Quick Look
Grade Level: 11 (10-12)
Time Required: 3 hours
Split over 4 days (45-minute class periods)
Expendable Cost/Group: US $5.00
Group Size: 4
Activity Dependency: None
Subject Areas: Biology, Chemistry, Life Science, Science and Technology

Summary
Students learn how nanoparticles can be creatively used for medical diagnostic purposes. They learn about buckminsterfullerenes, more commonly known as buckyballs, and about the potential for these complex carbon molecules to deliver drugs and other treatments into the human body. They brainstorm methods to track buckyballs in the body, then build a buckyball from pipe cleaners with a fluorescent tag to model how nanoparticles might be labeled and detected for use in a living organism. As an extension, students research and select appropriate radioisotopes for different medical applications.Engineering Connection
Biomedical engineers are exploring new methods for delivering drugs in the body using buckyballs. Researchers must also be able to trace nanomaterials like buckyballs in the body, and chemists and chemical engineers are using technology based on radioisotopes to track them inside a living organism, or in vivo. For example, researchers at Rice University are engineering nanotubes loaded with gadolinium-based compounds—a component of a contrasting agent—as a way to improve sensitivity in magnetic resonance imaging (MRI). Medical professionals use a similar labeling technique engineered for use in positron-emission tomography (PET) scans to follow nanotubes through the body—objects that must contain a radioactive isotope before they can be traced.
Learning Objectives
After this activity, students should be able to:
- Describe how researchers are using nanotechnology, specifically buckyballs, to deliver drugs and therapies in the body.
- Build a buckyball out of pipe cleaners and identify its tracer (a fluorescent segment visible under a black light).
- Research and identify appropriate radioisotopes for use in tracing buckyballs in vivo.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
-
Investigating or designing new systems or structures requires a detailed examination of the properties of different materials, the structures of different components, and connections of components to reveal its function and/or solve a problem.
(Grades 9 - 12)
More Details
Do you agree with this alignment?
-
The functions and properties of natural and designed objects and systems can be inferred from their overall structure, the way their components are shaped and used, and the molecular substructures of its various materials.
(Grades 9 - 12)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Students will develop an understanding of the attributes of design.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop an understanding of the role of society in the development and use of technology.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop an understanding of the characteristics and scope of technology.
(Grades
K -
12)
More Details
Do you agree with this alignment?
State Standards
Texas - Technology
-
Creativity and innovation. The student demonstrates creative thinking, constructs knowledge, and develops innovative products and processes using technology. The student is expected to:
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Research and information fluency. The student applies digital tools to gather, evaluate, and use information. The student uses a variety of strategies to acquire information from electronic resources, with appropriate supervision. The student is expected to:
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Critical thinking, problem solving, and decision making. The student uses critical-thinking skills to plan and conduct research, manage projects, solve problems, and make informed decisions using appropriate digital tools and resources. The student is expected to:
(Grades
9 -
12)
More Details
Do you agree with this alignment?
Materials List
For the teacher to use:
- computer with internet access and projector to show American Chemical Society (ACS) landmark lesson and ChemMatters videos
- whiteboards or easel with paper
For the teacher to prepare in advance:
- fluorescent paint, to label at least one pipe cleaner for each group; find fluorescent paint at craft stores such as Michaels, from $2.50 - $9.99, in small bottles or as a paint pen
Each group needs:
- scissors
- 24 pipe cleaners; 16 of one color, 8 of another; with 1-2 pipe cleaners per group painted with fluorescent paint beforehand; $8 for 350 at Michaels or $12.95 for 500 at Amazon
- pipe cleaners for arts and crafts are usually ~30.5 cm (12 inches) long
- 16 pipe cleaners will be cut in half, making 32 pieces total; of those pieces, 20 will be made into hexagons and 12 will be made into pentagons
- 8 pipe cleaners will be cut into 3.8 cm (1.5 inches) pieces, making 64 pieces total
- Make sure to have extra pipe cleaners of each color on hand
- clear adhesive tape (in case some pipe cleaners do not hold together)
- Buckyball Instructions
- access to the Internet
For the class to share:
- black light flashlight, such as the Escolite UV Flashlight Black Light for $10 from Amazon
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/rice-2253-human-bloodhounds-nanomaterials-body-buckeyballs] to print or download.Pre-Req Knowledge
A basic understanding of carbon chemistry and of radioisotopes and half-life.
Introduction/Motivation
Wouldn’t it be nice to be able to deliver a drug directly into the body to the location where it is needed most? Most pharmaceuticals that are swallowed or injected end up being absorbed or spread throughout the body via the bloodstream, rather than going specifically to the site where they are most effective. But with the discovery of the buckminsterfullerene nanoparticle, or what we’ll refer to as a “buckyball,” in the late 1980s, an entire new field of nanotechnology emerged, and with it the possibility of customized treatment and personalized medicine.
Buckyballs are carbon allotropes—a chemical element that can exists in two or more physical forms. They are made of 60 carbon atoms (its chemical formula is C60) and arranged in a spherical shape built of alternating hexagons and pentagons. The unique hollow shape of a buckyball makes it feasible to load them with pharmaceutical agents, and their carbon structure makes them compatible with human tissues and cells. Buckyballs may also be labeled to target specific areas in vivo, which enables them to target certain body tissues. But because they are made purely of carbon, buckyballs are difficult to track in the human body. In order for researchers to know how effective buckyball-based medicine is, they must be able to follow it in the body before, during, and after treatment.
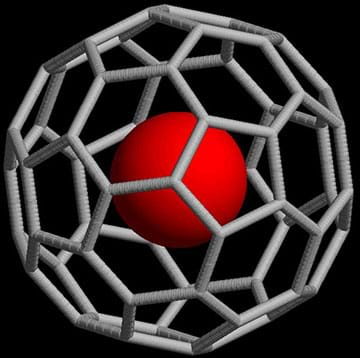
Over the next few class periods, we will learn about the discovery of buckyballs and their shape and structure. We will brainstorm ways to track buckyballs in the body, and we will build a buckyball that has been tagged to see if we can "track" it.
(Show the class the ChemMatters Episode 1: Nanotechnology’s Big Impact video linked through the ACS Discovery of Fullerene Landmark Lesson plan or by using the link directly.)
Procedure
Background
If time permits, conduct the Landmark Lesson Plan: Discovery of Fullerenes prior to this activity. This full lesson is accompanied by several activities on buckyballs and the historical discovery of fullerenes. Find more information on the ACS website.
Synopsis of buckyball history from the ACS: Harry Kroto, an organic chemist, was interested in studying long carbon chains that were discovered in interstellar space. In 1985, he collaborated with Robert Curl and Richard Smalley of Rice University to use an instrument that was designed to study clusters of atoms. The three scientists soon realized that they had discovered the long chains of carbon Kroto was interested in as well as previously unknown form of carbon in the form of an extremely stable, discrete molecule made up of 60 atoms. The team struggled to determine the structure of this unique molecule, but finally, after remembering the architect Buckminster Fuller and his geodesic dome, came up with a structure of interlocking pentagons and hexagons that created a spherical molecule with 60 carbon atoms. Their discovery earned them the Nobel Prize in Chemistry in 1996. Since the discovery of buckyballs, scientists have synthesized thousands of related compounds including nanotubes—hollow tubes that are cousins to the round buckyball. Practical applications of buckyballs occur in diverse fields such as energy, medicine, superconductivity, and optics.
Before the Activity
- Purchase fluorescent paint, a black light flashlight, and enough pipe cleaners for the class.
- Separate the pipe cleaners into two colors and cut them each in half. (Individual groups may have different colored pipe cleaners than other groups, as long as each group has pipe cleaners of two different colors.)
- Paint several of the pipe cleaner sections with the fluorescent paint and let the paint dry.
- Divide the pipe cleaners into groups for student teams to work on, making sure that each team has at least one or two painted pipe cleaners.
- Print out several copies of the Buckyball Instructions for making a pipe cleaner buckyball.
With the Students: Days 1-2
- With the entire class, discuss the concept of an allotrope and have students name allotropes of carbon (diamond, graphite, coal). Some students may already be familiar with fullerenes such as buckyballs and nanotubes. Discuss how the structure of allotropes affects their properties by comparing diamond, graphite and coal (FYI, refer to An Element of Many Forms: Allotropes of Carbon in the Landmark Lesson Plan: Discovery of Fullerenes).
- Discuss the history of fullerenes using information and, depending on time, activities from the Landmark Lesson Plan: Discovery of Fullerenes. Discuss the size and properties of buckyballs.
- Watch the ChemMatters Episode 1: Nanotechnology’s Big Impact video. Have students read the ChemMatters Magazine article: Nanotechnology’s Big Impact.
- The video and article mention that both mice and fruit flies have been exposed to nanomaterials such as nanotubes in various studies. Some studies showed that nanotubes are excreted harmlessly in urine, but other studies show that nanotubes that are inhaled may accumulate in the lungs and cause inflammation. Lead a discussion with students about the importance of researchers and doctors being able to track nanomaterials in the body in order to determine their effectiveness and whether/how they are expelled from the body after performing the desired task.
- Divide the class into groups of 3-4 students each and have them brainstorm methods of tracking buckyballs in the body. At the end of the brainstorming session, if radioactive tracers have not been discussed, introduce the use of radioisotopes and radioactive tracers as a method of detection nanotubes in MRI and PET scans. Many radioactive materials (radioisotopes) emit gamma rays, which can be detected with medical imaging and computer programs. They can be used to track the radioactive material in the body. If students have additional questions or are interested in this type of diagnostic therapy, see Positron Emission Tomography – Computed Tomography (PET/CT).
With the Students: Day 3
- Explain to students that we cannot use radioactive tracers in class, but that we will use the next best thing: fluorescence.
- Divide the class into groups of 3-4 students each. Give each group the Buckyball Instructions and distribute the allotted set of pipe cleaners. Remember that each group gets at least one fluorescent-painted pipe cleaner. Have the groups start constructing their buckyballs.
- Once the teams finish constructing their buckyballs, turn off the classroom lights. One group at a time, shine the black light flashlight on each segment of each group’s completed buckyball to detect the fluorescent segment on each buckyball. The flashlight activates the fluorescent paint and makes it glow in the dark. Have students recall the radioisotope in a nanotube. Their buckyballs are modeling the way PET scan would detect radioactive material that emits gamma rays. Have a class discussion about the effectiveness of the tags (are they visible or not?) and whether each buckyball needs more or fewer tagged segments.

- See Activity Extensions: access the information from the American Chemical Society National Historic Chemical Landmarks: Production and Distribution of Radioisotopes at Oak Ridge National Laboratory.
- Have students do further research to identify other radioisotopes that are used medically and have a class discussion to find the best radioisotope for drug researchers to use to tag buckyballs. A good radioisotope can a) be detected by PET scans because it gives off gamma rays; b) has a half-life between several hours and several days; c) is not so toxic that it cannot be administered to humans.
Vocabulary/Definitions
allotrope: A different physical form for the same element. For example, carbon naturally exists as diamond, graphite, and buckyballs.
buckyball: The common name for the buckminsterfullerene, a 60-carbon nanoparticle made up of twenty hexagons, twelve pentagons, connected at each vertex by a carbon atom. The buckyball was discovered in 1985 at Rice University.
fluorescence: The emission of light by a substance that has absorbed light or other electromagnetic radiation; a form of luminescence. Often, the emitted light has a longer wavelength, and therefore lower energy, than the absorbed radiation.
fullerene: The generic name for a molecule of carbon in the shape of a sphere, tube, ellipsoid, or other shape.
in vivo: A process, often during experimentation, performed or taking place in a living organism.
magnetic resonance imaging: A medical imaging technique, commonly known as a MRI, that uses strong magnetic fields, gradients, and radio waves to generate images of organs in the body. In contrast to other imaging techniques, magnetic resonance imaging does not use ionizing radiation.
nanotechnology: The science that deals with the manipulation of matter at the nanometer-sized scale. In order for a system or process to be considered “nanotech,” it must not only be effective at the nanoscale but must also have unique properties at that scale.
nanotube : A tube-like structure that may be large enough to serve as a pipe through which other nanoparticles can be channeled.
positron emission tomography: A nuclear medicine imagine technique, commonly known as a PET scan, which uses gamma rays from a radioisotope to create three-dimensional images in the body. This is often done in conjunction with a CT X-ray scan.
radioisotope: An unstable isotope of an element which, when it decays, gives off some form of radiation energy.
tag: In biomedical terms, a label that is attached to a molecule or nanoparticle that facilitates easy detection during drug delivery.
Assessment
Pre-Activity Assessment
Brainstorming: In groups of three of four, have students engage in open discussion. Remind them that no idea or suggestion is “silly.” Have them respectfully listen to all ideas. Ask the students:
- Have you ever heard of buckyballs? If so, what are they?
- Buckyballs are allotropes of carbon, made of 60 carbon atoms arranged in a spherical shape built of alternating hexagons and pentagons.
- Why would scientists need to track/trace a foreign delivery system (such as buckyballs) loaded with drugs in the human body?
- To determine the effectiveness of the buckyball-based medicine; that is, does the drug get delivered to the appropriate location?
- How can buckyballs, which are made of carbon, be tracked or traced in the human body?
- Attach radioactive tracers to the buckyballs. Many radioactive materials (radioisotopes) emit gamma rays, which can be detected, and in conjunction with MRI, PET, CT scans and computer programs, can be used to track the radioactive material in the body.
Activity Embedded Assessment
During Construction: Have students discuss the following questions within their groups:
- Why is the buckyball made of alternating pentagons and hexagons?
- This is the only structure that formed a closed C60 molecule when the university researchers were trying to determine its structure. Hexagons by themselves and pentagons by themselves did not results in the right number of carbon atoms.
- What other fundamental shapes could be used to construct the buckyball?
- The three scientists could have tried larger forms, such as octagons, but those are very rarely found in nature. They could have tried triangles, but those stressed forms of carbon would result in a molecule not as stable as the one discovered.
- Can an open-ended nanoparticle have a C-60 formula?
- No—in order to have purely carbon (without hydrogen or other elements), each carbon atom must be bonded only to other carbons. Because of carbon’s electron structure, it is not possible to achieve that with an open-ended molecule.
Post-Activity Assessment
Project Reflection: After checking for fluorescent tags on their buckyballs, have students again consider the questions they answered in the Pre-Activity Assessment, along with the following discussion questions:
- How would fluorescence work as an in vivo method of tracking nanoparticles? Does it depend on where the particles are in the body?
- Fluorescence is difficult to detect inside the body without powerful instruments. Imagine if we swallowed one of the fluorescent-painted pipe cleaners…how would we activate it? How would we see it?
- Would radioactive tags be more or less effective for tracking/tracing nanoparticles in the body? What sort of isotopes would work best? Does it depend on the length of time that the particles need to be tracked and the location in the body?
- Radioactive tags are more effective than fluorescence in the body, because gamma rays can easily pass through body tissues. The best radioisotopes would a) give off gamma rays so that they could be detected by PET scans; b) have a half-life between several hours and several days; c) not be toxic to humans in small doses.
Troubleshooting Tips
Have extra pipe cleaners available in case any are broken or bent beyond repair. Have some scotch tape in case some of the pipe cleaners do not stay together.
Activity Extensions
Lead a classroom discussion about radioactive isotopes, reminding students about the concepts of half-life and of types of radioactive decay. As a class, review the ACS information over the Production and Distribution of Radioisotopes and discuss the following issues:
- How does the radioisotope used for tracking depend on...
- its half-life?
- its toxicity?
- its availability?
- How does the radioisotope used for tracking depend on...
- the type of drug attached to the nanoparticle
- the location of the drug attached to the nanoparticle (on it or inside it, for example)
- the type of drug and/or disease being treated?
Activity Scaling
- For lower grades, have students simply build buckyballs and discuss allotropes, and learn that buckyballs are one of the allotropes of carbon.
- For higher grades, have students independently research different nanomaterial-based technologies used to diagnose and/or treat various diseases
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

By exploring nanotechnology through a scavenger hunt, students learn about the exciting new ways this technology is applied in different engineering fields. Students also hone their critical thinking skills by participating in the scavenger hunt as well as creating their own games from scratch.
References
Crivat, G. Taraska, J.W. “Imaging proteins inside cells with fluorescent tags.” 2012. Trends Biotechnol. 30(1): 8-16. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3246539/ (via NCBI.)
“Landmark Lesson Plan: Discovery of Fullerenes.” 2016 American Society of Chemistry. Accessed September 5, 2018. https://www.acs.org/content/acs/en/education/whatischemistry/landmarks/lesson-plans/discovery-of-fullerenes.html
Quardokus, Rebecca. “How to make a buckyball!” 2010. Notre Dame extended Research Community (NDeRC). Accessed September 5, 2018. http://freyr.phys.nd.edu/erc_blog/erc.nd.edu/blogs/rquardok/files/2010/12/buckyball_Directions1.pdf
Copyright
© 2018 by Regents of the University of Colorado; original © 2017 Rice UniversityContributors
Diana Gano; Donna TateSupporting Program
Nanotechnology RET, Department of Earth Science, School Science and Technology, Rice UniversityAcknowledgements
This material was developed in collaboration with the Rice University Office of STEM Engagement, based upon work supported by the National Science Foundation under grant no. EEC 1406885—the Nanotechnology Research Experience for Teachers at the Rice University School Science and Technology in Houston, TX. Any opinions, findings and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation or Rice University.
Last modified: September 13, 2018


User Comments & Tips