Quick Look
Grade Level: 11 (9-12)
Time Required: 45 minutes
Expendable Cost/Group: US $2.00 This activity uses one or two non-expendable (reusable) air quality monitors, which can be rented; see the Materials List and Other sections for details.
Group Size: 4
Activity Dependency:
Subject Areas: Earth and Space, Science and Technology

Summary
Students use next-generation air quality monitors to measure gas-phase pollutants in the classroom. They apply the knowledge they gained during the associated lesson—an understanding of the connection between air pollutants and their possible sources. Student teams choose three potential pollutant sources and predict how the monitor’s sensors will respond. Then they evaluate whether or not their predictions were correct, and provide possible explanations for any inaccuracies. This activity serves as a simple introduction to the low-cost air quality monitoring technology that students use throughout the associated activities that follow. Three student handouts are provided.Engineering Connection
During this activity, students investigate as if they were engineers, probing the link between sources and pollutants. By examining what pollutants are present in the air as well as possible sources, environmental engineers can determine where pollution is coming from, which is the first step towards control or mitigation strategies. Students also benefit from the hands-on learning practice of beginning with an action—measuring the gases—and then seeing the data plotted on a screen. In this way, students (and engineers) make the invisible visible and quantifiable.
Learning Objectives
After this activity, students should be able to:
- Explain the different sources of CO2 (biological and combustion), VOCs (combustion and volatilization), and NO2 (combustion).
- Use a simple air quality monitor (such as a Pod) to collect data.
- Analyze and interpret data qualitatively using plots.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
-
Analyze data using tools, technologies, and/or models (e.g., computational, mathematical) in order to make valid and reliable scientific claims or determine an optimal design solution.
(Grades 9 - 12)
More Details
Do you agree with this alignment?
Common Core State Standards - Math
-
Represent data with plots on the real number line (dot plots, histograms, and box plots).
(Grades
9 -
12)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Assess a technology that minimizes resource use and resulting waste to achieve a goal.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
State Standards
Colorado - Science
-
The interaction of Earth's surface with water, air, gravity, and biological activity causes physical and chemical changes
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Analyze and interpret data, maps, and models concerning the direct and indirect evidence produced by physical and chemical changes that water, air, gravity, and biological activity create
(Grades
9 -
12)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- pens or pencils
- Student Data Sheet, one per student
- Everyday Exposure Worksheet, one per student
- (optional) Connecting to the Big Picture Handout, one per student
To share with the entire class:
- 2 air quality monitors, such as a rented AQ-IQ Kit (containing 2 Pods, which are low-cost air quality monitors) from the University of Colorado Boulder, or another low-cost monitor with multiple gas-phase sensors; see the Other section for details on obtaining the Pod or alternate monitors
- pollutant sources; for a class of 28 students, provide about 10 items (2 of each pollutant type) that give off combustion byproducts or VOCs, such as lighters, candles, rubbing alcohol, wooden coffee stir sticks to burn, super glue, a Bunsen burner to burn natural gas, human breath, etc.
- safety glasses, enough for two teams of students testing with the two Pods
- computer and projector, for the teacher to plot and display class data
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/cub_airquality_lesson01_activity1] to print or download.Pre-Req Knowledge
Basic air quality background information on gas-phase pollutants and some of their sources, such as provided in the associated lesson, An Introduction to Air Quality Research.
Introduction/Motivation
Now you have the chance to test out what you learned about air quality monitoring during the associated lesson. Do you remember how we talked about what is in a cloud of smoke billowing from a diesel semi-truck? We know the emissions include carbon dioxide, but can we see it? (Listen to student responses.) No, because it is an invisible gas, right? Remember, all we can see is the particulates, and on cold days, the water vapor.
Today we will use air quality monitors in order to “see” some of the gases that we have discussed. In your groups, you will choose one item from an assortment of possible pollution sources, predict how the sensors will respond, and then we will plot all the data and discuss as a class what happened.
Procedure
Teacher Tips
Familiarize yourself with the air quality monitor you will be using and how to view data from it. If using a Pod air quality monitor, go through the user manual to understand how to run the monitors and check the data. It is a good idea to practice these steps before using the monitors with your class. The associated lesson, An Introduction to Air Quality Research, provides sufficient scientific background for teaching this activity.
Before the Activity
- Gather tools and supplies—the air quality monitors and pollutant sources.
- Make copies of the Student Data Sheet and Everyday Exposure Worksheet. The data sheet guides students through the experimental portion of the lab. The worksheet is suitable for students to complete during the 10-15 minutes when the teacher downloads and plots the data, or as a homework assignment. If you did not use the Connecting to the Big Picture Handout as a homework assignment with the associated lesson, you may want to also make copies of this handout and assign it as homework at activity end.
- If using a Pod, plug it in for 30 minutes prior to the activity to allow the sensors to warm up. Following the user instructions, practice taking, downloading and plotting some sensor data once before class, to make sure everything works.
- Set up a computer with a projector to display the plotted data for a class discussion.
- The procedures below describe doing the entire activity during one class period (collecting, plotting and analyzing Pod air quality monitor sensor data). Alternatively, you could split the activity between two class periods, which gives the teacher time to download and plot the data between class periods (takes 10-15 minutes), and then use the second class period to focus us on a class discussion of the results and data sheet reflection question completion.
With the Students: Experimental Data Collection
- Hand out the data sheets.
- Divide the class into groups of four to six students each (adjust accordingly, depending on your class size).
- Present to the class the Introduction/Motivation content.
- Direct students to read the instructions on the data sheet, choose the pollution sources they will use and make predictions.
- Give teams about 10 minutes for discussion and planning time within their groups.
- Direct groups to start running their sets of three experiments (that is, exposing the pollution sources to the air quality monitors).
- As needed, assist groups and answer questions while they run their experiments.
- Assist students with source placement. It is best if sources are placed close to the inlet and a little below, since emissions (particularly from combustion) rise; see Figure 1.
- Advise students to give the sensors 2-4 minutes to respond to each source.

With the Students: Data Plotting and Analysis
- Download and plot the data from the Pod (allow 10-15 minutes), following the user manual instructions. Do this in front of the entire class. If desired, hand out the worksheet for students to complete while the class data is being downloaded and plotted.
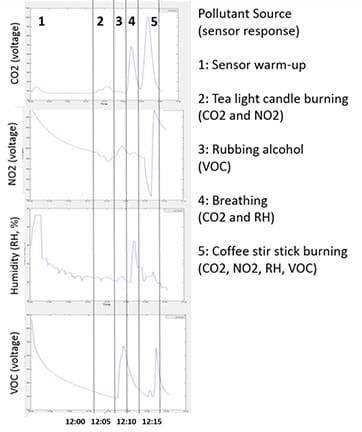
- Project the data (one Pod at a time) for the entire class to see. Work through the following questions and prompts. Figure 2 provides sample data and is the example referred to below.
A. Examining one sensor at a time (CO2, NO2 and VOCs), discuss as a class the overall trends. Note that in the Figure 2 example, section 1 is the warm-up period. Possible questions:
- When was the sensor warming up?
- When did we begin the activity?
- Compare the time you started the activity with the time shown in the plot.
B. Look for peaks associated with particular times and ask students what they believe was happening based on the actions recorded on their data sheets. Example questions based on the Figure 2 example data:
- Can anyone explain this spike in CO2 that occurs between 12:10 and 12:15? (Answer: This is the spike from breathing on the sensor; we also see a spike in relative humidity at this point, also from breathing.)
- What was happening at 12:10 vs. 12:15, because the VOC spike appears larger at time 12:10? (Answer: The first spike is from the rubbing alcohol and the second is from burning the wooden coffee stir stick; note that different VOCs and different amounts of VOCs are affecting the difference in sensor response during the two periods.)
C. Figure 2 provides example data along with explanations of the corresponding activities that generated that data. All of these plots were generated by uploading the raw data file to the plotting software. Note that the raw NO2 sensor signal responds inversely, so expect to see a dip in the raw signal if NO2 is present. Additional Figure 2 explanatory notes:
- Phase 1: Shows data from when the sensor was warming up.
- Phase 2: Shows data from a burning candle. Notice a small response from the CO2, and a matching inverse response from the NO2 sensor.
- Phase 3: Shows data from rubbing alcohol fumes. Notice a strong response from the VOC sensor, NO2 is recovering, and no response from the rest of the sensors.
- Phase 4: Shows data from breathing. Notice a strong response from the CO2 and RH sensor, while the VOC sensor recovers.
- Phase 5: Shows data from burning the wooden stir stick. Notice a strong response from CO2 and NO2, small response from RH, and delayed response from the VOC sensor.
- After the class-wide discussion, direct students to complete the data table by filling in the “Actual Response” from each sensor, and then answer the Reflection Questions at the end of the data sheet.
- Assign students a worksheet or handout as homework, as described in the Assessment and Activity Extension sections.
Vocabulary/Definitions
carbon dioxide: A colorless and odorless gas. A gas-phase pollutant. Composed of 1 carbon atom and 2 oxygen atoms. Generated by the respiration of animals and the combustion (burning) of fuels that contain carbon. Abbreviated as CO2.
carbon monoxide: A product of incomplete combustion that is deadly to humans in large quantities.
nitrogen dioxide: A gas-phase compound made of 1 nitrogen atom and 2 oxygen atoms. It is formed during high-temperature combustion from the nitrogen that exists in the air. High-temperature combustion also produces nitrogen monoxide (NO). The sum of the amount of NO and NO2 is the amount of NOx present; in other words NOx is a term that includes both NO and NO2.
particulate matter: Solid or liquid particles suspended in the air.
time series plot: A graph with data plotted over time that is used to evaluate patterns and behavior in data over time. In this activity, students use plots of raw sensor data over time for their analyses.
volatile organic compound: An organic chemical that has a high vapor pressure at ordinary room temperature, such that it volatizes (enters the gas phase) at room temperature and pressure. An example is formaldehyde (CH2O, 1 carbon, 2 hydrogens, and 1 oxygen atom). Abbreviated as VOC. VOCs are also gas-phase compounds. VOCs also include products of incomplete combustion (when a carbon-fuel is not completely burned, resulting in only CO2).
Assessment
Pre-Activity Assessment
Predictions: Before collecting data, have students complete in their groups the first two columns in the table on the Student Data Sheet—Source and Application Notes, and Predicted Response. Expect this to generate discussion around what pollutants are emitted by each source.
Activity Embedded Assessment
Data Collection & Reflection: During the activity, have students record their data on the Student Data Sheet and then answer the Activity Reflection questions on the sheet. Review the predictions, data, observations and answers on students’ completed data sheets to evaluate their understanding of the experiment concepts, procedure and significance.
Post-Activity Assessment
Informal Class Discussion: Review students’ responses to the Reflection Questions on the Student Data Sheet through an informal class discussion and ask what they learned from the activity.
Homework
Everyday Connections: After the activity, assign the Everyday Exposure Worksheet as homework to test students’ application of learned concepts and connect them to their own lives. The worksheet asks students to identify three sources of pollution that they routinely encounter, name the likely pollutants associated with them, and rank them according to the estimated amount of exposure. Alternatively, assign students to complete this worksheet while the teacher is downloading and plotting the data during the activity.
Safety Issues
During this activity, use eye protection, such as safety glasses.
When burning pollutant sources, take precautions such as tying back hair, rolling up long sleeves, conducting the activity under a fume hood if available, not letting flames get out of control, being aware of any smoke, and making sure that everything is completely extinguished.
Avoid inhaling any combustion products or vapors from any pollutant sources.
Troubleshooting Tips
If students are not careful about source placement near the Pod inlet, or do not leave the sources at the inlet long enough, no response may be recorded by the sensors. Classroom sources are unlikely to overwhelm the sensors, so inform students to not worry about this and be cognizant of getting the emissions into the monitor inlet.
To be prepared for any technical difficulties during class, you may want to conduct the activity on your own and create a set of backup data that you download and plot so it is ready to display. Then, if needed, pull up the sample data and tell students the pollutant sources that you used and have them guess when they occur in the time series plot.
Activity Extensions
Take-Home Handout: If you did not use the three-question Connecting to the Big Picture Handout as part of the associated lesson, consider using this handout as a homework assignment if you used the Everyday Exposure Worksheet during the downloading/plotting wait time during the activity. The questions are open-ended and intended to prompt students to reflect on connections between what they are learning and everyday life. If students are completing the entire AQ-IQ curriculum, these questions are recommended to help them begin to think about topics they may wish to focus on for their projects. If possible, take 10 minutes at the beginning of the next class period to discuss their answers to these questions.
Activity Scaling
For more advanced students, let them open the collected Pod data in a text file and import it into Microsoft® Excel®. Using the spreadsheet application, students can plot CO2, NO2, and VOCs against time. Note that this takes a much longer time, but is possible.
Additional Multimedia Support
Software to aid in plotting and examining the monitor data is explained in the Pod user manual. All downloads and additional assistance/information are available at https://www.colorado.edu/aqiq/. Alternatively, use Microsoft® Excel® to plot and visualize the data.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn the basics about the structure of the Earth’s atmosphere, the types of pollutants that are present in the atmosphere (primary, secondary, gas-phase compounds, particulate matter), and the importance of air quality research. They are also introduced to some engineering concepts such as...

Students are introduced to the concepts of air pollution, air quality, and climate change. The three lesson parts (including the associated activities) focus on the prerequisites for understanding air pollution. First, students use M&M® candies to create pie graphs that express their understanding o...
Other Related Information
AQ-IQ Air Quality Monitor Information
This curriculum was designed to support high school students’ use of a low-cost air quality monitor developed by the Hannigan Lab at the University of Colorado Boulder called a “Pod.” Pods can be rented and shipped from the university; see below for details. Alternatively, many of the activities, including the long-term project, can be completed with other air quality monitors—or no monitor. For example, students can design research projects that utilize existing air quality data instead of collecting their own, which is highly feasible since a great amount of data from around the planet is publically available. In addition, other low-cost monitors could be used instead of the Pods, ranging from purchasable to DIY; see the AQ-IQ unit for a list of options.
Is an air quality monitor needed for this activity?
Yes, this activity requires an air quality monitor such as the Pod, although a substitute monitor (see examples listed on the AQ-IQ unit) could also be used. Essentially you just need the ability to measure multiple pollutants.
Obtaining an AQ-IQ air quality monitor (Pod) from the University of Colorado Boulder
The air quality monitors—called Pods—are available in AQ-IQ Kits that can be rented from the Natural History Museum at the University of Colorado Boulder.
An AQ-IQ Kit is an easily transportable carry-on suitcase-sized container that holds two portable air quality monitors (= 2 Pods), a small laptop for processing data, a comprehensive user manual, and accessories such as power cords and batteries for portable monitoring. The cost is a $10 per week rental fee that supports museum expenses to store and rent the kits. Generally, if students are completing a long-term project, expect to rent a kit for 3-8 weeks.
To find out about the availability of AQ-IQ Kits and shipping options, or to schedule a rental or to rent the kits, contact the museum’s education office at https://www.colorado.edu/cumuseum/programs/schools-and-groups/outreach-materials (a phone number and email address are provided). After checking out the kits, the museum can connect you to a mechanical engineering lab at the university as a technical support resource for using the air quality monitors, troubleshooting and conducting student air quality research projects (as needed). Sometimes the lab partners with schools and provides university undergraduate students to assist teachers with technical issues and to mentor and assist high school students throughout their projects. See the AQ-IQ Program website to learn more and to contact them: https://www.colorado.edu/aqiq/.
Copyright
© 2013 by Regents of the University of ColoradoContributors
Ashley Collier; Katya Hafich; Daniel Knight; Michael Hannigan; Joanna Gordon; Ben Graves; Eric Ambos; Olivia Cecil; Victoria Danner; Erik Hotaling; Eric Lee; Drew Meyers; Hanadi Adel Salamah; Nicholas VanderKolkSupporting Program
AirWaterGas SNR Project Education and Outreach, College of Engineering, University of Colorado BoulderAcknowledgements
This material is based upon work by the AirWaterGas Sustainability Research Network Education and Outreach Project in the College of Engineering at the University of Colorado Boulder, supported by National Science Foundation grant no. CBET 1240584. However, these contents do not necessarily represent the policies of the National Science Foundation, and you should not assume endorsement by the federal government.
The authors also express their appreciation for the support of the University of Colorado’s Office of Outreach and Engagement.
Last modified: August 22, 2020




User Comments & Tips