reaction in qualitative terms — how did the reaction look, smell and sound? — on their worksheets.
- Give students time to clean up their workspaces and finish their worksheets.
- Conduct the creative writing post-activity assessment with the students or assign it as homework (see description in the Assessment section). Students need a sheet of paper and a writing utensil; markers, colored pencils and paint are optional.
Vocabulary/Definitions
atmosphere: The Earth's atmosphere is a layer of gases surrounding the planet and retained by gravity; contains roughly 78% nitrogen, 0.97% argon, 0.04% carbon dioxide; protects life on Earth by absorbing ultraviolet solar radiation and reducing temperature extremes between day and night.
carbon cycle: The exchange of carbon between the land, the oceans, the atmosphere and the Earth's interior.
Carboniferous period: A major division of the geological time scale that extends from the end of the Devonian Period (360 million years ago) to the beginning of the Permian Period (299 million years ago). Carboniferous rocks in Europe and eastern North America consist largely of limestone, sandstone, shale and coal beds. The Carboniferous coal beds provided much of the fuel for power generation during the Industrial Revolution.
erosion: The displacement of soil, mud rock and other particles by wind, water, ice and downward slope movement caused by gravity. Erosion is different from weathering because it involves movement. Although erosion is a natural process, in many places it is increased by human land use.
fossil fuels: Fossil fuels such as coal, petroleum products and natural gas are sources of ancient biomass that were formed millions of years ago from the decay of plant and animal matter.
global warming: The observed increase in the average temperature of the Earth's atmosphere and oceans in recent decades.
Jurassic period: The Jurassic period, also known as the Age of Dinosaurs, is a major unit of the geologic timescale that extends from the end of the Triassic Period (200 million years ago) to the beginning of the Cretaceous (145 million years ago).
photosynthesis: The process by which green plants create energy by absorbing solar energy and carbon dioxide from the atmosphere to produce carbohydrates (sugars). Plants "burn" these carbohydrates during respiration, which releases the energy contained in sugars to be used as fuel. Plants then release oxygen to the atmosphere, which is used for respiration by humans and other organisms.
respiration: The process by which an organism obtains energy through the reaction of oxygen with glucose to give water, carbon dioxide and ATP (energy).
subduction zone: An area on Earth where two tectonic plates meet and move towards one another, with one sliding underneath the other and moving down into the Earth's mantle. When an oceanic plate slides underneath a continental plate, this creates a zone with many volcanoes and earthquakes.
volcanic activity: Volcanic activity is caused by openings (or ruptures) in the Earth's surface or crust, which allows hot, molten rock, ash and gases to escape from deep below the surface. Volcanoes are usually found where two to three tectonic plates pull apart or come together. Volcanoes can also be caused by "hot spots" in the Earth's mantle. Volcanoes formed by hotspots are found elsewhere in the solar system, especially on rocky planets and moons.
weathering: The process of breaking down rocks, soils and their minerals through direct contact with atmospheric conditions such as heat, water, ice and pressure.
Assessment
Pre-Activity Assessment
Toss-A-Ball: Play a game to learn students' prior knowledge related to this activity. Divide the class into two teams and have one team start out with the ball. Ask one person from the team one of the questions from the list below. The teacher may want to ask additional questions to assess students' understanding of geological processes, global warming, carbon emissions into the atmosphere, and other real-world applications of the carbon cycle. If a team member answers the question correctly, his/her team receives one point and s/he has the opportunity to answer another question. If /when the team member answers incorrectly, s/he throws the ball to one of his/her teammates. If the teammate answers incorrectly, the ball is thrown to a person on the other team to give it a try.
Sample Questions
1. During what geological time period did dinosaurs live?
(Answer: Dinosaurs lived throughout the Mesozoic Era, which began 245 million years ago and lasted for 180 million years. It is sometimes called the Age of the Reptiles. The era is divided into three periods – the Triassic, Jurassic and Cretaceous.)
2. What element is the building block of life on Earth?
(Answer: Carbon)
3. What does the term "weathering" mean?
(Answer: Weathering is the process of breaking down rocks, soils and their minerals through direct contact with atmospheric conditions such as heat, water, ice and pressure.)
4. What is a subduction zone?
(Answer: A subduction zone is an area on Earth where two tectonic plates meet and move towards one another, with one sliding underneath the other and moving down into the Earth's mantle. When an oceanic plate slides underneath a continental plate, this creates a zone with many volcanoes and earthquakes.)
5. What is erosion?
(Answer: Erosion is the displacement of soil, mud rock and other particles by wind, water, ice and downward slope movement caused by gravity. Erosion is different from weathering because it involves movement.)
6. What is volcanic activity? What causes it?
(Answer: Volcanic activity is caused by openings (or ruptures) in the Earth's surface or crust, which allows hot, molten rock, ash and gases to escape from deep below the surface. Volcanoes are usually found where two to three tectonic plates pull apart or come together.)
Activity Embedded Assessment
Worksheet: Have students record measurements and follow along with the activity on their worksheet.s After students have finished their worksheets, have them compare answers with their peers.
Post-Activity Assessment
Worksheet Discussion: Discuss the results of the activity with students by reviewing the worksheet answers as a class. Did students find that adding vinegar to crushed chalk released some of the carbon contained in the chalk? Do they understand the chemical reaction? How is it possible that the carbon contained in the chalk is the same carbon that was exhaled by a dinosaur that lived long ago? Where is this carbon now? Why are engineers concerned about carbon in the atmosphere? What is global warming, and what can you do to help?
Creative Writing Practice: Have students write letters to a brontosaurus living the Jurassic Period from the perspective of a carbon molecule that was ingested by the brontosaurus in the form of a leafy fern and has recently found itself helping engineers to create technologies to help conserve energy. Ask students to explain the following:
Where is this carbon molecule today? For example, is the carbon molecule in an object, in the atmosphere, or in the process of moving from one place to another?
How did the carbon molecule arrive at its present state? Describe the events that occurred in the carbon molecule's life from the time of ingestion by the dinosaur to today. Was the carbon molecule previously in the atmosphere, as a CO2 molecule emitted from a respiring animal, a pile of decomposed wood or a power plant? Was the carbon molecule in a plant stalk or a human body?
What are the carbon molecule's plans for the future? Where is the best place for the carbon molecule to be if it does not want to contribute to global climate change? Have students imagine a technology that could be created by engineers to help reduce carbon emissions into the environment. How would this help reduce global warming?
Safety Issues
- Students should wear splash-proof goggles if conducting the activity using the Alternative Procedure.
Troubleshooting Tips
Because much of the chalk we use today is not natural chalk (pure calcium carbonate), the chemical reaction with chalk and vinegar is not as dramatic as it could be. One idea to evoke more of a "wow" reaction from the students is for the teacher to have the students crush the chalk and place it in a glass container, as described in the Procedures, and then add to the crushed chalk about one teaspoon of baking soda (sodium bicarbonate). The chemical reaction with the vinegar still produces CO2, but the reaction is more pronounced. Follow the rest of the procedures as written – that is, students should still record the mass of the chalk/baking soda mixture and determine how much carbon was released from the mixture as CO2. If conducting the alternative procedure, make sure students wear splash-proof goggles.
Activity Extensions
Carbon Sleuth: Give each student a carbon-containing object or an image of that object and ask them to figure out how the carbon exists in the object and where the carbon came from. Suggested objects: chalk, leafy plants, sea shells, a piece of wood, humans and other animals, pencil lead, a diamond, coal, a balloon full of air, etc. Students may take this activity home to conduct independent research and/or use the school library or Internet, if available. See the References section for some appropriate student websites.
Activity Scaling
For more advanced students, have them draw two carbon cycles – the geological carbon cycle and the biological/physical carbon cycle. Ask them to explain the differences between the two cycles, how they are interconnected, and the human role in each cycle. Have them explain why engineers and scientists are working to understand and rebalance the carbon cycle.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students are introduced to the concept of energy cycles by learning about the carbon cycle. They learn how carbon atoms travel through the geological (ancient) carbon cycle and the biological/physical carbon cycle.

Students learn and discuss the advantages and disadvantages of renewable and non-renewable energy sources. They also learn about our nation's electric power grid and what it means for a residential home to be "off the grid."
References
Wilson, Jim. National Aeronautics and Space Administration. Accessed May 25, 2007 http://earthobservatory.nasa.gov/
U.S. Environmental Protection Agency, Kids Site, October 23, 2006. Accessed May 29, 2007. http://www.epa.gov/climatechange/kids/
University Center for Atmospheric Research, Global Climate Change. Accessed October 14, 2006. http://www.ucar.edu/learn/1_4_2_16t.htm
Other Related Information
This activity was selected to be part of the Climate Literacy & Energy Network (CLEAN) reviewed and annotated digital library collection. CLEAN, an NSF National Science Digital Library (NSDL) Pathway, seeks out exemplary digital resources that relate to key climate and energy concepts, are scientifically robust and current, and are easily accessible online. The focus is on strong learning activities, with solid pedagogical scaffolding for grades 6-16. This resource passed a rigorous peer-review process as part of being selected to be included in the collection in April 2011. For the complete review, see: http://cleanet.org/resources/41913.html .
Copyright
© 2007 by Regents of the University of ColoradoContributors
Lauren Cooper; Malinda Schaefer Zarske; Janet YowellSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under grants from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and the National Science Foundation (GK-12 grant no. 0338326). However, these contents do not necessarily represent the policies of the Department of Education or the National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: August 31, 2020



User Comments & Tips