Summary
Students apply the concepts of conduction, convection, and radiation as they work in teams to solve two challenges. One problem requires that they maintain the warm temperature of one soda can filled with water at approximately human body temperature, and the other problem is to cause an identical soda can of warm water to cool as much as possible during the same 30-minute time period. Students design their engineering solutions using only common everyday materials, and test their devices by recording the water temperatures in their two soda cans every five minutes.
Engineering Connection
Engineers encounter problems of warming and cooling liquids in many situations. For prepared beverages, this might require maintaining specific cold or hot temperatures, but either way, the principles applied are the same. Students approach the activity challenges as if they were engineers, using heat transfer principles and the engineering design process to achieve their goals.
Learning Objectives
After this activity, students should be able to:
- Describe everyday examples of ways people try to cause or prevent heating and cooling by conduction, convection, and radiation.
- Give examples of materials that serve well for mechanisms of conduction, convection, and radiation.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-1. Define the criteria and constraints of a design problem with sufficient precision to ensure a successful solution, taking into account relevant scientific principles and potential impacts on people and the natural environment that may limit possible solutions. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Define a design problem that can be solved through the development of an object, tool, process or system and includes multiple criteria and constraints, including scientific knowledge that may limit possible solutions. Alignment agreement: | The more precisely a design task's criteria and constraints can be defined, the more likely it is that the designed solution will be successful. Specification of constraints includes consideration of scientific principles and other relevant knowledge that is likely to limit possible solutions. Alignment agreement: | All human activity draws on natural resources and has both short and long-term consequences, positive as well as negative, for the health of people and the natural environment. Alignment agreement: The uses of technologies and any limitations on their use are driven by individual or societal needs, desires, and values; by the findings of scientific research; and by differences in such factors as climate, natural resources, and economic conditions.Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-2. Evaluate competing design solutions using a systematic process to determine how well they meet the criteria and constraints of the problem. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Evaluate competing design solutions based on jointly developed and agreed-upon design criteria. Alignment agreement: | There are systematic processes for evaluating solutions with respect to how well they meet the criteria and constraints of a problem. Alignment agreement: | |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-4. Develop a model to generate data for iterative testing and modification of a proposed object, tool, or process such that an optimal design can be achieved. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Develop a model to generate data to test ideas about designed systems, including those representing inputs and outputs. Alignment agreement: | Models of all kinds are important for testing solutions. Alignment agreement: The iterative process of testing the most promising solutions and modifying what is proposed on the basis of the test results leads to greater refinement and ultimately to an optimal solution.Alignment agreement: | |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-PS3-4. Plan an investigation to determine the relationships among the energy transferred, the type of matter, the mass, and the change in the average kinetic energy of the particles as measured by the temperature of the sample. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Plan an investigation individually and collaboratively, and in the design: identify independent and dependent variables and controls, what tools are needed to do the gathering, how measurements will be recorded, and how many data are needed to support a claim. Alignment agreement: Science knowledge is based upon logical and conceptual connections between evidence and explanations.Alignment agreement: | Temperature is a measure of the average kinetic energy of particles of matter. The relationship between the temperature and the total energy of a system depends on the types, states, and amounts of matter present. Alignment agreement: The amount of energy transfer needed to change the temperature of a matter sample by a given amount depends on the nature of the matter, the size of the sample, and the environment.Alignment agreement: | Proportional relationships (e.g. speed as the ratio of distance traveled to time taken) among different types of quantities provide information about the magnitude of properties and processes. Alignment agreement: |
Common Core State Standards - Math
-
Reason abstractly and quantitatively.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Fluently add, subtract, multiply, and divide multi-digit decimals using the standard algorithm for each operation.
(Grade
6)
More Details
Do you agree with this alignment?
-
Recognize and represent proportional relationships between quantities.
(Grade
7)
More Details
Do you agree with this alignment?
-
Construct and interpret scatter plots for bivariate measurement data to investigate patterns of association between two quantities. Describe patterns such as clustering, outliers, positive or negative association, linear association, and nonlinear association.
(Grade
8)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Students will develop an understanding of the attributes of design.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop an understanding of engineering design.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop abilities to apply the design process.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Brainstorming is a group problem-solving design process in which each person in the group presents his or her ideas in an open forum.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Some technological problems are best solved through experimentation.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Apply the technology and engineering design process.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Refine design solutions to address criteria and constraints.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Develop innovative products and systems that solve problems and extend capabilities based on individual or collective needs and wants.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Use instruments to gather data on the performance of everyday products.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Create solutions to problems by identifying and applying human factors in design.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
State Standards
North Carolina - Math
-
Reason abstractly and quantitatively.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Fluently add, subtract, multiply, and divide multi-digit decimals using the standard algorithm for each operation.
(Grade
6)
More Details
Do you agree with this alignment?
-
Recognize and represent proportional relationships between quantities.
(Grade
7)
More Details
Do you agree with this alignment?
-
Construct and interpret scatter plots for bivariate measurement data to investigate patterns of association between two quantities. Describe patterns such as clustering, outliers, positive or negative association, linear association, and nonlinear association.
(Grade
8)
More Details
Do you agree with this alignment?
North Carolina - Science
-
Illustrate the transfer of heat energy from warmer objects to cooler ones using examples of conduction, radiation and convection and the effects that may result.
(Grade
6)
More Details
Do you agree with this alignment?
-
Understand characteristics of energy transfer and interactions of matter and energy.
(Grade
6)
More Details
Do you agree with this alignment?
-
Explain the suitability of materials for use in technological design based on a response to heat (to include conduction, expansion, and contraction) and electrical energy (conductors and insulators).
(Grade
6)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- 2 clean, empty, 12-ounce soda cans, ideally all the same type, plus 2 extra cans for controls
- laboratory thermometer (liquid immersion; accurate to 0.5 or 1°C)
- an assortment of "useful junk," such as fabric scraps (various sizes), socks from the lost & found, packing peanuts of several types, pieces of foam (various sizes), construction paper (both light and dark colors), bubble wrap, newspapers, quilt batting, old overhead transparencies, rubber tubing, drinking straws, funnels, aluminum foil, large zipper-type plastic bags, and anything else that might be used as insulating or conducting material, or to absorb or reflect radiation
- scissors, if possible
- glue, one bottle, if possible
- roll of tape, if possible
- (optional) timer or stopwatch
Pre-Req Knowledge
Students should have a good understanding of the concepts of conduction, convection and radiation, and be able to read a thermometer.
Introduction/Motivation
(The introduction and background information in the associated lesson, What's Hot and What's Not, provides adequate content introduction to this activity. Once students' understanding of the heat transfer concepts are verified, introduce the activity.)
Today you and your team will be acting as if you were engineers by applying your understanding of the concepts of conduction, convection, and radiation to a design challenge: To design and build devices that keep one can of water as warm as possible, while cooling another can of water as much as possible.
The cans will be filled with water that is at about 35 °C, which is very close to human body temperature. For 30 minutes, you will monitor the temperatures in each of your cans, recording the temperatures every five minutes. At the end of the 30 minutes, you will determine how much the temperature changed in each of the cans.
If you really understand what you learned about conduction, convection, and radiation, then the temperature in one of your cans should change very little, and the temperature in the other can should decrease a lot.
(If students need additional motivation, present the activity as a competition, with a small prize for the team that is best able to keep one can of water warm, and another small prize for the team that is able to cool one can of water the most.)
Procedure
Part 1: Introduce the Engineering Design Process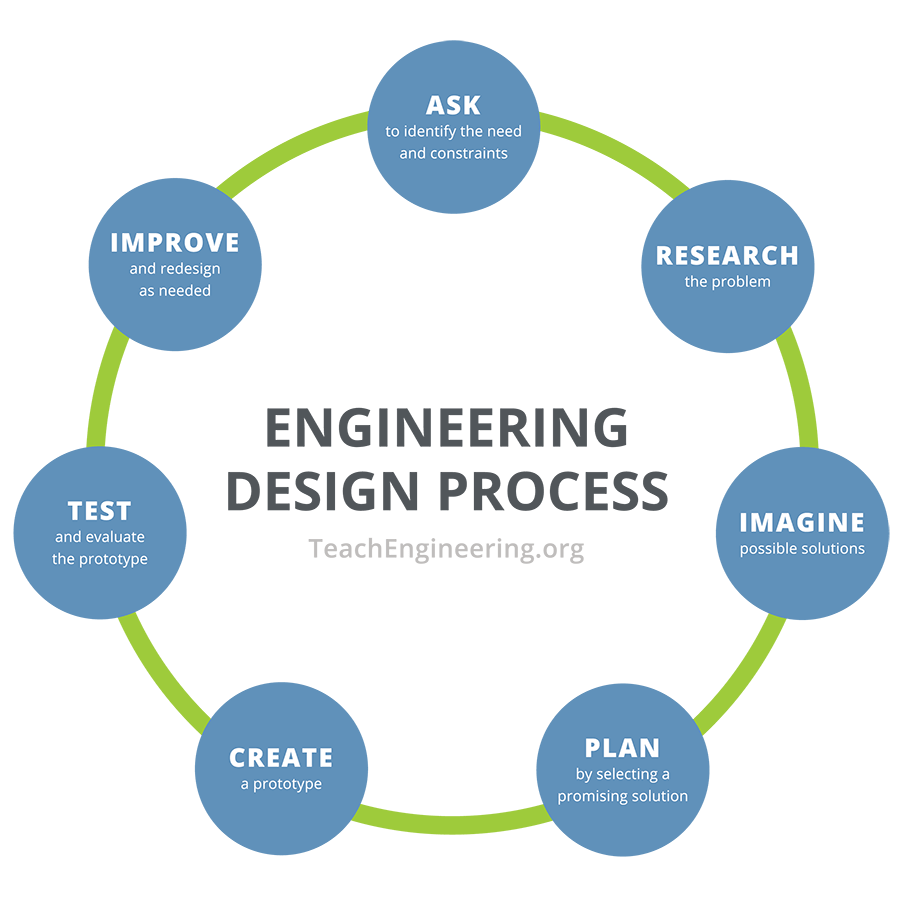
Part 2: Designing the warming and cooling devices
Assemble all the "useful junk" materials, as well as tools (scissors, tape, glue) at a table. Let students examine the materials, and then, working in teams of four students each, give groups 20 minutes complete the third, fourth, and fifth steps of the engineering design process to brainstorm, plan, and construct their cooling and warming devices. Allow students to use empty cans during the planning and construction phases, which they will need to be able to remove and replace with filled cans for the testing phase.
You may want to impose some limitations on the types and/or amounts of materials used, which is no different than the limitations placed on engineers in their real-world problem solving (often called design requirements and constraints). For example, it is a good idea to forbid human-made containers or devices such as insulated lunch boxes, thermoses, flashlights, classroom radiators or air conditioners. The intent is for students to "start from scratch" rather than use existing technology. Obviously, flames would not be allowed. You may or may not permit water to be used, and if so, designate what temperature the water may be, such as room temperature or 35 °C. Do not let students pour out their 35 °C water and replace it with hot or cold water from the tap! In fact, you might want to require that the original water must stay in the can for the duration of the experiment.
While students are designing and constructing, prepare the soda cans by filling a sink or large basin with water that is about 38-40 °C. Then add all the cans (two per team plus two extra) and make sure each is filled completely with the warm water. By the time students are ready to use the cans, the water will have cooled a few degrees, and thus at a good temperature for the activity.
Part 3: Keeping cans warm and letting them cool
After they have created their prototype, the students are ready to complete the sixth step of the engineering design process: test and evaluate prototype. When all groups are ready to test their warming-cooling devices, issue the water-filled cans and thermometers. If possible, give each team one thermometer for each can. If they need to share one thermometer between two cans, be sure they wait two minutes before reading the thermometer after switching between cans. Standard laboratory liquid thermometers that are accurate to 0.5 or 1°C are recommended. If possible, issue a timer to each team; otherwise, let students be responsible for keeping track of their five-minute intervals. Remind students to come up with an organized way to keep track of their time and temperature data.
In addition to the student test cans, fill two or three additional cans with the 35° water and leave them sitting undisturbed in a central location. These cans serve as controls; assign a student to record the temperatures in these cans at the same five-minute intervals.
Part 4: Data analysis and results presentations
Have students prepare line graphs of the temperature data for each of their two cans. The temperature of the water inside the cans should be on the y-axis (since it depended on how long the cans had been cooling) and time should be on the x-axis (since time always proceeds independently of anything else). Have students graph both sets of data on one set of axes, using different symbols or colored lines for each of their cans. They should also graph the temperature data from one of the two control cans on the same set of axes. This way they have a visual representation that not only compares how their "hot" and "cold" cans behaved, but also compares how well their warming and cooling arrangements performed compared to the "control" can that had had nothing done to it.
Then ask each group to make a brief presentation to the class, in which team members present their graphs and summarize the performances of their warming and cooling devices by calculating the temperature change of each during the 30 minutes and comparing these to the temperature changes in the control cans. As part of their presentations, team members should also show their warming and cooling devices or arrangements, and explain how they either tried to take advantage of conduction, convection and radiation, or how they tried to eliminate them. Students may have difficulty articulating the roles of conduction, convection, and radiation in their devices, so be prepared to help them understand these roles in their own and other teams' devices.
Expect that most of the successful warming devices students design will involve insulating cans with fabric, paper or some other material. Insulation serves to prevent air from moving over the surfaces of the can, thereby eliminating or greatly reducing its ability to lose heat by convection. We wear sweaters, fleece and down or fiber-filled coats in the winter for the same reason. Another method students might use is to insulate their cans and run a tube from next to the can, through the insulation, and out to the mouth of a student, whose job it is to breathe warm air through the tube for the duration of the experiment. Students using this arrangement are trying to prevent heat loss by convection, while at the same time using forced convection to carry heat to the can.
Other students might wrap their cans in dark fabric or paper and set them on a sunny windowsill, taking advantage of radiation. Once a group with a large, athletic male among its members, had the young man run around the outside of the building for five minutes in order to work up a sweat. Then they had him lie on a table with their can held firmly in his armpit! Their method was a very effective example of warming by conduction.
As for cooling devices, students might simply place their cans on the concrete floor in a cool corner of the classroom, and take advantage of conduction (from the warm can to the cool floor) and radiation (from the warm surface of the can to the cooler space surrounding it). They can enhance this method of cooling, however, by fanning the can with a piece of paper for forced convection. If the use of water is permitted, they might even wet their cans and fan them dry, an example of letting evaporative cooling take place, which might merit further discussion.
Take time between presentations, or as a wrap-up discussion, to clarify the ways that the groups applied the various heat transfer concepts, including the use of convection. Which teams used similar methods with different materials? In terms of meeting the design challenge, which were most successful at heating? Which were most successful at cooling?
Vocabulary/Definitions
conduction: The transfer of heat by molecular motion through a solid or a liquid, from a region of high temperature to a region of lower temperature.
convection: The movement of heated molecules of a gas or a liquid from a heat source to another area, due to density differences within the gas or liquid.
radiation: The transfer of heat energy by waves of visible or infrared light moving through space.
Assessment
Engineering Solutions Results Presentations: Have each group make a brief class presentation, presenting data graphs and summarizing the performances of their warming and cooling devices. Require the presentations to indicate the temperature gains/losses compared to the control cans, explain the design decisions made in the creation of their warming and cooling devices or arrangements, and explain how they tried to take advantage of conduction, convection and radiation, or how they tried to nullify them.
The performance of the devices or situations students design in order to keep one can of water warm while cooling the other can of water provides a means of assessing their abilities to understand and apply the ideas of conduction, convection and radiation. If the water in a team's "warm" can did not stay several degrees warmer than the water in the control cans, or if the water in their "cool" can did not reach a temperature several degrees lower than that of the control can, the students involved might benefit from further discussion of the concepts and how they can be applied to practical problems.
Investigating Questions
As students design and test their warming and cooling arrangements, ask questions to help them apply what they know about conduction, convection and radiation to the problems at hand, such as:
- Is this arrangement intended to help your can stay warm (or get cool) by conduction (or convection or radiation)? How does it do that?
As students test their arrangements, ask questions to help them begin analyzing their data, such as:
- How much has the water temperature changed so far?
- How does the temperature in your can compare to the temperature in the control cans? Does that mean that your arrangement is working (to keep your can warm, or help your can get cooler)?
Troubleshooting Tips
After five or 10 minutes, some students may find that the arrangements they devised, either to keep a can warm, or cause a can to cool, are not performing well. They may want to change their tactics immediately, and this is okay. In fact, real engineering solutions are seldom perfected on the first attempt, and mid-course corrections are commonplace. Direct them to make adjustments and improvements and begin the testing protocol again.
Check to make sure students are reading their thermometers accurately, especially if their warming or cooling devices seem to be performing exceptionally well.
Activity Extensions
After completing the activity and discussing the results, students may generate new ideas for approaches to keeping cans of water warm or causing them to cool. Permitting students to repeat the activity enables them to more fully experience the engineering design process, in which solutions are typically modified numerous times before a satisfactory design is achieved. If students repeat the activity, you may also want to give them the option of bringing materials in from home.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

With the help of simple, teacher-led demonstration activities, students learn the basic physics of heat transfer by means of conduction, convection and radiation. They also learn about examples of heating and cooling devices, from stove tops to car radiators, that they encounter in their homes, scho...

Students learn about the definition of heat as a form of energy and how it exists in everyday life. They learn about the three types of heat transfer—conduction, convection and radiation—as well as the connection between heat and insulation.

Students learn the scientific concepts of temperature, heat and the transfer of heat through conduction, convection and radiation, which are illustrated by comparison to magical spells found in the Harry Potter books.

With the help of simple, teacher-led demonstration activities, students learn the basic concepts of heat transfer by means of conduction, convection and radiation. Then they apply these concepts as they work in teams to solve two lab problems: 1) maintain the warm temperature of one soda can filled ...
Copyright
© 2013 by Regents of the University of Colorado; original © 2004 Duke UniversityContributors
Mary R. Hebrank , project writer and consultantSupporting Program
Engineering K-PhD Program, Pratt School of Engineering, Duke UniversityAcknowledgements
This content was developed by the MUSIC (Math Understanding through Science Integrated with Curriculum) Program in the Pratt School of Engineering at Duke University under National Science Foundation GK-12 grant no. DGE 0338262. However, these contents do not necessarily represent the policies of the NSF, and you should not assume endorsement by the federal government.
This activity was originally published, in slightly modified form, by Duke University's Center for Inquiry Based Learning (CIBL). Visit http://ciblearning.org/ for information about CIBL and other resources for K-12 science and math teachers.
Last modified: May 31, 2022








User Comments & Tips