
Summary
Students use a thermal process approach to design, build and test a small-scale desalination plant that is capable of significantly removing the salt content from a saltwater solution. Students use a saltwater circuit to test the efficiency of their model desalination plant and learn how the water cycle is the basis for the thermal processes that drive their desalination plant.Engineering Connection
Environmental engineering is the application of science and engineering principles to improve air, water and land resources; provide healthy water, air, and land for human habitation; and remediate polluted sites. Environmental engineers work with other types of engineers to design and install water desalination plants around the world as our demand for clean water increases.
Learning Objectives
After this activity, students should be able to:
- Describe the thermal process of water desalination.
- Explain the engineering design process.
- Work on a team.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-3. Analyze data from tests to determine similarities and differences among several design solutions to identify the best characteristics of each that can be combined into a new solution to better meet the criteria for success. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Analyze and interpret data to determine similarities and differences in findings. Alignment agreement: | There are systematic processes for evaluating solutions with respect to how well they meet the criteria and constraints of a problem. Alignment agreement: Sometimes parts of different solutions can be combined to create a solution that is better than any of its predecessors.Alignment agreement: Although one design may not perform the best across all tests, identifying the characteristics of the design that performed the best in each test can provide useful information for the redesign process—that is, some of the characteristics may be incorporated into the new design.Alignment agreement: | |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-4. Develop a model to generate data for iterative testing and modification of a proposed object, tool, or process such that an optimal design can be achieved. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Develop a model to generate data to test ideas about designed systems, including those representing inputs and outputs. Alignment agreement: | Models of all kinds are important for testing solutions. Alignment agreement: The iterative process of testing the most promising solutions and modifying what is proposed on the basis of the test results leads to greater refinement and ultimately to an optimal solution.Alignment agreement: | |
International Technology and Engineering Educators Association - Technology
-
Make two-dimensional and three-dimensional representations of the designed solution.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Refine design solutions to address criteria and constraints.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Develop innovative products and systems that solve problems and extend capabilities based on individual or collective needs and wants.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
State Standards
Colorado - Science
-
Identify properties of substances in a mixture that could be used to separate those substances from each other
(Grade
7)
More Details
Do you agree with this alignment?
-
Develop and design a scientific investigation to separate the components of a mixture
(Grade
7)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- No. 10 tin can (this large size can is ~6 3/16-in diameter x 7-in height [15.72 cm x 17.78 cm] and is most commonly used by food service companies for tomatoes, ketchup, pie filling, refried beans, pizza sauce, etc.; ask for this size at restaurants or your school's cafeteria; Indian restaurants buy this size to make some of their food, so ask for their used cans before they throw them out; alternative: any other large tin can, such as a 2-lb coffee can)
- 1 piece of aluminum foil, 12 x 12 in (30 x 20 cm)
- 2 plastic containers (any type will do)
- 5-10 Popsicle sticks
- plastic soda bottle, 20 fl oz (566 ml) size
- hot plate
- 3-5 cups, 9 fl oz (266 ml) or 16 fl oz (473 ml) size
- eye protection (goggles or safety glasses)
- oven mitt, to safely move tin can on hot plate
- saltwater circuit, as created in the associated Saltwater Circuit activity
- Design Worksheet, one per team
- Reflection Worksheet, one per person
For the entire class to share:
- roll of aluminum foil
- salt, enough for 20-25 g per group
- water, enough for 32 fl oz (946 ml) per group
- duct tape
- masking tape
- measuring cups or graduated cylinders, to measure ml of water
- triple beam or digital scale, to measure grams of salt
- ice, enough for about 2-4 cups per group (expect to use 1 large bag of ice for two class periods; sometimes school cafeterias have an ice maker; otherwise bags of ice are available at grocery stores and gas stations)
- cooler, to keep the ice cold
- projector, to show the attached Water Desalination Plant Presentation PowerPoint
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/cub_desal_lesson01_activity2] to print or download.Introduction/Motivation
How much of the water on our planet do you think is available for drinking? (Give students a few seconds to think about this.) Of the total water available on our planet, less than 1% is safe for drinking. In fact, the world's water consumption rate is doubling every 20 years, outpacing by two times the rate of population growth. It is projected that by the year 2025, water demand will exceed supply by 56%.
Luckily for us, the oceans are full of water — except that we just can't fill a glass with ocean water and drink it! Engineers have been working on technologies to transform ocean water into drinking water. One technology they have created is based on a thermal process that is a controlled approximation to the naturally-occurring water cycle. What is really cool about this technology is that the water cycle occurs by nature on our planet and engineers have thought of ways to mimic this process on a smaller scale.
During the next few days, you are going to design and build a model system that takes in saltwater and removes as much of the salt as possible. You will also test the efficiency of your system using a saltwater circuit. We are going to use the engineering design process to help us create our desalination plant. Has anyone ever heard of the engineering design process? Do you know what it is? (Take suggestions from students.) It is a series of steps that engineers follow, kind of like how scientists follow the scientific method, but these steps are used to solve solving engineering design problems.
Procedure
Background
The thermal desalination approach involves heating saltwater until it evaporates and then forcing the evaporated water to condense. The condensed water is clean water. This type of process is used to produce more than 85% of the desalinated water in the world. This approach mimics the water cycle; in fact, each step in a thermal process can be linked to a stage of the water cycle (see the attached Water Desalination Plant Presentation PowerPoint).
In this activity, students design a model seawater desalination plant using a hot plate as the heat source (see an example in Figure 1).Refer to additional background information for this activity in the associated lesson, Ocean Water Desalination, and associated activity, Saltwater Circuit.
An important aspect of this project is to give students opportunities to make mistakes. It is common for students to not consider in their initial designs all aspects of how the desalination plant will ultimately work. But through a few design iterations, they improve their designs, until their plants work. This process mirrors the real world in which the steps of the engineering design process are followed and repeated to correct design deficiencies.
The basic steps of the engineering design process (sometimes called the engineering design loop) include: identify and understand the need, research the problem, brainstorm and develop possible solutions, select the most promising solution, construct a prototype/model, test and evaluate the prototype/model, and redesign and improve. A successful model or prototype serves as the mock-up for a full-scale product or system. Engineering teams often iterate a design many times before determining the final solution to a challenge. See more information on the engineering design process at https://www.teachengineering.org/k12engineering/designprocess.
Before the Activity
- Gather materials.
- Cut the aluminum foil into 12 x 12 in (30 x 30 cm) pieces, one per team. Cut a few extra sheets in case some teams are unable to build a working plant on their first try and need more than one piece.
- Make copies of the Design Worksheet (one per team) and the Reflection Worksheet (one per person).
- For Day 1, set up a projector to show the PPT presentation.
- For Day 2 and Day 3, have a cooler full of ice.
- Divide the class into groups of three or four students each.
With the Students—Day 1
- Show students the attached Water Desalination Plant Presentation (PowerPoint).
- Show students the materials available to build their model desalination plants. Have one or two sets available for them to examine. Hand out the Design Worksheets, one per team.
- Review the steps of the engineering design process (write them on the board).
- Give teams time to brainstorm ideas and come up with labeled diagrams and drawings of what they intend to create with the given materials. Encourage them to get several ideas down on scrap paper. Use the worksheet to document the final design.
- Review and approve designs at the end of class. Acceptable designs are detailed, labeled diagrams that show what the model plant will be made of and look like.
With the Students—Day 2
- Give teams time to construct their model desalination plants.
- Once a team has completed their model, have the team begin testing. Give each team 20 to 35 grams of salt, 32 fl oz (946 ml) of water and 2-4 cups of ice. (Use about a 3:1 ratio of water to salt. During the testing phase, set hot plates to their highest setting.)
- Once a team has its plant running, have team members take turns visiting other groups' stations to ask questions about their designs, and provide the comparative research information and ideas requested on page 3 of the Design Worksheet.
- After the desalination plants have been running for 20 minutes or so, have the team check to see if they are collecting water. Answer the questions on the Design Worksheet.
- If no water is collected, direct students to analyze their design and redesign with improvements.
- Before the class period is up, have students make notes on their design worksheet on what worked and what didn't. Let them know that the next class period will be the final class period to get a working desalination plant.
- For teams that successfully collect water, have them use their saltwater circuit to test the water for salt. If the light bulb turns on, then salt is present in the recovered water. If the light bulb does not turn on, then is no salt (or significantly reduced amount of salt) is present in the recovered water. Success!
- While going from team to team, note the kinds of designs and their major features, so as to prepare for the next class period when you will begin with an overview of different designs.
With the Students—Day 3
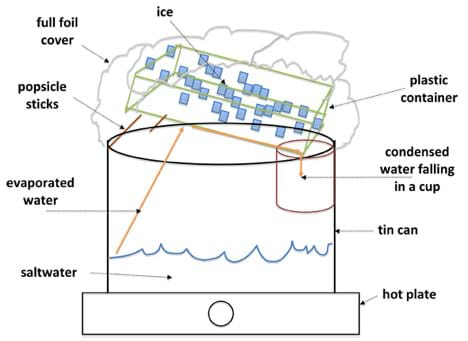
- To help students reach a working design, spend about 10 minutes at the beginning of the class drawing three different designs on the board and leading a class discussion about what would work and not work. You could use Figures 2 and 3 (one is workable, one is not); you could also incorporate student design examples observed from step 8 on Day 2.
- Give teams about five minutes to revise and improve their designs.
- Replenish any materials students may need and give students time to build and test their revised model desalination plants.
- It is possible that not all teams will achieve a working design by the end of Day 3. This does not mean the team has failed. In engineering --- much is learned from failures. Ask each team to write a summary paragraph describing their model plant and its performance.
- Conclude by having each student complete the Reflection Worksheet, as described in the Assessment section.
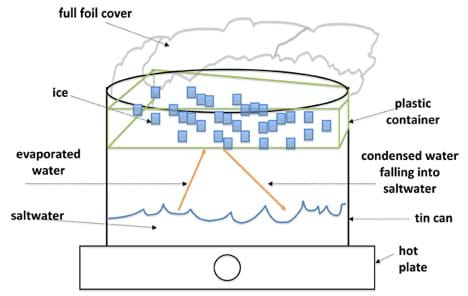
The design drawn in Figure 2 does not have a chance to work; the water that is evaporated by the hot plate condenses on the bottom of the plastic bin and falls back into the original saltwater solution. The design team failed to consider how to collect the water that condenses at the bottom of the plastic bin.
The design shown in Figure 3 could work; the water evaporated by the hot plate condenses at the bottom of an angled plastic container that directs the clean water into a collector cup.
Vocabulary/Definitions
engineering design process: A series of steps that engineering teams use to guide them as they solve problems. Sometimes called the engineering design loop.
input: The object(s) going into a system.
iteration: Repeating a series of steps to get closer to a desired outcome (that is, re-design, re-test, re-build to get nearer to an optimal engineering solution to a specific problem). Also: A version of the final product or solution. For example: Our third iteration passed the saltwater circuit test.
model: A small-scale object that represents another, often larger object. Often used in testing or perfecting the design of a final product or system.
output: The object(s) coming out of a system.
system: An object(s) that receives inputs and transforms them into output(s).
Assessment
Pre-Activity Assessment
Class Discussion: After showing the Water Desalination Plant Presentation PowerPoint, lead a class discussion.
Activity Embedded Assessment
Design Worksheet: Teams use the worksheet to sketch their design schematics and details, collect comparative information on the designs of other teams, and answer questions about their testing results.
Post-Activity Assessment
Team Writing Assignment: Whether or not teams achieved a working design, ask them to conclude their work by composing a summary paragraph describing their model plant and its performance.
Reflection Worksheet: By answering the four questions on this worksheet, students individually describe what they learned in this activity and how it can be improved. Review worksheets to gauge students' mastery of the subject matter. Questions: Describe the thermal water desalination processes; With which part(s) of the design process did you help? What did you learn in this activity? How would you change this activity for future classes?
Safety Issues
- Use eye protection during this activity.
- Hot plates can get extremely hot. Watch that students do not touch or throw water on them.
Activity Scaling
- For Lower grades, show a demonstration of the model desalination plant, test its water output with a saltwater circuit, and make comparisons to the natural water cycle.
- For upper grades, allow teams to choose the saltwater concentration for their model plant.
Additional Multimedia Support
Show students a two-minute video that describes the work General Electric has done in Africa to provide water for human use. See:
https://www.youtube.com/watch?v=USRRqmICTJE&feature=related
Show students a two-minute video of a water desalination plant in Israel. See:
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn about the techniques engineers have developed for changing ocean water into drinking water, including thermal and membrane desalination. They learn how processes can be viewed as systems, with unique objects, inputs, components and outputs, and sketch their own system diagrams to desc...

Students build a saltwater circuit, which is an electrical circuit that uses saltwater as part of the circuit. Students investigate the conductivity of saltwater, and develop an understanding of how the amount of salt in a solution impacts how much electrical current flows through the circuit.

Students examine in detail the water cycle components and phase transitions, and then learn how water moves through the human-made urban environment. Students show their understanding of the process by writing a description of the path of a water droplet through the urban water cycle, from the dropl...

Students learn about water bodies on our plant, including their uses and qualities. They learn several ways that engineers help people to maintain and conserve water sources.
References
Thirsty? How 'bout a cool, refreshing cup of seawater?, USGS Water Science for Schools, Updated March 29, 2010. U. S. Geological Survey, U.S. Department of the Interior. Accessed May 1, 2010. http://ga.water.usgs.gov/edu/drinkseawater.html
Wikipedia.org, Wikipedia Foundation Inc., Water Desalination. Accessed May 1, 2010. http://wikipedia.org
Copyright
© 2009 by Regents of the University of Colorado.Contributors
Juan Ramirez Jr.; Carleigh Samson; Stephanie Rivale; Denise W. CarlsonSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under a grant from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and National Science Foundation GK-12 grant no. 0338326. However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: June 30, 2020







User Comments & Tips