Quick Look
Grade Level: 11 (10-12)
Time Required: 30 minutes
Lesson Dependency: None
Subject Areas: Chemistry

Summary
Students learn how to classify materials as mixtures, elements or compounds and identify the properties of each type. The concept of separation of mixtures is also introduced since nearly every element or compound is found naturally in an impure state such as a mixture of two or more substances, and it is common that chemical engineers use separation techniques to separate mixtures into their individual components. For example, the separation of crude oil into purified hydrocarbons such as natural gas, gasoline, diesel, jet fuel and/or lubricants.Engineering Connection
Almost all materials in the universe are found naturally in impure states such as mixtures of two or more substances. In chemistry and chemical engineering, a separation process is commonly used to transform a mixture of substances into two or more distinct materials. The separated products might differ in chemical properties or some physical property, such as size or crystal modification or other separation into different components. One example of separation application is crude oil, which is a mixture of various hydrocarbons. While valuable in this natural form, demand is greater for the various purified hydrocarbons such as natural gases, gasoline, diesel, jet fuel, lubricating oils, asphalt, etc. Chemical plants commonly have from 40% to 70% of both capital and operating costs in separations.[1] Since separations are ubiquitous in chemical plants and petroleum refineries, chemical engineers must be familiar with a variety of separation methods.
Learning Objectives
After this lesson, students should be able to:
- Define element, mixture and compound.
- Explain the differences between pure substances and mixtures.
- Explain the characteristics of the mixtures.
- Give some examples of elements, mixtures and compounds.
- Define the homogenious and heterogeneous mixtures and give some examples.
- Explain in general how mixtures can be separated.
- Name some separation techniques.
- Explain how chemical engineers apply these separation methods to purify various hydrocarbons such as natural gases, gasoline, diesel, jet fuel, lubricating oils, asphalt, etc., from raw crude oil.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
International Technology and Engineering Educators Association - Technology
-
Chemical technologies provide a means for humans to alter or modify materials and to produce chemical products.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Materials have different qualities and may be classified as natural, synthetic, or mixed.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
State Standards
Texas - Science
-
classify matter as pure substances or mixtures through investigation of their properties.
(Grades
10 -
12)
More Details
Do you agree with this alignment?
Worksheets and Attachments
Visit [www.teachengineering.org/lessons/view/uoh_sep_mixtures_less1] to print or download.Introduction/Motivation
Pure Substances vs. Mixtures
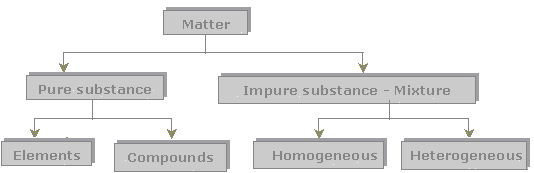
Everything around us is made of matter. Matter can be classified in to two broad categories: pure substances and mixtures.
Lavoisier, a French chemist, classified pure substances into elements and compounds on the basis of quantitative studies. He showed that when we heat mercuric oxide it changes into mercury and oxygen.[2]
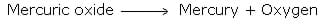
Mercuric oxide is a compound because it decomposes into simpler substances, whereas mercury and oxygen cannot be further decomposed into anything simpler as they are elements.
Pure Substances
1. Elements
All matter is composed of elements that are fundamental substances that cannot be broken down by chemical means. Element is defined as a substance that can not be further reduced as to simpler substances by ordinary processes. Another definition of element: a material that is composed of only one type of atom. The elements hydrogen, carbon, nitrogen and oxygen are the elements that make up most living organisms. Some other elements found in living organisms are magnesium, calcium, phosphorus, sodium and potassium.
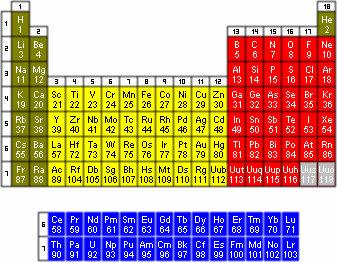
Of the 114 known elements, 92 occur in nature. By the late 1800s many elements had already been discovered. Dmitri Mendeleev, a Russian chemist, proposed an arrangement of known elements based on their atomic masses. The modern arrangement of the elements, the periodic table of elements, is a tabular display of the known chemical elements (see Figure 1) in which they are arranged according to their atomic numbers. The elements are arranged by electron structure so that many chemical properties vary systematically across the table. Each element is listed by its atomic number and chemical symbol.
2. Compounds
A compound is a pure substance composed of two or more different atoms chemically bonded to one another. That means that it can not be separated into its constituents by mechanical or physical means and only can be destroyed by chemical means. For example if we bring a magnet near a sample of iron sulphide, the iron present in the iron sulphide can not be separated.
Properties of a compound differ entirely from those of its constituent elements. Water is composed of hydrogen and oxygen. However, the properties of hydrogen and oxygen (both gases) are different from water (liquid). Hydrogen is combustible, oxygen is a supporter of combustion whereas water (made up of both hydrogen and oxygen) puts out a flame.
Energy changes are involved in the formation of a compound. For example, iron and sulphur react only when heat is supplied. The constituent elements in a compound are in a fixed proportion by weight. In water, hydrogen and oxygen are present in a fixed ratio of 1:8 by weight.
A compound is a homogeneous substance. That is, it is same throughout in properties and composition. Compounds also have fixed melting and boiling points.
Mixtures
A mixture is a material containing two or more elements or compounds that are in close contact and are mixed in any proportion. For example, air, sea water, crude oil, etc. The constituents of a mixture can be separated by physical means like filtration, evaporation, sublimation and magnetic separation. In the preparation of a mixture, energy is neither evolved nor absorbed. A mixture has no definite melting and boiling points. The constituents of a mixture retain their original set of properties. For example, sulphur dissolves in carbon disulphide and a magnet attracts iron filings. To help illustrate mixtures and different types refer to the associated activity Element, Mixture, Compound for students to discuss materials they use in their daily lives to gain a better understanding of pure substances vs. mixtures, and homogeneous vs. heterogeneous mixtures.
Examples of mixtures:
- Solid in liquid: Sugar and coffee
- Liquid in liquid: Water and alcohol
- Gas in liquid: Soda
- Gas in solid: Air entrapped in soil
- Gas in gas: Air containing hydrogen, oxygen, nitrogen, carbon dioxide, etc.
- Solid in solid: Metal alloys
1. Homogeneous Mixtures
The prefix "homo" indicates sameness. A homogeneous mixture has the same uniform appearance and composition throughout its mass. For example, sugar or salt dissolved in water, alcohol in water, etc.
2. Heterogeneous Mixtures
The prefix "hetero" indicates difference. A heterogeneous mixture consists of visibly different substances or phases. The three phases or states of matter are gas, liquid and solid. A heterogeneous mixture does not have a uniform composition throughout its mass.
Separation of Components of Mixtures
Most materials found in nature are in the form of mixtures. In engineering, a separation process is used to transform a mixture into two or more distinct products. This is done by considering that different components of the mixture may have different properties such as:
- size
- density
- solubility
- electrical charge
- boiling point
Depending on the raw mixture, various processes can be employed to separate the mixtures. Often, two or more of these processes must be used in combination to obtain the desired separation. In addition to chemical processes, mechanical processes are sometimes applied. In the example of crude oil, one upstream distillation operation feeds its two or more product streams into multiple downstream distillation operations to further separate the raw crude, and so on, until final products are purified.
Example separation techniques for mixtures:
- Filtration is used for the separation of solids from fluids (liquids or gases) by interposing a medium through which only the fluid can pass.
- Distillation for mixtures of liquids with different boiling points.
- Chromatography separates dissolved substances by different interaction with (that is, travel through) a material.
- Centrifugation and cyclonic separation, separates based on density differences.
- Drying, removes liquid from a solid by vaporization.
- Magnet separation technique uses magnet to separate iron particles from a mixture. (Refer to the associated activity Eat Iron?!! for a fun experiment to show students how the iron component of this heterogeneous mixture (cereal) retains its properties and can thus be separated by physical means.)
Chemical engineers use these separation techniques to purify naturally found substances or isolate them from other substances. For example, crude oil, also called petroleum, is a complex mixture of carbon and hydrogen (hydrocarbons) that exists as a liquid in the Earth's crust. Chemical engineers apply various distillation methods to purify various hydrocarbons such as natural gases, gasoline, diesel, jet fuel, lubricating oils, asphalt, etc., from the raw crude oil. Water purification is another good example of application of separation techniques.
Lesson Background and Concepts for Teachers
Several types of mixtures exist:
- Solutions are mixtures made by mixing a solute and a solvent, like salt in water. The solute is the substance that dissolves. The solvent is the substance that does the dissolving. Solutions are homogeneous.
- Suspensions are heterogeneous mixtures of a solid and a liquid in which the solid does not dissolve, like sand in water. Suspensions settle when left standing undisturbed.
- Emulsions are a special type of suspension. This mixture consists of two liquids that do not mix, like oil and water. Since the liquids do not mix, emulsions are heterogeneous. Emulsions settle into layers when they are left standing undisturbed.
- Colloidal Dispersions are mixtures with characteristics partway between a solution and a suspension, like mayonnaise. Colloidal dispersions may appear homogeneous but are actually heterogeneous. Colloidal dispersions do not settle when left standing undisturbed for a period of time.
Associated Activities
- Element, Mixture, Compound - Students discuss materials they use in their daily lives to gain a better understanding of pure substances vs. mixtures, and homogeneous vs. heterogeneous mixtures.
- Eat Iron?! - Students use strong magnets to find the element of iron in iron-fortified breakfast cereal flakes. They see how the iron component of this heterogeneous mixture (cereal) retains its properties and can thus be separated by physical means.
Vocabulary/Definitions
compound: A pure chemical substance consisting of two or more different chemical elements.
element: A substance consisting one type of atom.
heterogeneous mixture: A mixture that consists of visibly different substances or phases.
homogeneous mixture: A mixture that has the same uniform appearance and composition throughout its mass.
mixture: A substance consisting mixing two or more material.
solute: Material dissolved in a solution.
solution: A homogeneous mixture composed of two or more substances.
solvent: A liquid, solid, or gas that dissolves another solid, liquid, or gaseous solute.
Assessment
Post-Lesson Test: Administer the Mixtures Test to gauge student comprehension of the lesson content and concepts.
Lesson Extension Activities
Explain to students the distillation process for crude oil. Refer to the following source of information on the topic:
- How Oil Refining from Crude Oil Works http://science.howstuffworks.com/environmental/energy/oil-refining2.htm
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

To gain an understanding of mixtures and the concept of separation of mixtures, students use strong magnets to find the element of iron in iron-fortified breakfast cereal flakes. Through this activity, they see how the iron component of this heterogeneous mixture (cereal) retains its properties and ...

Students gain a better understanding of the different types of materials as pure substances and mixtures and learn to distinguish between homogeneous and heterogeneous mixtures by discussing an assortment of example materials they use and encounter in their daily lives.

This lesson plan introduces students to the properties of mixtures and solutions. It includes teacher instructions for a class demonstration that gives students the chance to compare and contrast the physical characteristics of some simple mixtures and solutions.

Students examine the periodic table and the properties of elements. They learn the basic definition of an element and the 18 elements that compose most of the matter in the universe. The periodic table is described as one method of organization for the elements.
References
Humphrey, J. L. and G. E. Keller II. Separation Process Technology. New York, NY: McGraw-Hill, 1997. (Industrially oriented book that includes performance, selection and scale up information)
Copyright
© 2013 by Regents of the University of Colorado; original © 2010 University of HoustonContributors
Parnia Mohammadi; Roberto DimaliwatSupporting Program
National Science Foundation GK-12 and Research Experience for Teachers (RET) Programs, University of HoustonAcknowledgements
This digital library content was developed by the University of Houston's College of Engineering under National Science Foundation GK-12 grant number DGE-0840889. However, these contents do not necessarily represent the policies of the NSF and you should not assume endorsement by the federal government.
Last modified: July 1, 2019






User Comments & Tips