Quick Look
Grade Level: 11 (9-12)
Time Required: 45 minutes
(See timing note in the Procedure section)
Expendable Cost/Group: US $3.00 This activity also uses some non-expendable (reusable) items such as microscopes and lab equipment; see the Materials List for details.
Group Size: 3
Activity Dependency:
Subject Areas: Biology, Chemistry, Life Science, Science and Technology
NGSS Performance Expectations:

| HS-ETS1-2 |
| HS-PS1-6 |

Summary
Students learn how crystallization and inhibition occur by examining calcium oxalate crystals with and without inhibitors that are capable of altering crystallization. Kidney stones are composed of calcium oxalate crystals, and engineers and doctors experiment with these crystals to determine how growth is affected when a potential drug is introduced. Students play the role of engineers by trying to determine which inhibitor would be the best for blocking crystallization.Engineering Connection
Crystals are present in many forms in the human body, and some can cause damage. Kidney stones form when the concentration of certain ions reaches high levels of supersaturation, forming crystals that block waste from exiting the body. The body naturally attempts to inhibit crystallization with other ions, however more help is necessary in most cases. Biomedical and chemical engineers and doctors design drugs to inhibit crystallization and prevent future blockages.
Learning Objectives
After this activity, students should be able to:
- Identify which inhibitor is more effective.
- Explain why one molecule is more likely to inhibit than another.
- Identify to which face an inhibitor binds.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-ETS1-2. Design a solution to a complex real-world problem by breaking it down into smaller, more manageable problems that can be solved through engineering. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Design a solution to a complex real-world problem, based on scientific knowledge, student-generated sources of evidence, prioritized criteria, and tradeoff considerations. Alignment agreement: | Criteria may need to be broken down into simpler ones that can be approached systematically, and decisions about the priority of certain criteria over others (trade-offs) may be needed. Alignment agreement: | |
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-PS1-6. Refine the design of a chemical system by specifying a change in conditions that would produce increased amounts of products at equilibrium. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Refine a solution to a complex real-world problem, based on scientific knowledge, student-generated sources of evidence, prioritized criteria, and tradeoff considerations. Alignment agreement: | The structure and interactions of matter at the bulk scale are determined by electrical forces within and between atoms. Alignment agreement: In many situations, a dynamic and condition-dependent balance between a reaction and the reverse reaction determines the numbers of all types of molecules present.Alignment agreement: Criteria may need to be broken down into simpler ones that can be approached systematically, and decisions about the priority of certain criteria over others (trade-offs) may be needed.Alignment agreement: | Much of science deals with constructing explanations of how things change and how they remain stable. Alignment agreement: |
International Technology and Engineering Educators Association - Technology
-
Students will develop an understanding of the role of troubleshooting, research and development, invention and innovation, and experimentation in problem solving.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop an understanding of and be able to select and use medical technologies.
(Grades
K -
12)
More Details
Do you agree with this alignment?
State Standards
Texas - Science
-
evaluate models according to their limitations in representing biological objects or events; and
(Grades
9 -
11)
More Details
Do you agree with this alignment?
-
in all fields of science, analyze, evaluate, and critique scientific explanations by using empirical evidence, logical reasoning, and experimental and observational testing, including examining all sides of scientific evidence of those scientific explanations, so as to encourage critical thinking by the student;
(Grades
9 -
12)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- 1 20-ml scintillation vial with a polypropylene screw cap (such as case of 500 vials for $164 at amazon.com; reusable)
- microscope slide (such as large plain economy 3 x 2-inch slides, $25.90 for pack of 144 at tedpella.com; one slide makes 10 smaller square slides)
- stir plate
- stir bar, 5 mm length (such as a pack of 10 for $50.70 at sigmaaldrich.com; reusable)
- Calcium Oxalate Worksheet, one per student
- COM Procedure Handout, one per group (only necessary if enough time is available for students to make the calcium oxalate crystals instead of the teacher making them; see the Procedure section for details)
To share with the entire class:
- microscopes with 40x optical lenses (borrow from biology lab or $85 each at amscope.com), ideally about 12 per class so students can rotate through 12 different crystal slides; may need fewer or more, depending on the number of crystal slides grown and how you run the class
- 20 plastic coverslips (such as 100-pack for $6.85 at tedpella.com)
- diamond glass cutter or file, to cut glass slides (such as one for $3.50 at lowes.com)
- 1 bottle NaCl (such as 25 gram bottle for $30 at sigmaaldrich.com)
- 1 bottle calcium chloride dihydrate (such as 25 grams for $36.40 at sigmaaldrich.com)
- 1 bottle sodium oxalate (such as 100 grams for $43.30 at sigmaaldrich.com)
- 3 gallons deionized water (this is enough for 10 ml per group making crystals and 1 liter for each of the 8 stock solutions; you may not need this much)
- 1 bottle acetone (such as 1 liter for $59.50 at sigmaaldrich.com)
- 2 boxes kimwipes
- 2-3 tweezers
- 1 bottle bovine serum albumin (such as 1 gram for $43.20 at sigmaaldrich.com)
- 1 bottle transferrin (such as 100 mg for $86 at sigmaaldrich.com)
- 1 bottle sodium citrate dihydrate (such as 5 grams for $30.50 at sigmaaldrich.com)
- 1 bottle chondroitin sulfate A (such as 5 grams for $57.20 at sigmaaldrich.com)
- 1 bottle dimethyl hydroxyglutaric acid (such as 5 grams for $80.80 at sigmaaldrich.com)
- oven/incubator, capable of 60 °C (140 °F)
- scale, capable of measuring mg quantities
- 2 scoopula spatulas
- 1000-μL pipette
- 1 box of weigh paper
- 1 1000-ml round-bottom flask
- 1 25-ml round-bottom flask
- 2 1000-ml jars
- 5 25-ml jars
- petri dishes, one for each glass slide of crystals (such as a case of 500 60-mm diameter dishes for $70 at http://www.sks-science.com/)
- permanent markers
- (optional) cell phones or iPads, to take photographs of the crystals by holding the phone or iPad camera to the microscope eyepiece, so you can measure the crystals using PowerPoint®
Supplies note: These bottles of chemicals will last for many years and are enough for 200 or more groups.
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/uoh_crystals_lesson01_activity2] to print or download.Pre-Req Knowledge
Familiarity with the human body's urinary system, how kidneys remove waste, crystallization, inhibition and drug design. Before conducting this activity, students should have learned and discussed the background information provided in the PowerPoint® presentation in the associated lesson, Body Full of Crystals, so they are informed about what they will see under the microscopes.
Introduction/Motivation
How can our understanding of crystals and the crystallization process help us provide medical care for people? (Listen to student ideas.) As an example, most kidney stones are composed of calcium oxalate crystals. Researchers are currently investigating, creating and testing new drugs with the intent to prevent the formation of kidney stones, since statistically more than 50% of the people who have passed one kidney stone are expected to have re-occurrences. As part of this research, chemical engineers examine how organic molecules, or peptides, bind to the calcium oxalate crystals. For today's activity, you will put yourselves in the shoes of current researchers to determine which molecules have the potential to be future drugs.
Procedure
Timing
This activity can be done two different ways, depending on whether you have just 50 minutes or more time—an additional 75-minute period. If you have 50-minutes plus an additional 75 minutes with the students, follow the Procedure below to have students in groups of three grow the calcium oxalate crystals themselves; it is a simple procedure in which they just add NaCl and the stock solutions together at the correct times. After the crystals are grown, students individually examine them using the microscopes and compare to other groups who added different inhibitors or no inhibitors. If you only have 50-minutes, prepare the calcium oxalate crystals with and without inhibitors before conducting the activity with students. Thus, making the crystals is a team activity and analyzing the crystals is an individual activity.
Before the Activity
Gather materials and make copies of the Calcium Oxalate Worksheet, one per student.
If only a 50-minute class period is available:
- Following the instructions on the COM Procedure Handout, make calcium oxalate crystals with and without inhibitors for two sets of 6 microscopes (12 total). (It is also described below in the "75-Minute Class Period" sections.).
- Set out the microscopes with one-half of the room having one set of slides and the other side of the room having the other set.
If 75-minute plus 50-minute class periods are available:
- Make copies of the COM Procedure Handout, one per group.
- Use a diamond glass cutter to cut the microscope slides into 1 x 1 cm squares.
- As necessary, mark out areas for each class in the incubator showing where to place their vials. Turn on the incubator and set it to 60 °C.
- Make the stock solutions of calcium chloride, sodium oxalate, albumin, transferrin, chondroitin sulfate A sodium salt, sodium citrate dehydrate, and dimethyl hydroxyglutaric acid for students using round bottom flasks as described in the procedure handout. Prepare 1-liter solutions for calcium chloride and sodium oxalate. Prepare 25-ml solutions for all of the inhibitors.
With the Students—For Only 50-Minute Class Period
- Help students find the crystals in the microscope by placing them on top of the thin plastic coverslips underneath the microscopes. (The pieces may not be large enough to sit over the opening of the optical lens, while the plastic coverslip does.) Have students take turns using the microscopes to view all of the crystals with different inhibitors. Refer to Figure 1 for the shapes, sizes and pervasiveness of crystals expected to be seen under the microscope.
- Direct students to fill out the worksheet table with their own drawings that show how each crystal looks with inhibitors.
- (optional) For help in making comparisons, have students take pictures with cell phones or iPads and use PowerPoint® to measure the widths and lengths of the crystals. Record these measurements on the drawn crystals on the worksheet.
- Have students complete the worksheets.
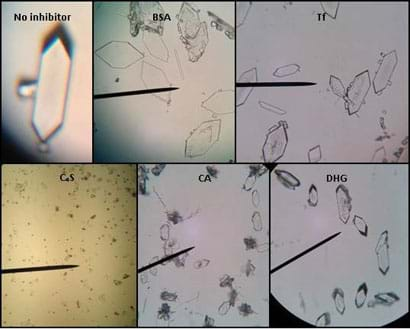
Figure 1. Calcium oxalate crystals grown in the presence of inhibitors to determine which are more effective at blocking crystallization. Please note, the images do not have scale bars to be able to compare sizes.
With the Students—For 75-Minute Plus 50-Minute Class Periods
- While students look at the procedure handout in groups, go over the procedure as a class. Hand out the worksheets.
- Direct students to gather the following materials for their groups: 20-ml vial, stir bar and glass slide.
- Make sure students use permanent markers to label their vials with their names and inhibitors.
- Have students proceed independently to conduct the experiment, guided by the procedure handout. Monitor students as they perform the entire experiment.
- Students measure out the amount of sodium chloride and add the necessary deionized water (see procedure handout, step 2) to the vial while stirring.
- Using a pipette, add the amount of calcium chloride. Let the solution stir for 1 minute then place in the 60 °C oven for 1 hour.
- Remove the vial from the oven and use a pipette to add the inhibitor solution and sodium oxalate solution to the vial. Let the solution stir for a minute. Remove the stir bar using a magnetic stir bar retriever. (To prevent contamination, make sure this is cleaned between groups if shared.)
- Use tweezers to place the square glass slide in the bottom of the vial. Cap the vial and place it in the incubator, in the area identified for your class.
- Three days later, have groups remove their square glass pieces from the vial, wash with deionized water and left to air dry overnight in a petri dish or some type of holder that can be propped open.
- Help students find the crystals in the microscope by placing them on top of the thin plastic coverslips underneath the microscopes. (The pieces may not be large enough to sit over the opening of the optical lens, while the plastic coverslip does.) Have students take turns using the microscopes to view all of the crystals with different inhibitors. Refer to Figure 1 for the shapes, sizes and amounts of crystals expected to be seen under the microscope.
- Direct students to fill out the worksheet table with their own drawings that show how each crystal looks with inhibitors.
- (optional) For help in making comparisons, have students take pictures with cell phones or iPads and use PowerPoint® to measure the widths and lengths of the crystals. Record these measurements on the drawn crystals on the worksheet.
- Have students complete the worksheets.
Vocabulary/Definitions
calcium oxalate: A molecule that comprises the most common type of kidney stone.
crystal: A solid material that consists of an ordered pattern in all directions.
dehydration: The excess loss of body water.
efficacy: A measure of how much a drug is able to inhibit. For example, causing 100% inhibition is high efficacy.
face: A side of a crystal.
growth: The third phase of crystallization during which more molecules attach to the stable nuclei, causing the crystal to grow in size.
inhibition: The disruption of normal crystal growth, or blockage of further growth.
kidney: An organ in the urinary system that removes waste products, and regulates electrolyte levels and blood pressure.
kidney stone: A crystalline structure that forms in the kidney due to supersaturation, causing blockages to waste removal.
kink: A position on a crystal surface where molecules are most likely to attach.
nucleation: The second phase of crystallization during which clusters of molecules become a stable nuclei. The molecules arrange themselves into an ordered pattern, which is the basic building structure of the crystal.
potency: A measure of how much of a drug produces a large amount of inhibition.
solubility: The maximum amount of solute that can be dissolved into a solvent.
solute: In a solution, the component that is being dissolved into another.
solvent: In a solution, the component that another component is dissolved into.
step: In a crystal, a layer or sheet of molecules stacked on top of each other.
supersaturation: A condition in which the amount of solute dissolved in a solvent is above the solubility.
terrace: On a crystal face, the flat surface between steps.
toxicity: A measure of the degree of harmfulness of a molecule to humans.
urinary system: The organ system that removes waste from the body and regulates electrolytes, blood pressure and blood pH.
Assessment
Pre-Activity Assessment
Group Pop Quiz: Divide the class into groups and give each a small white board and a marker. Ask the class questions about crystals that were taught during the PowerPoint® presentation from the associated lesson, Body Full of Crystals (such as the discussion questions, below) and have the groups write down the answers. Reward the group with the most correct answers (extra credit or a treat).
Discussion Questions: Lead a class discussion. Ask students:
- Where is a molecule most likely to bind on a crystal surface to grow to inhibit? (Answer: At a kink.)
- What is a type of crystal that forms in the body? (Possible answers: Cataracts, kidney stones [calcium oxalate, cystine, uric acid or struvite], hemozoin, calcification [bones], insulin, etc.)
- How does temperature affect crystallization? (Answer: Higher temperatures increase the rate of crystallization.)
- During the crystallization process, which stage has an energy barrier that needs to be overcome before crystals grow? (Answer: The second stage, nucleation.)
- Show students the graph from the last slide in the associated lesson's PowerPoint® file and ask: Which is more potent and which has more efficacy? (Answer: Blue is more potent and red is more efficacious.)
Activity Embedded Assessment
Observations in Table: Have students fill out the Calcium Oxalate Worksheet table by drawing what crystals look like for each inhibitor and how they look without an inhibitor. Review their answers to gauge their comprehension of the subject matter.
Post-Activity Assessment
Analysis & Conclusion: Have students complete the Calcium Oxalate Worksheet questions to determine which inhibitor is best suited as a future drug and why. Review their answers to assess their depth of comprehension.
Investigating Questions
What happens when the concentration of the inhibitor is changed? (Answer: The concentration of the inhibitor affects the crystal growth. If more inhibitor is added, less growth occurs and the size of the crystals may be smaller and/or fewer crystals created. The morphology/shape of the crystals may be more distorted in comparison to those grown without an inhibitor. If less inhibitor is added, the crystals are closer in size and shape to crystals grown without an inhibitor.)
Safety Issues
When cutting the microscope slides, the glass has the potential to cut skin, so be careful and cautious when snapping them apart.
Troubleshooting Tips
The oven temperature need not be stable, but higher temperatures cause the crystallization to go faster, so try to stay at or above 60 °C or crystals may not appear after three days.
Activity Extensions
As an extension, attempt the same experiment using different concentrations of the inhibitors to see how the change in concentration might affect the amount of inhibition from the same molecule. Try starting at 10 μg/ml and increasing the concentration by 10 μg/ml to make a potency and efficacy graph by plotting percent inhibition as a function of inhibitor concentration, just as engineers and researchers do.
To make a graph, have students make the crystals as performed previously, but with different inhibitor concentrations. Then have them examine the crystals by microscope and take photographs. If the same optical lens is used (40x), students can compare sizes and shapes from the images.
Activity Scaling
For lower grades, prepare the crystals in advance and then have students look at them with the microscopes. As a class, work through the worksheet to help students understand that different crystal shapes result depending on where blocking molecules bind. Use different-sized blocks to demonstrate how molecules halt growth in one direction, but not another.
Additional Multimedia Support
Microscopic, real-time calcium oxalate surface growth (29 seconds; Movie4.avi; also provided as an mpeg attachment): https://www.youtube.com/watch?v=RVl5wyS6Y94
Microscopic, real-time crystal surface growth of cystine (12 seconds; Movie3.mov): https://www.youtube.com/watch?v=x2HoNCF9pu8
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn about various crystals, such as kidney stones, within the human body. They also learn about how crystals grow and ways to inhibit their growth. They also learn how researchers such as chemical engineers design drugs with the intent to inhibit crystal growth for medical treatment purpo...

Students see and learn how crystallization and inhibition occur by making sugar crystals with and without additives in a supersaturation solution, testing to see how the additives may alter crystallization, such as by improving crystal growth by more or larger crystals.

Students are challenged to think as biomedical engineers and brainstorm ways to administer medication to a patient who is unable to swallow. They learn about the advantages and disadvantages of current drug delivery methods—oral, injection, topical, inhalation and suppository—and pharmaceutical desi...
References
Farmanesh, Sahar, Sriram Ramamoorthy, Jihae Chung, et al. (2014) "Specificity of Growth Inhibitors and their Cooperative Effects in Calcium Oxalate Monohydrate Crystallization." Journal of the American Chemical Society. Vol. 136, No. 1, pp. 367-376. http://pubs.acs.org/doi/abs/10.1021/ja410623q
Copyright
© 2014 by Regents of the University of Colorado; original © 2014 University of HoustonContributors
Andrea Lee, Megan KetchumSupporting Program
National Science Foundation GK-12 and Research Experience for Teachers (RET) Programs, University of HoustonAcknowledgements
This digital library content was developed by the University of Houston's College of Engineering, based upon work supported by the National Science Foundation under GK-12 grant no. DGE 0840889. Any opinions, findings and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation.
Last modified: May 10, 2017





User Comments & Tips