Summary
Using their knowledge of the phases of matter, the scientific method, and polymers, student teams work as if they are chemical engineers to optimize the formula for slime. Hired by the fictional company, Slime Productions, students are challenged to modify the chemical composition of the basic formula for slime to maximize its "bounce factor."
Engineering Connection
Similar to chemical engineers, students take raw materials and turn them into useful products. Then, working with project budget constraints, they apply their knowledge of polymers and methodically test their slime mixtures to modify their properties while minimizing waste and unnecessary expenditures.
Learning Objectives
After this activity, students should be able to:
- Perform and modify experimental procedures based on qualitative data from previous experiments.
- Organize and record qualitative data.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-ETS1-2. Design a solution to a complex real-world problem by breaking it down into smaller, more manageable problems that can be solved through engineering. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Design a solution to a complex real-world problem, based on scientific knowledge, student-generated sources of evidence, prioritized criteria, and tradeoff considerations. Alignment agreement: | Criteria may need to be broken down into simpler ones that can be approached systematically, and decisions about the priority of certain criteria over others (trade-offs) may be needed. Alignment agreement: | |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-2. Evaluate competing design solutions using a systematic process to determine how well they meet the criteria and constraints of the problem. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Evaluate competing design solutions based on jointly developed and agreed-upon design criteria. Alignment agreement: | There are systematic processes for evaluating solutions with respect to how well they meet the criteria and constraints of a problem. Alignment agreement: | |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-4. Develop a model to generate data for iterative testing and modification of a proposed object, tool, or process such that an optimal design can be achieved. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Develop a model to generate data to test ideas about designed systems, including those representing inputs and outputs. Alignment agreement: | Models of all kinds are important for testing solutions. Alignment agreement: The iterative process of testing the most promising solutions and modifying what is proposed on the basis of the test results leads to greater refinement and ultimately to an optimal solution.Alignment agreement: | |
International Technology and Engineering Educators Association - Technology
-
Analyze how an invention or innovation was influenced by its historical context.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Conduct research to inform intentional inventions and innovations that address specific needs and wants.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
State Standards
National Science Education Standards - Science
-
Scientists formulate and test their explanations of nature using observation, experiments, and theoretical and mathematical models. Although all scientific ideas are tentative and subject to change and improvement in principle, for most major ideas in science, there is much experimental and observational confirmation. Those ideas are not likely to change greatly in the future. Scientists do and have changed their ideas about nature when they encounter new experimental evidence that does not match their existing explanations.
(Grades
5 -
8)
More Details
Do you agree with this alignment?
Texas - Science
-
in all fields of science, analyze, evaluate, and critique scientific explanations by using empirical evidence, logical reasoning, and experimental and observational testing, including examining all sides of scientific evidence of those scientific explanations, so as to encourage critical thinking by the student;
(Grades
10 -
12)
More Details
Do you agree with this alignment?
-
organize, analyze, evaluate, make inferences, and predict trends from data; and
(Grades
10 -
12)
More Details
Do you agree with this alignment?
Materials List

Each group needs:
- white school glue, 100 ml
- sodium borate (borax), 100 g, available at grocery stores
- 10 disposable cups, 4-8 oz size
- 10 Popsicle sticks (or wooden dowels)
- Slime Productions Budget Sheet
- Slime Productions Challenge Worksheet
- blank paper and pencils
To share with the entire class:
- access to the Internet to view or print the four-page article, "The Science of Slime"
- balances, for weighing ingredients
- scoopula or spoons, for measuring borax and glue
- (optional) food coloring, to create colorful slime! (may want gloves if use food coloring)
- (optional) white lab coat and nametag, for teacher use
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/uoh_opslime_activity1] to print or download.Pre-Req Knowledge
Perform this activity after reviewing the scientific method with a special focus on experimental design. Students should be familiar with basic chemistry, including polymers.
Introduction/Motivation
(To get students engaged in this activity, enter the classroom wearing a white lab coat with a name tag that states your name and "Slime Productions" in green letters.)
You're hired! My name is (insert your name), and I am the research director for Slime Productions. My company has hired you and your engineering firms to help Slime Productions with one of our projects. I created the 10-formula for slime several years ago, however the chemical composition of that formula does not meet our new specifications. Your chemical engineering challenge is to refine the formula.
Procedure
Background
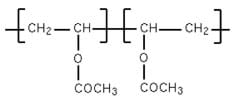
In this activity, students produce one variety of "slime" from a recipe that uses white school glue, which contains polyvinyl acetate. Polyvinyl acetate is an example of a polymer. Figure 1 shows a small section of polyvinyl acetate, which consists of two monomers.
The other key ingredient is sodium tetraborate decahydrate (referred to in the Materials List as sodium borate or borax). When mixed with the white glue, the borax solution creates links between different chains of polyvinyl acetate, which is a phenomenon known as cross-linking. Due to the cross-linking, the mixture's viscosity increases, creating what we commonly call "slime."
Slime is a non-Newtonian fluid, meaning its viscosity can be influenced by factors other than temperature and pressure. By altering the concentrations (or amounts) of borax and white glue, the properties of the slime can be altered. During this activity, students work to find the best ratios among borax, white glue and water, in order to meet the requirements and constraints of their client, Slime Productions.
Before the Activity
- Gather materials and make copies of the Slime Productions Budget Sheet and Slime Productions Challenge Worksheet.
- Be prepared with the ability (computers or printouts) to have students read the online article.
- (optional) Have ready a lab coat and nametag.
With the Students – Day 1
- Conduct the Introduction/Motivation section (~ seven minutes).
- Review the steps of the engineering design process, as it applies to this activity. See the Assessment section for details.
- Have students read the four-page article, "The Science of Slime." (See References section for details.)
- Review or introduce students to the scientific concepts associated with the production of slime by reviewing the vocabulary terms.
- Divide the class into groups of two or three students each. Each group represents an engineering firm hoping to be hired by Slime Productions.
- Further describe the chemical engineering challenge from Slime Productions:
- Slime Productions wants a "bouncier" formula (bouncier than 10-formula).
- In order to be hired, your engineering team must determine which ingredient is responsible for the bounciness of slime and then optimize the formula.
- All the engineering teams are in competition, and only one engineering firm will be hired!
- Slime Productions will hire the engineering firm that best meets the following criteria: 1) correctly identify the bounce factor, 2) maximize the height of the first bounce of 20 g of slime, and 3) conduct experiments with the lowest cost possible.
- Hand out and review the budget sheet.
- Hand out the worksheet. Say: To help you get started, I have arranged for you to synthesize samples of the 10-formula slime. Give students five minutes to make their own samples following these steps (which are also on the worksheet):
- In a chemical container (a paper cup), dissolve 10 grams of sodium borate into 10 grams of water.
- In a second container (a paper cup), dissolve 10 grams of white glue into 10 grams of water.
- Use stirring tools (Popsicle sticks) to mix the components in each of the cups.
- Then, pour the contents of one cup into the other cup.
- Continue stirring.
- Then, give students time to brainstorm ideas and conduct one or two trials of their own slime formulas. Make sure students know that they are altering the quantities of the slime ingredients to determine the best formula for bouncier slime. During the trials, remind students to:
- Record all expenses on the budget sheet, and record accurate measurements and test results.
- Always use 20 g of slime for each trial, so that the engineering firms can compare results!
- At the end of Day 1, ask teams to turn in all documentation for their experimental trials—since it is confidential information.
With the Students - Day 2
- Relay to students any suggestions from your review of the team records from Day 1. Hand back the documentation.
- Give students 40 minutes to continue conducting trials. Remind them to document their results and findings for their client, Slime Productions.
- With 10-15 minutes left, remind students to:
- Record their final formulas, being sure to identify which ingredient is responsible for the bounce factor.
- Test and record the first bounce height of their final formulas by recording the first bounce height three separate times (in meters). Instruct students to take the average of those three first bounce heights. The calculated average is the number reported to the client.
- Tally all expenses for the experimental trials based on items recorded on their budget sheets.
- Assign student teams to prepare professional presentations of their results for submission to Slime Productions, as described in the Assessment section. Evaluate results and declare a winner.
Vocabulary/Definitions
cross-linking: Bonds that link one polymer chain to another.
monomer: A molecule that can be bonded to other identical molecules to form a polymer.
Newtonian fluid: A fluid that has a viscosity that is only dependent upon temperature and pressure, such as water.
non-Newtonian fluid: A fluid that has a viscosity that can be changed by other factors, such as stirring.
optimize: To find the best compromise solution among several, often conflicting or competing, requirements or factors.
polymer: A substance that has a molecular structure built up chiefly (or completely) from a large number of similar units bonded together.
viscosity: The resistance of a fluid to flow, a measure of the thickness of a liquid.
Assessment
Pre-Activity Assessment
Engineering Design Process Practice: Ask students what they know about the engineering design process. Tell them that these are the overall steps that they (and all engineers) follow to make the bounciest slime for their client. If students have never done this before, have them first guess what the steps might be. Then review the stages. Before or during the activity, explain how each step of the engineering design process corresponds to what they are doing:
- identify the need/problem (Slime Productions' challenge to create a bouncier slime formula)
- research and brainstorm (read the scientific article; learn how to make the basic 10-formula slime, write down ideas for many possible slime formula variations with your teammate)
- develop possible solutions and select the best solution (compare your possible solutions to each other; decide which seems to best meet all the requirements and constraints)
- construct a prototype (create slime with your new formula)
- test and evaluate the solution (use a bounce test to test your new formula; record results)
- communicate the solution (document exactly what you did and the results)
- redesign (from what you learn, go back to step 2 and brainstorm again, following the next steps to keep making adjustments, test and evaluate again, until satisfied that you have a winning solution)
Activity Embedded Assessment
Teacher Observations: Observe the student groups to see if they are:
- using information from "The Science of Slime" article to guide their decision making
- recording their experimental procedures and data in an organized fashion
- demonstrating an understanding of the engineering design process by following the seven steps outlined as a class during the pre-activity assessment
Day 1 Documentation: At the end of the first day, collect and review the groups' documentation (worksheets, observations, lab notebooks, budget sheets, etc.). From this information, note any groups that need more assistance or are struggling with the application of their knowledge of the engineering design process, the engineering challenge or the scientific concepts.
Post-Activity Assessment
Final Submission: Require groups to gather together into professional presentations the following information for submission to Slime Productions. Then check to see that teams met the requirements, and which was most successful in meeting the challenge within budget.
- A final slime formula that includes a detailed list of materials, "recipe" and procedure
- Identify the ingredient responsible for the bounce factor (Answer: Borax)
- Bounce height data for 20 g of slime (trials and calculated average) (Rating: less than .05 meters = below average; between .05 and .10 meters = fair; .10 meters or higher = excellent!)
- Budget sheet with a total expense amount (Constraint: $6,000 per group)
Troubleshooting Tips
It can be time consuming to measure the glue. To save time, pre-measure units of glue, especially for Day 1 experimental trials.
If adding food coloring to create colored slime, it is important that students either wear gloves or only use stirrers, so as to avoid staining skin and clothing.
Both the glue and the slime are easier to clean up after they have dried.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!References
Rohrig, Brian. "The Science of Slime." Published December 2004. ChemMatters. American Chemical Society, Washington DC. Vol. 22, No. 4, pp. 13-16. Accessed January 19, 2012. (A four-page article about slime: It oozes. It's goopy. Sometimes it's liquid, sometimes solid. Learn about viscosity, Newtonian and non-Newtonian fluids, but most of all enjoy yourself. Plus a how-to-make-slime activity with photos.) http://www.acs.org/content/dam/acsorg/education/resources/highschool/chemmatters/articlesbytopic/solidsliquidsgases/chemmatters-dec2004-slime.pdf
Copyright
© 2013 by Regents of the University of Colorado; original © 2011 University of HoustonContributors
Leslie StilesSupporting Program
National Science Foundation GK-12 and Research Experience for Teachers (RET) Programs, University of HoustonAcknowledgements
Created through the University of Houston's Cullen College of Engineering's NSF Research Experience for Teachers (RET) Program, grant no. 1130006. However, these contents do not necessarily represent the policies of the National Science Foundation and you should not assume endorsement by the federal government.
Last modified: April 30, 2020




User Comments & Tips