Quick Look
Grade Level: 6 (5-6)
Time Required: 45 minutes
Lesson Dependency: None
Subject Areas: Chemistry
NGSS Performance Expectations:

| MS-ESS3-4 |

Summary
Students are introduced to acids and bases, and the environmental problem of acid rain. They explore ways to use indicators to distinguish between acids and bases. Students also conduct a simple experiment to model and discuss the harmful effects of acid rain on our living and non-living environment, as well as how engineers address acid rain. Students can also learn how persuasive techniques are used to develop an argument, and create an environmental case study.Engineering Connection
Environmental and chemical engineers examine pollutants to find the substance's pH, which helps them determine the kind of impact it may have on human health and the environment. For example, the effects of acid rain include damage to water, wildlife, plants, forests, crops, buildings, statues, bridges and vehicles. Reinforce the students' learning of the effects of Acid Rain through the activity, Acid Rain Effects. Knowing the pH of pollutants also helps to decipher the initiating chemical and environmental causes behind a pollution problem. Students can learn how to distinguish between acids and bases in the activity, Acid (and Base) Rainbows, to learn more about the pH. In response, engineers develop useful technologies to alleviate pollution sources, such as catalytic converters that remove certain poisonous gases from vehicle exhaust fumes. Have students expand their knowledge by constructing an environmental case study to persuade others about such potential dangers in the activity Is That Legal? A Case of Acid Rain.
Learning Objectives
After this lesson, students should be able to:
- Describe some general characteristics and common examples of acidic, basic and neutral solutions.
- Understand that engineers use acids and bases in a variety of ways.
- Identify cause-effect relationships with acid rain and the environment.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ESS3-4. Construct an argument supported by evidence for how increases in human population and per-capita consumption of natural resources impact Earth's systems. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This lesson focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Apply scientific ideas to construct an explanation for real-world phenomena, examples, or events. Alignment agreement: | Typically as human populations and per-capita consumption of natural resources increase, so do the negative impacts on Earth unless the activities and technologies involved are engineered otherwise. Alignment agreement: | Cause and effect relationships may be used to predict phenomena in natural or designed systems. Alignment agreement: All human activity draws on natural resources and has both short and long-term consequences, positive as well as negative, for the health of people and the natural environment.Alignment agreement: Scientific knowledge can describe the consequences of actions but does not necessarily prescribe the decisions that society takes.Alignment agreement: |
International Technology and Engineering Educators Association - Technology
-
Students will develop an understanding of the effects of technology on the environment.
(Grades
K -
12)
More Details
Do you agree with this alignment?
Worksheets and Attachments
Visit [www.teachengineering.org/lessons/view/cub_air_lesson06] to print or download.Introduction/Motivation
To introduce students to the concepts of pH, acids and bases, conduct the following hands-on class activity.
Before Class
Prepare enough of the following items for each student:
- dried apricots (1 piece, about 1 cm2)
- grapefruit pieces (1 piece, about 1 cm2)
- lemon pieces (1 piece, about 1 cm2)
- crackers (1 saltine)
- water (enough for a sip)
- milk (enough for a sip)
- molasses (enough for a small taste on a spoon)
- 1 small plastic spoon
- 4 small paper cups
Prepare the following in-class visual aids, as either overhead projector transparencies or student handouts: the attached pH Values of Common Substances, Acid Rain Formation — Small Scale, and Acid Rain Formation — Large Scale.
With the Students
- Ask students to suggest definitions of the following terms: pH, acid, base (alkaline) and neutral.
- Ask them to consider where they may have heard these terms before. (Possible responses: Testing the pool/hot tub/aquarium, pH-balanced shampoo, deodorants, scrapbooking, etc.)
- Tell students that they interact with acids and bases every day.
- Distribute the food samples to students.
- Ask the students to sample each item. Have them rank the foods in order of which tasted the least bitter (sour) to the most bitter (sour). This step can be done formally on paper or more informally as a class.
- After they rank the items, ask them to group the foods based on characteristics of taste.
- Write the pH for each test item on the blackboard (see Table 1).
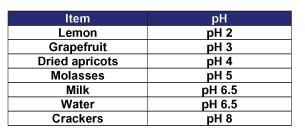
- Explain the general idea of a pH scale to the students. The pH scale ranges between 0 and 14, with 7 being neutral. Mention that acids have a pH of less than 7 and bases have a pH of greater than 7. Refer to the pH Values of Common Substances attachment.
- Do the groups that the students suggested agree with this new information? Can they come up with some common taste descriptions of acids and bases? (Rule of thumb: Acids are bitter, bases are often sweeter.)
- Explain that the pH scale is based on the number 10. Explain that the molasses has a pH of 5. The dried apricot has a pH of 4 and is 10 times more acidic than the molasses. Grapefruit (pH 3) is 10 times more acidic than the apricots and 100 times (10 x 10) more acidic than the molasses. The lemons (pH 2) are 1,000 times (10 x 10 x 10) more acidic than the molasses. Does this information agree with their taste descriptions?
- Take a class vote. Have students vote by holding thumbs up for true and thumbs down for false. Count the votes and write the totals on the board. Give the right answer. Ask the students:
- True or False: Scientists and engineers often test substances by tasting them. (Answer: False. Scientists and engineers never test substances by tasting them. They use special paper that changes color depending on if the substance is an acid or base [students can do this in the Acid (and Base) Rainbows activity], or they use electronic pH probes.)
Lesson Background and Concepts for Teachers
All substances are divided into three categories — acids, bases and neutral substances. Refer to the Acid (and Base) Rainbows activity to help students distinguish between the categories. Acids and bases are some of the most important substances on Earth. There are strong and weak acids and bases, and their strengths are described by the pH scale.
There are three theories that define acids and bases:
- The Arrhenius theory states that acids ionize in water to produce H+ ions and that bases ionize to produce OH- ions.
- The Bronsted-Lowery theory states that acids donate protons and bases accept protons.
- The Lewis theory states that acids are electron-pair acceptors and a bases are electron-pair donors.
Strong acids and bases completely dissociate in water (no acid/base left in original solution). Weak acids partially dissociate into water so that some acid is left.
Acids and bases combine in neutralization reactions. Strong acids combine with weak bases and strong bases combine with weak acids.
The pH Scale
The pH scale is used to describe the hydrogen-ion (H+) concentration of an aqueous solution. The concentration of hydrogen ions is measured on a logarithmic scale. If you use a calculator: pH = - log [H3O+] (also seen as - log [H+]). The pH scale ranges between 0 and 14, with 7 being neutral. A solution with a pH below 7 is acidic, and a solution with a pH greater than 7 is basic (alkaline). Each one-unit change in pH is a tenfold increase (or decrease) in the strength of the acid or base. A change from pH 5 to pH 3, for example, would be a 100 times (10 x 10) increase in acidity.
It may be helpful to use the concept of money to explain relative acid/base strength. Assign a neutral pH (7) the value of 1 cent. A pH of 6 (or 8) is ten times stronger, so it is worth 10 cents. A pH of 5 (or 9) is ten times stronger than that, so it is worth 100 cents (or $1). A pH of 4 (or 10) is 10 times stronger than that, so it is worth $10.
See the pH Values of Common Substances attachment for more information.
Indicators
Indicators are tools used to measure/indicate the pH of a substance. A color change in the indicator corresponds to different pH levels.
Litmus paper is frequently used to investigate the pH of different substances. Litmus paper is infused with litmus, a water-soluble blue powder derived from certain lichens (a type of fungus). The paper color changes depending on the pH of the substance with which it is in contact. The more basic a substance, the bluer the indicator turns. The more acid the substance, the redder the paper turns. There is wide-range pH litmus paper available as well as acid/base specific paper (pink litmus paper turns blue for bases, blue litmus paper turns pink for acids).
Another naturally occurring indicator is red cabbage juice. The pigment in the cabbage juice is what actually acts as the indicator. Red cabbage juice starts out purple and becomes more and more red as a solution becomes more acidic. It becomes blue, green or yellow in basic solutions.
Red cabbage contains two main types of plant dyes: anthocyanin and flavonol. Anthocyanin pigments are red in strongly acidic solution, blue in neutral and weakly basic solutions, and colorless in strongly basic solutions. Weakly acidic solutions contain some of the red form and some of the blue form and thus appear purple. Flavonol pigments are colorless in acidic and neutral solutions, and yellow in basic solutions. Weakly basic solutions thus contain both blue (anthocyanin) and yellow (flavonol) dyes, and appear to be green. The pH corresponding to various colors varies slightly with concentration, solvent, age and variety of cabbage. Most flowers and fruits contain anthocyanin as pigments.
Acid Rain
When environmental and chemical engineers examine a pollutant, they find out whether the substance is an acid or base in order to know what kind of reactions it causes. Acid rain is an environmental problem that concerns many engineers. The effects of acid rain include damage to the limestone and marble in statues and buildings; weakening of the exposed metal on bridges and cars; damage to bodies of water, wildlife, plants, forests and crops; and the contamination of the drinking water supply. Students conduct their own investigation with living and non-living items with the Acid Rain Effects activity. Researchers are investigating other possible effects of acid rain on human health.
Robert Angus Smith, an English chemist, first used the term "acid rain" in 1852 when he noted the connection between the acidity of London's rainfall and its polluted skies. Most scientists agree that "normal" rainfall has a pH of 5.6. Rain in the atmosphere reacts with carbon dioxide (CO2) to form a weak carbonic acid, altering the rain pH to 5.6 (instead of a pH of 7).
Acid rain is defined as any form of wet precipitation (rain, snow, fog, dew or sleet) that has a pH less than 5.6 (on a scale of 0 to 14, with 7 being neutral). Large quantities can also be deposited in a dry form through dust. Acid rain is more acidic than normal rain and forms through a complex process of chemical reactions involving air pollution and water molecules in the air. The two most important pollutants that contribute to the formation of acid rain are nitrogen oxides (NOx) and sulfur dioxide (SO2), which react with moisture in the atmosphere to form nitric and sulfuric acid. See the Acid Rain Formation — Small Scale attachment.
The sulfur and nitrogen compounds that contribute to acid rain primarily come from combustion products (burning coal and oil) from large industrial and utility sites. Emissions also come from automobiles and other forms of transportation, and other industrial processes such as smelting. See the Acid Rain Formation — Large Scale attachment.
One way that we can help prevent acid rain is by burning less fossil fuel. We can also make laws that prevent large factories from burning fossil fuels or that require them to limit (minimize) their pollutant output. Engineers have developed many useful technologies for this purpose, but the companies must adhere to the laws. For example, emissions from cars have been reduced because cars now have catalytic converters that remove the poisonous gases from exhaust fumes.
Unfortunately, pollutants that contribute to acid rain may be carried hundreds of miles by wind before being deposited on the Earth. Because of this, it is sometimes difficult to determine the specific sources of these acid rain pollutants. For example, Canada has an acid rain problem because of manufacturing in the mid-western states of the U.S. Sulfur dioxides are produced in the industries in Ohio, Illinois and Pennsylvania and are carried over the land by the weather patterns. The acids then combine with rain over Canada and the Adirondack mountains in New York, making those lakes lifeless. Elsewhere, even though southern Norway has little heavy industry, 80% of its lakes are devoid of life or are on the critical list. Students can practice crafting their own case study for environmental policies using the Is That Legal? A Case of Acid Rain activity.
Associated Activities
- Acid and Base Rainbows - Students are introduced to the differences between acids and bases, and how to use indicators (pH paper and red cabbage juice) to distinguish between them.
- Acid Rain Effects - Students conduct a simple experiment to model and explore the harmful effects of acid rain (vinegar) on living (green leaf and eggshell) and non-living (paper clip) objects.
- Is That Legal? A Case of Acid Rain - In this literacy activity, students learn how persuasion techniques are used to develop an argument for or against a topic. Students learn how a case study is used as an analysis tool as they develop an environmental case study.
Lesson Closure
Gather together a small amount of vinegar, an eyedropper and a new stick of chalk. Place a few drops of vinegar at different places on the chalk stick and observe what happens. Challenge students to use what they have learned to explain why this happens. (Note: Vinegar is acetic acid and chalk is made of limestone.)
Even though acid rain is harmful, if acids ceased to exist, we would die! Why? Hydrochloric acid is found in the digestive fluid in the stomach, and it is what helps us digest food. Other acids and bases are very useful in our lives. Some bases are so strong that they open clogged drains or clean ovens without the need to scrub. Acids and bases are some of the most important substances on Earth!
Give the Not So Neutral Quiz. Review the answers during the next class period.
Vocabulary/Definitions
acid: A substance whose pH is less than 7.
acid rain: Any form of wet precipitation (rain, snow, fog, dew or sleet) that has a pH less than 5.6 (on a scale of 0 to 14, with 7 being neutral). Generally created by an interaction of coal and oil combustion products with water vapor in the atmosphere.
alkaline: Another term for a base.
arrhenius theory: States that acids ionize in water to produce H+ ions and that bases ionize to produce OH- ions.
base: A substance whose pH is greater than 7.
Bronsted-Lowery theory: States that acids donate protons and bases accept protons.
indicator: A tool used to measure/indicate the pH of a substance. A color change in the indicator corresponds to different pH levels.
Lewis theory: States that acid is an electron-pair acceptor and a base is an electron-pair donor.
litmus paper: An acid-base indicator made of paper infused with litmus (a water-soluble blue powder derived from certain lichens, a type of fungus) whose color changes depending on the pH of the substance with which it is in contact. There are type specific papers and wide range papers available.
neutral: A substance with a pH of 7.
pH: A measure of the acidity or alkalinity of a solution, numerically equal to 7 for neutral solutions, increasing with increasing alkalinity and decreasing with increasing acidity. The pH scale is used to describe the hydrogen ion (H+) concentration of an aqueous solution. [p(otential of) H(ydrogen)].
Assessment
Pre-Lesson Assessment
Brainstorming: As a class, have the students engage in open discussion. Remind students that in brainstorming, no idea or suggestion is "silly." All ideas should be respectfully heard. Encourage wild ideas and discourage criticism of ideas. Have them raise their hands to respond. Ask students to suggest definitions of the terms: pH, acid, base (alkaline) and neutral.
Question/Answer: Ask students questions and have them raise their hands to respond. Write their answers on the chalkboard.
- Where have you heard the term pH? Acid? Base (alkaline)? Neutral? (Possible responses: testing the pool/hot tub/aquarium, pH-balanced shampoo, deodorants, scrapbooking, etc.)
Post-Introduction Assessment
Voting: Ask a true/false question and have students vote by holding thumbs up for true and thumbs down for false. Count the votes and write the totals on the board. Give the right answer.
- True or False: Engineers often test substances by tasting them. (Answer: False. Scientists and engineers never test substances by tasting them. They use special paper that changes color depending on whether the substance is an acid or base [students can do this in the Acid (and Base) Rainbows activity], or they use electronic pH probes.)
Lesson Summary Assessment
Quiz: Give the students the Not So Neutral Quiz. Collect and grade. Return quizzes to students and review the answers during the next class period.
Concept Juggle: Have students stand in a circle and toss the ball to each other. Each time they toss the ball, have them name a base. One round can be "name a base," the next round can be "name an acid," "a cause or effect of acid rain," and so on.
Letter to the Industry: Have students write a letter to Unclean Industry, a paper manufacturing company that is not abiding by federal regulations of air pollutants released each year. Writing as engineers, have the students explain the harmful effects of acid rain in terms of pH and other observations from this lesson.
Homework
Internet Search: Research the history of acid rain. Include information on the causes of acid rain, when we first became aware of the problem, what problems have been caused by acid rain, what measures have been taken to combat acid rain. Has the situation improved? Lead a small discussion of class findings during the next class period.
Lesson Extension Activities
Research the history of acid rain. Include information on the causes of acid rain, when we first became aware of the problem, what problems have been caused by acid rain, what measures have been taken to combat acid rain. Has the situation improved?
Make a class mural to show the acid rain cycle.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn the basics of acid/base chemistry in a fun, interactive way by studying instances of acid/base chemistry found in popular films such as Harry Potter and the Prisoner of Azkaban and National Treasure. Students learn what acids, bases and indicators are and how they can be used, includi...

Students are introduced to the differences between acids and bases and how to use indicators, such as pH paper and red cabbage juice, to distinguish between them. They learn why it is important for engineers to understand acids and bases.

Students hypothesize whether vinegar and ammonia-based glass cleaner are acids or bases. They create designs on index cards using these substances as invisible inks.

Students are introduced to the primary types of erosion—chemical, water, wind, glacier and temperature. Students investigate examples of each erosion type and discuss how erosion changes the surface of the Earth.
References
Acid Rain Lesson Plan (Grades 6-8). Updated December 18, 2003. National Park Service. Originally found at: www.nature.nps.gov. Accessed August 16, 2004. (Activity adapted from this source.)
The Acid Rain Report. Acid Rain Report, Tripod and Angelfire. Accessed September 17, 2020. http://www.angelfire.com/ks3/acidrainreport/acid.html
Ebbing, Darrell D. General Chemistry. Fifth Edition. Geneva, IL: Houghton Mifflin Company, 1996.
Investigations in Science – Ecology. Huntington Beach, CA: Creative Teaching Press, 1995.
The pH Factor. Miami Museum of Science. Accessed September 17, 2020. www.miamisci.org/ph
Richardson, Lois. Acid Rain: The Disappearing Statue. Updated July 2, 2004. Lesson Plans for Teachers, TCEQ, Texas Natural Resource Conservation Commission. Accessed September 18, 2006. Originally found at: www.tceq.state.tx.us.
Sambal's Science Web. Gondar Design Science. Accessed August 16, 2004.
Silberberg, Martin S. Chemistry: The Molecular Nature of Matter and Change, New York, NY: McGraw – Hill Higher Education, 2000
ZoomSci (Activities from the show). PBS Kids, WGBH. Accessed September 17, 2020. Originally found at: www.pbskids.org. (Now archived at: https://www.pbslearningmedia.org/collection/zoom/)
Copyright
© 2004 by Regents of the University of Colorado.Contributors
Amy Kolenbrander; Janet Yowell; Natalie Mach; Malinda Schaefer Zarske; Denise CarlsonSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under a grant from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and National Science Foundation GK-12 grant no. 0338326. However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: May 21, 2021






User Comments & Tips