Quick Look

Summary
Using benchtop experimental techniques, students are allowed options of varying degrees of complexity to test for polyvalency or polyresistance. These investigations allow students to gain an understanding of organisms’ ability to survive challenging conditions, exploit possible evolved adaptations, find new hosts, or establish themselves in new biomes. The implications are to discover how antibiotic resistance may be overcome and how vital crops may be improved using traditional artificial selection methods.Engineering Connection
Biomedical engineers help to improve access to safe, effective, and affordable medicines, medical technologies, and devices through the development of new, innovative solutions. Biomedical engineers apply their knowledge of analytical and design principles to the development, testing, and analysis of novel and improved medical treatments and devices.
Whether it’s optimizing the composition of a particular substance or developing new products that are stronger and more reliable, chemical and biological engineers develop exciting new technologies for a variety of industries, including energy, agriculture, and environmental science.
Learning Objectives
After this activity, students should be able to:
- Compare polyvalency by quantifying the effectiveness of a bacteriophage to kill multiple targets.
- Develop hypotheses, select degrees of exposure to an antagonistic condition, and analyze the results.
- Analyze organisms’ polyresistance to various destructive elements.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-LS4-4. Construct an explanation based on evidence for how natural selection leads to adaptation of populations. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Construct an explanation based on valid and reliable evidence obtained from a variety of sources (including students' own investigations, models, theories, simulations, peer review) and the assumption that theories and laws that describe the natural world operate today as they did in the past and will continue to do so in the future. Alignment agreement: | Natural selection leads to adaptation, that is, to a population dominated by organisms that are anatomically, behaviorally, and physiologically well suited to survive and reproduce in a specific environment. That is, the differential survival and reproduction of organisms in a population that have an advantageous heritable trait leads to an increase in the proportion of individuals in future generations that have the trait and to a decrease in the proportion of individuals that do not. Alignment agreement: | Empirical evidence is required to differentiate between cause and correlation and make claims about specific causes and effects. Alignment agreement: Scientific knowledge is based on the assumption that natural laws operate today as they did in the past and they will continue to do so in the future.Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-LS4-6. Create or revise a simulation to test a solution to mitigate adverse impacts of human activity on biodiversity. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Create or revise a simulation of a phenomenon, designed device, process, or system. Alignment agreement: | Changes in the physical environment, whether naturally occurring or human induced, have thus contributed to the expansion of some species, the emergence of new distinct species as populations diverge under different conditions, and the decline–and sometimes the extinction–of some species. Alignment agreement: Humans depend on the living world for the resources and other benefits provided by biodiversity. But human activity is also having adverse impacts on biodiversity through overpopulation, overexploitation, habitat destruction, pollution, introduction of invasive species, and climate change. Thus sustaining biodiversity so that ecosystem functioning and productivity are maintained is essential to supporting and enhancing life on Earth. Sustaining biodiversity also aids humanity by preserving landscapes of recreational or inspirational value.Alignment agreement: When evaluating solutions it is important to take into account a range of constraints including cost, safety, reliability and aesthetics and to consider social, cultural and environmental impacts.Alignment agreement: Both physical models and computers can be used in various ways to aid in the engineering design process. Computers are useful for a variety of purposes, such as running simulations to test different ways of solving a problem or to see which one is most efficient or economical; and in making a persuasive presentation to a client about how a given design will meet his or her needs.Alignment agreement: | Empirical evidence is required to differentiate between cause and correlation and make claims about specific causes and effects. Alignment agreement: |
Materials List
Materials for Option 1
- personal protection equipment (PPE)
- autoclave
- culture chambers
- laminar flow benches (or a comparable sterile workbench)
- storage under refrigeration
- micropipettes
- Parafilm M®
- petri dishes
- live bacterial hosts are available from Carolina.com, such as Escherichia coli and Pseudomonas putida (2-4 bacterial hosts are recommended)
- Carolina.com has phage culture kits
Materials for Option 2
Each group needs:
- 20 mung beans seeds / group
- NaCl
- distilled water
- petri dishes
- cotton gauze/sterile paper towels
- 500 mL - 1 L beakers
- stirring rod
- hot plate
- strainer with handle
- fine-mesh bag
- twine
- access to sink with cold water
- small stone or other weight
- thermometer
- forceps or tweezers
- timer
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/rice-2789-polyvalency-polyresistance-organisms-bacteriophage-activity] to print or download.Pre-Req Knowledge
Students should be familiar with standard laboratory equipment and procedures for biological investigations, including heating liquids, mixing, and dissolving substances, taking precise mass measurements, recording temperatures, and performing calculations to prepare concentrations.
Introduction/Motivation
We are familiar with the extinction of the dinosaurs. Why do some species survive catastrophic and cataclysmic events, but others go extinct? Why do infectious diseases affect different individuals differently? Are there genetic reasons that allow some individuals, or populations of a species, to survive while others die?
Today you will design experiments to empirically test for polyvalency in bacteriophages or challenge the limits of organisms to survive different hostile conditions.
Biological polyvalency is the ability of an organism to infect or destroy multiple toxins or organisms. Bacteria have an ancient enemy: bacteriophages, which are sometimes simply referred to as phages. Bacteriophages are viruses that are usually specific to the bacterial strain they lyse and destroy. (Review lysis, if necessary.) New research into phages that are polyvalent shows promise to destroy bacteria, especially for antibiotic resistant bacteria (ARB).
Bacteria exposed to bacteriophages may have some level of survivability. Survivors may evolve immunities during this timeless dance between predator and prey, antagonist and protagonist. Resistance to multiple negative agents—pathogens or environmental conditions—allows greater survival of the species when confronted with a range of challenges, especially when these challenges occur simultaneously and overwhelm a population.
Today you will explore polyvalency by comparing the effectiveness of a bacteriophage to kill multiple targets; specifically, you will select bacteriophages that are effective against different bacteria. Alternatively, you may explore the polyresistance of organisms to various destructive elements, selecting the survivors from a population exposed to several antagonists. The subject may be exposed to these antagonists simultaneously or sequentially. In small groups, you will determine the degree of exposure to an antagonistic condition, develop hypotheses, and analyze the results.
A guiding principle of evolutionary theory is that genetic variability may allow for resistance or susceptibility to antagonists. Thus, the survivors of exposure to a normally lethal antagonist are those naturally selected for the continuation of their species; all other factors being equal, the variability found in germplasm may allow for some survivors. For bacteriophages, it allows them to lyse (and reproduce) by exploiting multiple strains of bacteria.
Another guiding principle in this investigation is the unknown. Outcomes are not predetermined, and you can set certain parameters with the teacher’s guidance. (This student inquiry is based on basic protocols; it is not intended to be a recipe for predetermined outcomes.)
Procedure
Background
With the degree of differences in high school laboratory resources, several of this investigation’s options are flexible enough to offer proof of concept for polyvalency and polyresistance for the instructor. Students will investigate bacteriophages or organisms for polyvalency or polyresistance. The test organisms could be bean or radish seed, protists, small crustaceans such as brine shrimp, worms (nematodes, earthworms), or even bacteriophages. The antagonists may include different concentrations of saline, bleach, triclosan, strains of bacteria, or different temperatures. Four options are presented below.
With limited time and resources, only small sampling sizes can be tested for genetic variability that may lead to survival from antagonists. A few bean seeds or a small sampling of bacteriophages does not fully represent the potential biodiversity in a species, much less in any population of that species. This should be emphasized and included in their reports, along with background research and a discussion.
Option 1: Bacteriophage-Bacteria. This is for labs equipped for, and students and instructors familiar with, Biosafety Level 2. Facilities should have laminar flow benches, micropipettes, microwave ovens for agar, or similar equipment. This option may take three to four consecutive days.
Option 2: Mung bean germination with heat and salinity. This is for laboratories, instructors, and students with limited access to, or experience with, the facilities or practices needed with the bacteriophage option. Still, it should prove to be a useful investigation for how genetic variability can be manifested through tolerances to unfavorable conditions.
Option 3: Fusarium. This option is to test mung beans for resistance to a fungal disease called wilt, caused by the fungus Fusarium, and followed by Option 4, which combines heat, salinity, and the wilt disease.
Control subjects should be included within a portion of the research for Options 2, 3 and 4.
**NOTE** Selected images for the options are at the end.
With the Students
Option 1 Bacteriophage – Bacteria
This investigation is based on research conducted at Rice University’s Nanosystems Engineering Research Center on Nanotechnology-Enabled Water Treatment (NEWT). The NEWT investigations isolated bacteriophages capable of lysing selected pernicious ARB. Typical bacteriophage protocols introduce the phages into a complex mixture of the multiple targeted species. This approach has had limited success. An alternative approach was found to be more successful at targeting multiple bacterial hosts. The alternative approach introduces phages sequentially to the targeted strains of ARB, selecting for efficacy through each progressive strain of ARB. The final selection was found to be effectively polyvalent.
Instead of introducing the bacteriophages into a community of the three targeted ABR bacteria, greater success was found by introducing the phages to one strain, selecting successful phages from that trial, introducing these to a second target strain, and then to a third strain. The final selection was polyvalent against all three target bacteria.
The teacher decides which organisms to use based on the school’s hardware and equipment, ethical code, and level of academic rigor. For example, phages and bacteria can be ordered from scientific supply houses if the school has biohazard waste disposal protocols; brine shrimp could be used if the school’s ethics code allows dosing at lethal concentrations; quickly germinating seeds could be used as an alternative to animals. The following instructions are for bacteriophages.
Before the Activity
- Hard agar should be prepared and kept sterile. For 1 L: prepare 10g Bactotryptone #211705; 11 g Agar, pure; 5 g CaCl. Autoclaved. While still hot, add 2 mL of 1M CaCl2; keep sterile. The hard agar is applied at a rate of 16 – 20 mL per petri dish. This should be done inside the flow bench while the agar is still hot and kept sterile.
- Soft agar has the bacteria and bacteriophage added to it while warm (116 °F); set the bottle of soft agar in a large beaker of warm water on a hot plate.
- For 0.5 L, prepare 5 g Bactotryptone; 3 g Agar; 2.5 g KCl. It is applied in a thin layer atop the hard agar layer.
With the Students
Part 1
- Sort the class into groups of three to four students each.
- Explain that there is a traditional method to test for bacteriophage polyvalency, and a new method.
The purpose of the investigation is to test for polyvalency. This is achieved by two protocols:
- Traditional: blending all the target bacteria together and adding the bacteriophage, or
- Sequential: applying it to one target bacterium and then the others in sequence.
The following instructions allow the students to continue with either of these two protocols.
- In a 15 mL centrifuge tube (or similar sterile container), add 5 mL of soft agar, 100 microliters of the bacterial culture(s), and 1,000 microliters (μL) of TSB plus 10 microliters of the bacteriophage. Quickly blend these with the tip of the micropipette and then pour over the hard agar layer.
- Cover the petri dish immediately and seal the edge with a strip of Parafilm M®. Incubate at 104 °F overnight. It is recommended that students write their sample data on the bottom of the dish using a marker.
Measuring Results
Always wear PPE and follow Level 2 bio-lab protocols from start-up through disposal of wastes.
- The next day, remove the petri dishes but keep them sealed with the Parafilm M®. Looking into the petri dish, count the spots that appear. The spots are plaques—colonies of the bacteriophage that have lysed the bacteria and successfully controlled it.
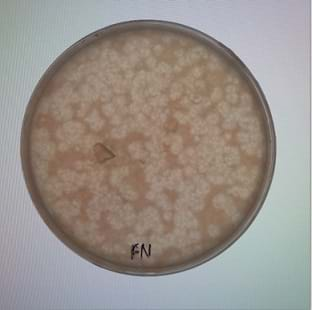
Bacteriophages. The “lawn” of the petri dish was inoculated with the target bacterium with the subject bacteriophage blended into it. The plaques (spots) are places where the bacteriophage is concentrated; these are also the locations where the bacteriophage was most effective at lysing and killing the bacteria. - A. If the traditional test was performed with multiple target bacteria, then only count the plaques and the test is finished. Dispose of all materials as biohazardous waste.
B. If this was the alternative test and the first of a sequence of tests, remain inside the flow bench and open the petri dish. With a sterile (flamed) scalpel or wire loop, carefully remove the area where plaques formed—only remove the top-most layer of the soft agar. 
Place these excised plaques into a 15 mL centrifuge tube and seal, performing all this work inside the flow bench.
Set the centrifuge tubes ready to add warm soft agar again.
Have ready new hard agar petri dishes and repeat steps with the second or third strain of bacteria to be tested.
- After all sequencing is done (2-4 target bacteria), compare the plaque counts for each test procedure—the complex with all target bacteria together and the sequenced bacteria.
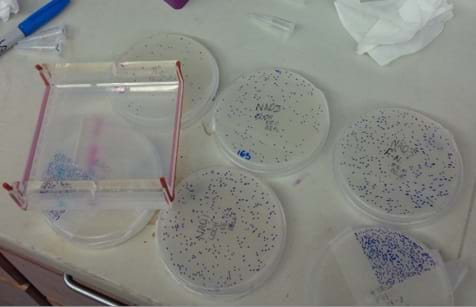
Meticulous counting of many plaques assists the researcher in learning the effectiveness of the bacteriophage against a given bacterium.
Polyvalency is achieved by lysing more than three of the target bacterial cultures. A complete write-up with background research and the significance of the relationship between bacteria and phages is customary, along with potential novel applications for phages to manage ARB.
Repeat Steps 1-3. It may be that no bacteriophages could lyse a bacterium or bacteria. Dispose of all contaminated materials as biohazard waste.
Option 2: Mung Bean Germination with Heat and Salinity
**NOTE** While polyvalency is the ability to destroy various hosts, a similar concept is the ability to resist or tolerate multiple antagonists. This investigation tests survivability after exposure to multiple antagonists in a non-biohazard laboratory setting.
**NOTE** Mung beans typically germinate in 3-5 days with sufficient moisture and at room temperature; they will remain pale and have extended shoots if germinated in the dark.
Background
Two investigations are presented, one with heat and the other with salinity as the antagonist. The equipment needed is relatively common in classroom laboratories.
Investigation 1: Heat Screening
Beans or radish seeds can be germinated while exposed to various concentrations of toxic substances or conditions. For example, it is not uncommon to heat-treat seed to kill pathogens such as bacterial canker and leaf rots present on the surface of or within the seed. Usually, a temperature of about 122 or 125 °F is used for 20 to 25 minutes. (An Internet search will reveal various established protocols, such as: https://www.iasj.net/iasj?func=fulltext&aId=50468.)
However, how would agriculturists select for seed that is infected with bacteria that have developed tolerance to 125 °F?
Precise controls are needed to effectively kill the bacteria without killing the seed’s germ. The tolerances are tight, and this may be an application for finding seed that is tolerant of higher temperatures to account for evolved heat-resistance in bacterial pathogens. Once the students are aware of the typical limits for seed heat treatment, they may opt for testing with higher temperatures, shorter exposure times, etc. This should be their choice.
Investigation 2: Salinity Screening
Plants with halophilic qualities have evolved to tolerate salty soils, yet many crops are intolerant of soil salinity. A problem in drier climates where crops are irrigated, soil salinization can significantly reduce crop yields, even taking some fields out of production (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4336437/). Beans are one crop that is not very tolerant of salt soils, according to Knott’s Handbook for Vegetable Growers, as adapted from E. V. Maas, ‘‘Crop Tolerance,’’ California Agriculture (October 1984). Beans may be a good choice to use as a test subject for tolerance of both heat and salinity. 

**NOTE** The threshold salinity for beans is 1.0 dS/m, at which point damage is observed and yields are reduced.
Mung bean is easily germinated in moist sand media, on damp cotton gauze or paper towels placed flat inside petri dishes. The seed is considered very sensitive to salt.
With the Students
Heat Screening
- Select seeds (maybe 5 seeds in 4 sets, 20 seeds) and subject them to the hot water immersion while constantly stirring the water. Students are given the known protocols, and they decide which other temperatures or times to test. It is suggested that 3-5 sets be tested at no more than 36 °F intervals with a time of less than 20 minutes, i.e., high temperatures and much shorter times.
- Set up the hot plate near the sink.
- Procedure (allowances may be made to let the students design the process):
- With the water stirred and stabilized for several minutes at the target temperature, place the seeds inside the mesh bag with the weight and tie with the twine.
- Leave roughly 30 cm of twine hanging outside the beaker.
- Place the bag with the seeds into the beaker of water and immediately begin the timer.
- Constantly stir the water for even temperature distribution.
- When time is up, remove the bag by grasping the twine and pulling it out and placing it into the strainer.
- Immediately place the strainer holding the hot bag under the cold water and liberally rinse it to stop the heating process and remove excess heat from the seeds.
- Rinse under distilled water and let the seeds drain excess water for a consistent time (30 seconds? Let the students learn the value of consistent protocols) before proceeding.
- Have the gauze or paper towels cut to fit inside the petri dishes; dampen this media with distilled water.
- Place the heat-treated seeds onto the sterile gauze or paper towels with forceps or tweezers, avoiding damaging the softened seed.
- Cover the petri dish and place at room temperature out of direct sunlight.
- Wait for observed damage upon germination, 3-5 days, or note failure to germinate.
- Record results.
Salinity Screening
- Select seeds (maybe 5 seeds in 4 variable sets, 20 seeds).
- Prepare saline solutions of distilled water and NaCl at various concentrations; all solutions should be at room temperature.

Table 1 Equivalents for Salt Concentrations - Immerse the seeds inside the tied mesh bag into a beaker with the desired concentration of salinity.
- Allow the seeds to soak (students choose the time, ~2 minutes), stirring once or twice to wet them. Then remove the seeds; do not rinse under tap water, but allow excess water to drain from the seeds.
- Moisten the gauze or paper towels with the same saline concentration and place the seeds onto the media inside a petri dish; the media should be damp but not saturated.
- Cover the petri dish and place at room temperature out of direct sunlight.

It is best to trim the media so it does not stick out beyond the closed petri dish, as the paper towel will wick moisture out from the petri dish. 
The trials were set into a dark closet to await germination, which typically takes 3-5 days. - Wait for observed damage upon germination, or note failure to germinate.
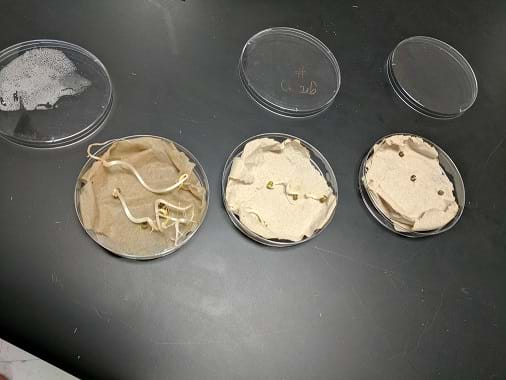
The results post treatment after five days. Moisture consistency is important, as mung bean seeds were less likely to germinate on dry media.

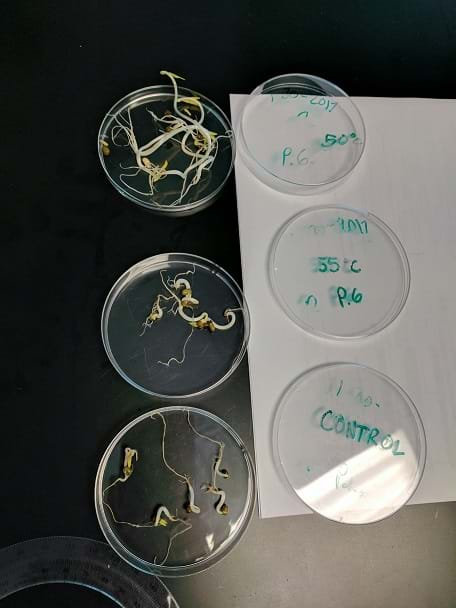
Heat and Salinity Screening
Have the students develop their own method for testing the seed by combining the two treatments.
Options 3 & 4: Fungal Wilt Resistance & Multiple Antagonists Screening
Fusarium oxysporum is available from Carolina.com. It is the fungus species that causes a serious crop disease commonly called wilt. Fusarium resistance is an ongoing area of study for many crops, and mung beans may be utilized to test for resistance. Following a protocol similar to the one for bacteriophage polyvalency, mung bean seed can be germinated inside a petri dish with agar that is inoculated with Fusarium. Because the concentration of CFU of the Fusarium may not be available, an assumption can be made that there is enough viable antagonist for infectability of all seed in the inoculated agar media. Note: Fusarium is a plant disease pathogen. Be careful to use proper sanitation protocols and dispose of any contaminated materials as biohazard waste.
Fusarium Screening
Assume 15 mL of agar per petri dish, 30 dishes, and 5 seeds per dish.
Prepare 500 mL of agar at 116 °F, adding the Fusarium and stirring to mix it thoroughly, then distributing it quickly to the 30 dishes, 15 mL each. Add 5 beans that have been soaking in sterile distilled water (for ~5 minutes) to the dishes, cover, and set out of direct sunlight at room temperature until germination. Observe changes and select seeds that appear to be resistant to this fungal disease.
Multiple Antagonists Screening
Seed that has been heated at various temperatures, as described in the Heat Screening section, or that has been soaked in salinized solution, as described in the Salinity Screening section, may be used in the Fusarium test.
Example: Seed subjected to 126 °F, 130 °F, 133 °F, and 137 °F is placed into the Fusarium-inoculated petri dishes and observed for tolerance to both high temperature and fungal infectability.
Vocabulary/Definitions
antibiotic resistant bacteria : Bacteria that mutated in response to one or more antibiotics and are now resistant to their control.
bacteriophages: Viruses that infect bacteria and are generally considered species or strain specific.
Fusarium: A genus of fungi that is mostly harmless, although several species are serious pathogens of important crops.
germination : The process of a seed becoming activated and beginning growth as a seedling plant.
lysis: The disintegration of cells resulting in cell death.
polyresistance: Being unaffected by more than one disinfectant or antibiotic.
polyvalent bacteriophages: Phages that can infect bacteria from different genera.
salinity: The concentration of salt (sodium chloride) in water.
Assessment
Pre-Activity Assessment
In a brainstorming session, ask students the following:
- How do unfavorable conditions affect a species’ survival?
- What role would genetic variability play for a species to resist challenging conditions?
- What purpose does manipulating an experiment’s variables have in understanding responses to changes?
Activity Embedded (Formative) Assessment
As students work in their groups and begin to set the parameters for testing their subjects, ask each group about their experimental design and the use of multiple variables. Provide insight about how testing different variables within the same experimental setup may prove challenging. Redirect and guide groups to apply practical experimental design.
Post-Activity (Summative) Assessment
In composing their lab report, essential questions addressing polyvalency or polyresistance should be fully addressed as appropriate for the experiment chosen by each group. Different groups should have selected different phages and bacteria, antagonistic conditions, or challenging treatments for their test subjects. These essential questions, and possible answers, are below. Students’ answers should reflect their ability and level of preparation and may be much more developed than the brief answers provided.
- What is the relationship between bacteriophages and bacteria? (Answer: Bacteriophages are viruses that lyse and destroy bacteria to replicate themselves.)
- How does polyvalency allow bacteriophages to have a survival advantage? (Answer: By having multiple bacteria to lyse, phages can improve their chances of replication.)
- How does genetic variability affect a species’ survival under unfavorable conditions? (Answer: Due to differences in the genetic makeup of an organism, it may have an advantage and survive during unfavorable conditions while most of its species’ members are killed.)
- How could bacteriophage polyvalency change the outcome of a bacteria’s population? (Answer: For bacteriophages, polyvalency means they could lyse and destroy various strains of bacteria.)
- What purpose does manipulating this experiment’s variables have in establishing resistance thresholds? (Answer: By applying various antagonistic challenges to the subjects, students may select for those that exhibit resistance to the chosen conditions by surviving or thriving. For bacteriophages, the challenge is to introduce them to different bacterial hosts and then select for polyvalency. For seeds, this is seen by subjecting them to Fusarium and different heat treatments, or saline concentrations.)
- How would the compounded results of multiple negative effects affect a species, and how does this mimic conditions in real life? (Answer: By exposing the subjects to various challenges, the students are mimicking the antagonisms to living organisms in real life. In nature, organisms are subjected to a wide range of environmental and organic challenges. From excess moisture to excess heat and/or excess cold, and from desiccation to predation, infections, and blights, organisms must have the naturally occurring genetic variability that, despite existential threats, has allowed species to survive over time.)
Safety Issues
- Having students handle heated objects, especially glass (beakers, thermometers, etc.), can be hazardous.
Troubleshooting Tips
- The teacher should allow guidance for salinity treatments based on Table 1 above.
- Students should be aware that heat kills seeds as well as pathogens. Overheating seed (too high a temperature or for too long a time) will kill the seed’s embryo.
- Subjecting the seed to heated saline at different concentrations of NaCl and at different temperatures creates a situation that is difficult to test due to the growing number of variable sets. This should become apparent to the students as they plan their protocols, leading them to develop a scheme whereby they effectively test the subjects without overburdening their investigation. It is also a reason why scientific study investigates one variable at a time.
- In natural selection settings, seedlings germinate in environments with many pathogens and unfavorable conditions present. It may become apparent to the students that this is one reason why most plants produce so many seeds, and why open pollination succeeds in producing genetic diversity. Seed companies use artificial selection to develop cultivars for many reasons, such as greater yields, color, flavor, packing ability, etc. They also select for resistance or tolerance to disease, salinity, temperature extremes, etc., subjecting seeds and plants to a host of antagonists, with many of them not succeeding. Artificial selection has given us improved crops in many ways over the past several millennia.
-
Option 1 Bacteriophage – Bacteria is the more rigorous of the investigations and is suited for laboratories equipped to handle sterile equipment and have Level 2 Biohazard controls in place. Few high school facilities can meet these protocols.
Option 2: Mung Bean Germination with Heat and Salinity was successfully accomplished at a Class 6A high school, and Options 3 & 4: Fungal Wilt Resistance & Multiple Antagonists Screening should be equally practicable in most high school settings.
Activity Extensions
For enrichment, these experiments could be carried out as a semester-long period investigation of the polyvalency of bacteriophages or of the generational changes for mung beans to demonstrate polyresistance.
By increasing the number of individuals in the test, students could observe and record data showing that greater test subjects provide better data than smaller, possibly biased, test samples.
Note:
These investigations are suited for higher-level biology, environmental science, or agronomy students, such as AP, IB, or introductory college level science courses. As such, students are expected to create their own lab data collection methods and record their data as raw data/field reports. Finished lab reports should be student generated. Comparative analyses data tables of the different groups’ results may be created by the teacher.
Activity Scaling
Depending on the equipment and capability of the students, the four options allow for a variety of learning rigors to suit the level of students.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!Copyright
© 2024 by Regents of the University of Colorado; original © 2017 Rice UniversityContributors
Carlton C. ColmenaresSupporting Program
Research Experience for Teachers, Nanotechnology Enabled Water Treatment Systems (NEWT), Rice UniversityAcknowledgements
This material was developed based upon work supported by the National Science Foundation under grant no. EEC 1406885—the Nanotechnology Research Experience for Teachers at the Rice University School Science and Technology in Houston, TX. Any opinions, findings and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation.
Special thanks to AP Environmental Science students and the microbiology lab research team at Rice University of Dr. Pedro Alvarado, Pingfeng Yu and Lingli Li for their guidance in lesson creation, Dr. Carolyn Nichol, and Christina Crawford Alston for their unfailing support of the NEWT and RET programs.
Last modified: April 29, 2024



User Comments & Tips