Quick Look
Grade Level: 7 (7-8)
Time Required: 1 hour
Lesson Dependency: None
Subject Areas: Life Science, Physical Science
NGSS Performance Expectations:

| MS-ESS2-4 |

Summary
Students learn about the techniques engineers have developed for changing ocean water into drinking water, including thermal and membrane desalination. They begin by reviewing the components of the natural water cycle. They see how filters, evaporation and/or condensation can be components of engineering desalination processes. They learn how processes can be viewed as systems, with unique objects, inputs, components and outputs, and sketch their own system diagrams to describe their own desalination plant designs.Engineering Connection
Environmental engineers apply science and engineering principles to improve air, water and land resources; to provide healthy water, air and land for human habitation; and to remediate polluted sites. With less than 1% of our planet's water safe for drinking, and the world's water consumption rate doubling every 20 years, it is projected that by 2025 water demand will exceed supply by 56%. To mediate this problem, engineers develop methods by which ocean water can be transformed into drinking water. Around the world, environmental engineers work together with other types of engineers to design and install water desalination plants.
Learning Objectives
After this lesson, students should be able to:
- Explain how the water cycle works.
- List technologies engineers have developed to desalinate ocean water.
- Be able to draw and interpret a system diagram.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ESS2-4. Develop a model to describe the cycling of water through Earth's systems driven by energy from the sun and the force of gravity. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This lesson focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Develop a model to describe unobservable mechanisms. Alignment agreement: | Water continually cycles among land, ocean, and atmosphere via transpiration, evaporation, condensation and crystallization, and precipitation, as well as downhill flows on land. Alignment agreement: Global movements of water and its changes in form are propelled by sunlight and gravity.Alignment agreement: | Within a natural or designed system, the transfer of energy drives the motion and/or cycling of matter. Alignment agreement: |
International Technology and Engineering Educators Association - Technology
-
Create an open-loop system that has no feedback path and requires human intervention.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Create a closed-loop system that has a feedback path and requires no human intervention.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Illustrate how systems thinking involves considering relationships between every part, as well as how the system interacts with the environment in which it is used.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
State Standards
Colorado - Science
-
Identify properties of substances in a mixture that could be used to separate those substances from each other
(Grade
7)
More Details
Do you agree with this alignment?
-
Develop and design a scientific investigation to separate the components of a mixture
(Grade
7)
More Details
Do you agree with this alignment?
Worksheets and Attachments
Visit [www.teachengineering.org/lessons/view/cub_desal_lesson01] to print or download.Introduction/Motivation
(Advance preparation: Have ready in the classroom a projector to show students the attached Water Desalination Presentation PowerPoint. Make copies of the attached Water Cycle Remix Worksheet, as a warm-up review of the water cycle as you start the lesson [or as a quick quiz], and the attached Design Your Own Desalination Plant Worksheet for the end of the lesson. See the Assessment section for details.)
(Begin by handing out the Water Cycle Remix Worksheet for students to complete first.)
How many times in a day do you go to a water faucet to get a drink? (Give students a minute to think about this.) At many places on our planet, people do not have this luxury. In fact, about 1.5 million people lack ready access to water every day. That's enough people to fill the 2008 Summer Olympic Birds Nest Stadium, a 91,000 seat stadium, about 16 times!
Do you think you could live your current lifestyle if you did not have access to clean water? Clean water is a part of our life from the moment we wake up in the morning until we go to bed at night. In addition to needing clean water to drink, we also need clean water to eat. Farmers use clean water to irrigate crops and feed livestock. Imagine if you were not able to have your favorite food at dinner; you might be very unhappy.
Does anyone have any ideas about where we could find a large volume of water that might be able to be turned into drinking water? (Give students a few minutes to think about this.) How about the ocean? Does anyone own the ocean? (Answer: No) Do you think you could just go out to the ocean and fill a bottle of water for drinking? (Answer: No) The ocean is full of saltwater. If you drink large quantities of saltwater, you become severely dehydrated. The more you drink, the thirstier you get. And, dehydration can lead to seizures, unconsciousness and brain damage, along with overtaxing your kidneys, which can be fatal.
Today we are going to learn how seawater can be turned into water suitable for drinking. At the core of this process is something called a filter. Has anyone ever used a filter? A filter is an object that separates components from a given mixture. Maybe you have used one in class or at home. (Give the students a few minutes to think about this.) Filters are in use all around us, every day. Whenever you drink filtered water, whenever you are in a car (air filters, oil filters, etc.), and whenever you use an air conditioner or furnace, you are benefiting from devices that at some point use a filter.
We are going to study filters that remove salt from saltwater. We are also going to learn about the way our planet naturally produces semi-clean water, and learn about technologies engineers have developed to turn saltwater into safe drinking water.
(Show the attached PowerPoint presentation. Then hand out the attached Design a Desalination Plant Worksheet). Following the lesson, conduct the hands-on associated activity Saltwater Circuit to have students learn about its real-world application as a desalination plant tool to test for the removal of salt from ocean water. After practicing the fundamentals of this process, students can then use the associated activity Water Desalination Plant to design, build and test a model saltwater desalination plant, using a saltwater circuit to test its success in removing salt from the water.
Lesson Background and Concepts for Teachers
Desalination Methods
To produce water suitable for drinking and irrigation, water desalination removes salt and minerals from seawater. The natural version of this process is the water cycle. Two fundamental human-made desalination methods are thermal technologies and membrane technologies.
Natural Water Desalination (the Water Cycle) — Water is the only substance that can exist as gas, liquid and solid in the naturally occurring temperature ranges on Earth. In fact, in some instances in the natural environment, water can be present in all three phases. The amount of water on our planet stays approximately constant, which means that the amount of water on our planet today is the same as the quantity present during the age of the dinosaurs. The water on Earth plays a major role in the naturally occurring phenomena called the water cycle, which can be described by four major steps: evaporation, condensation, precipitation and collection:
- Evaporation: During this stage, water from rivers, lakes and oceans transforms from liquid to water vapor, a gas. This transformation happens because the sun heats the Earth's surface, causing water to evaporate.
- Condensation: As water vapor rises into the Earth's atmosphere, it condenses on particles in the air. The water vapor condenses when air temperature decreases, as a result of higher elevation, forming clouds in the sky.
- Precipitation: As clouds become heavier, they release the condensed water particles, which fall back to the Earth as rain, snow or hail (depending on air temperature).
- Collection: The water that falls over land irrigates plants, trees and crops, and eventually makes its way back to rivers, lakes and oceans.
During the hydrologic cycle, water that evaporates from the ocean leaves its salt content behind because the salt in the water does not evaporate. So, the water cycle is, in essence, filtering saltwater, creating freshwater for the natural irrigation of plants, trees and crops.
Human-Made Desalination Systems — Engineers are challenged to develop cost effective ways to produce large quantities of water suitable for drinking and crop irrigation for regions of the world that suffer from water shortages. Water desalination is expensive, requiring large amounts of energy and specialized equipment to convert saltwater into drinking water. Currently, it costs $650 per acre-foot to processes seawater, compared to $200 per acre-foot to processes natural water for human consumption.
Thermal Desalination: This approach involves heating saltwater until it evaporates and then forcing the evaporated water to condense. The condensed water is clean water. This type of process is used to produce more than 85% of the desalinated water in the world. This approach mimics the water cycle; in fact, each step in a thermal process can be linked to a stage of the water cycle. An example:
- Multi-Stage Flash Distillation: Water is fed through a series of chambers, each at a different pressure. Water boiling in a high-pressure chamber is fed to a chamber of lower pressure, which causes the transferred water to evaporate quickly. Evaporation is quick because the boiling point of water at low-pressure is lower than the boiling point at high-pressure. The evaporated water is collected and condensed, producing clean water.
Membrane Desalination: This approach is the least expensive and works by forcing water through membrane sheets that prevent large saltwater molecules or ions from passing through the membrane. Two examples:
- Reverse Osmosis: Seawater is fed through a membrane at a pressure of 600 to 1000 psi (pounds per square inch). The membrane is designed in multiple layers in order to catch as many of the salt ions as possible.
- Electro-Dialysis: An electric potential is applied to a membrane that water is fed through. Since salt reduces to positively and negatively charged ions when dissolved in water, the electric potential causes the positive ions to travel towards the negative pole of the electric potential and the negative ions to travel towards the positive pole of the electric potential.
Both of these desalination techniques produce clean water for human consumption, but they also produce a waste product called brine. Brine is a sludge that has high concentrations of salt. As a result, the current desalination techniques are not 100% efficient.
Systems Interpretation
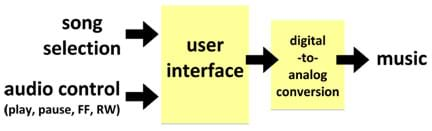
We can view processes, such as saltwater desalination, as objects composed of systems that perform certain tasks to achieve a desired goal. A system is an object that receives inputs and transforms it into output(s). Systems are all around us and can be viewed both high- and low-resolution levels. For example, when listening to music on an MP3 player, the user selects a song, hits play, and music comes out of the speakers; this is a low-resolution view of the system that produces music from audio data files (see Figure 1). A high-resolution view of the same system might be: the user selects a song, hits play, data is converted from digital to analog format, and music comes out of the speakers. This second description provides more information and is, therefore, considered a higher-resolution view of the system that produces music from audio data files (see Figure 2).
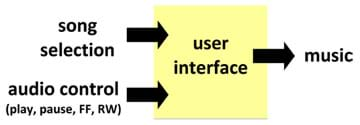
Both of these system descriptions can be easily sketched as a system diagram. A system diagram is a box diagram with labeled inputs and outputs (see Figures 1 and 2). System diagrams are typically a first step in engineering design and analysis, helping engineers get an understanding of the flow of a system.
Although the word "system" typically refers to something electrical or mechanical, systems are all around us. From the process in which you get dressed in the mornings, to the way you cook dinner in the evenings, any process can be viewed from a "systems perspective."
Associated Activities
- Saltwater Circuit - Students build a saltwater circuit and investigate the conductivity of water with different salt concentrations. They learn about its real-world application as a desalination plant tool to test for the removal of salt from ocean water.
- Water Desalination Plant - Students design, build and test a model saltwater desalination plant, using a saltwater circuit to test its success in removing salt from the water. They see how an engineering thermal desalination process mimics the natural water cycle.
Lesson Closure
So how dirty is dirty before you decide you cannot drink water? Does the water have to be clear or can it be kind of cloudy? (Give the students a minute to think about this.) At many places on our planet, people cannot go to a sink and fill a cup of water for drinking. Clean water is a precious and limited resource.
Can anyone tell me why we would be interested in removing salt from saltwater? (Possible answer: Many people face water shortages at the same time that so much water is available in our oceans — if we could only remove the salt.) We have lots of water in our oceans that cannot be used for human consumption. Can anyone tell me what might happen if someone drank ocean water? (Answer: That person would become dehydrated [and more thirsty!].) Since our planet has a fixed amount of water and the world's population continues to grow, we need to think about how to better use the resources available to us.
Engineers create technologies that help make the world a better place, and in the case of water desalination, they develop methods to make ocean water safe for drinking and other human uses. What are the two major technologies used to desalinate water? (Answer: Thermal and membrane.) If you think about it, the ideas behind these technologies came from the science concepts you learned in middle school. Figuring out how to produce clean water is a big challenge and one day you might be able to contribute to solving this problem. If you want to work on making the world a better place, then maybe engineering is for you.
Vocabulary/Definitions
brine: Water saturated with high quantities of salt.
dehydration: The removal of water from an object or organism. For people, refers to the abnormal loss of water from the body.
desalinate: To remove salts and other chemicals from (seawater or soil, for example). Also called desalinize.
desalination: Any process that removes salt from water. The term is usually applied to processes that remove salt from seawater to make it available for human consumption and agriculture.
filter: An object used to separate components in a given mixture.
seizure: When the brain tells muscles to move uncontrollably.
system: An object that receives inputs and transforms them into output(s).
system diagram: A diagram of the system that has all inputs and outputs labeled.
Assessment
Pre-Lesson Assessment
Water Cycle Remix: Have students complete the Water Cycle Remix Worksheet as a quick warm-up exercise on the water cycle. Or, use the worksheet as a quick quiz on the water cycle.
Post-Introduction Assessment
Class Discussion: While showing the Water Desalination Presentation (PPT), create an open environment in which students can ask questions and participate.
Lesson Summary Assessment
Design Your Own: To gauge students' understanding of the water desalination methods and of systems diagrams, have them complete the Design Your Own Desalination Plant Worksheet.
Lesson Extension Activities
Tomatoes and Salt: As an at-home activity, direct students to place a tomato on top of some paper towels or on a plate. Add a handful of salt on top of the tomato and observe what happens to the tomato after about 10 minutes. (Answer: After about 10-15 minutes, the tomato should be dry and the paper towel will be soaked with tomato juice. The salt removed water from the tomato cells and is an example of what would happen to human cells if one drinks large amounts of saltwater.)
Additional Multimedia Support
Show students a graphic of the water cycle at https://en.wikipedia.org/wiki/File:Water_cycle.png
Show students a four-minute news clip from Monterey, CA, describing offshore desalination technology. See: https://www.youtube.com/watch?v=GGUbRKf6aPY
Show students an animation of how seawater is converted into drinking water. See: https://www.youtube.com/watch?v=nbPNw3JaL7w&feature=related
This General Electric Co. website describes water desalination and the need for the development of this technology: https://www.suezwatertechnologies.com/images/multimedia/desal/index_flash.html
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students build a saltwater circuit, which is an electrical circuit that uses saltwater as part of the circuit. Students investigate the conductivity of saltwater, and develop an understanding of how the amount of salt in a solution impacts how much electrical current flows through the circuit.

Students learn about water bodies on our plant, including their uses and qualities. They learn several ways that engineers help people to maintain and conserve water sources.

Students use a thermal process approach to design, build and test a small-scale desalination plant that is capable of significantly removing the salt content from a saltwater solution. Students use a saltwater circuit to test the efficiency of their model desalination plant and learn how the water c...
References
Desalination, Existing facilities and facilities under construction. Wikipedia: the free encyclopedia. Accessed May 29, 2010. http://en.wikipedia.org/wiki/Desalination
Desalinize. Definition from Answers.com. Accessed May 29, 2010. http://www.answers.com/topic/desalinize
The Straight Dope: What would happen to you if you drank seawater? Published November 11, 2003. Straight Dope Science Advisory Board. Accessed May 29, 2010. http://www.straightdope.com/columns/read/2131/what-would-happen-to-you-if-you-drank-seawater
Texas Water. Texas A&M AgriLife. Water Resources Education, Texas A&M University. Accessed May 1, 2010. http://texaswater.tamu.edu/
Thirsty? How 'bout a cool, refreshing cup of seawater? USGS Water Science for Schools, Updated March 29, 2010. U. S. Geological Survey, U.S. Department of the Interior. Accessed May 1, 2010. http://ga.water.usgs.gov/edu/drinkseawater.html
Wikipedia.org, Wikipedia Foundation Inc., Accessed May 1, 2010. (Source of vocabulary definitions with some adaptation.) http://wikipedia.org
Copyright
© 2009 by Regents of the University of Colorado.Contributors
Juan Ramirez Jr.; Carleigh Samson; Stephanie Rivale; Denise W. CarlsonSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under a grant from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and National Science Foundation GK-12 grant no. 0338326. However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: June 24, 2019





User Comments & Tips