Quick Look
Grade Level: 7 (6-8)
Time Required: 1 hours 45 minutes
Note: 50 minutes for the initial activity, two days (48 hours) for incubation of Petri dishes and 50 minutes to record and analyze results
Expendable Cost/Group: US $4.00
Group Size: 2
Activity Dependency: None
Subject Areas: Biology, Chemistry, Life Science
NGSS Performance Expectations:

| MS-LS1-1 |
Summary
Students culture cells in order to find out which type of surfactant (in this case, soap) is best at removing bacteria. Groups culture cells from unwashed hands and add regular bar soap, regular liquid soap, anti-bacterial soap, dishwasher soap, and hand sanitizer to the cultures. The cultures are allowed to grow for two days and then the students assess which type of soap design did the best job of removing bacteria cells from unwashed hands. Students extend their knowledge of engineering and surfactants for different environmental applications.
Engineering Connection
Engineers spend a lot of time trying to prevent the spread of bacteria cells that make humans sick. One example of products designed by engineers is surfactants or soaps that remove bacteria cells from our hands. Surfactants are compounds that act as wetting agents to decrease the interface tension of two liquid, such as oil and water. Engineers design surfactants for a variety of applications including detergents, emulsifiers, paints, adhesives, agrochemicals, biocides, and soil and water remediation.
Learning Objectives
After this activity, students should be able to:
- Describe how surfactants work to remove surface tension between two compounds.
- Understand how engineers use processes to evaluate the effectiveness of different designs.
- Describe the effect of adding surfactant to a prokaryotic cell.
- List several engineering applications of surfactants in everyday life.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-LS1-1. Conduct an investigation to provide evidence that living things are made of cells, either one cell or many different numbers and types of cells. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Conduct an investigation to produce data to serve as the basis for evidence that meet the goals of an investigation. Alignment agreement: | All living things are made up of cells, which is the smallest unit that can be said to be alive. An organism may consist of one single cell (unicellular) or many different numbers and types of cells (multicellular). Alignment agreement: | Phenomena that can be observed at one scale may not be observable at another scale. Alignment agreement: Engineering advances have led to important discoveries in virtually every field of science, and scientific discoveries have led to the development of entire industries and engineered systems.Alignment agreement: |
Common Core State Standards - Math
-
Fluently divide multi-digit numbers using the standard algorithm.
(Grade
6)
More Details
Do you agree with this alignment?
-
Fluently add, subtract, multiply, and divide multi-digit decimals using the standard algorithm for each operation.
(Grade
6)
More Details
Do you agree with this alignment?
-
Find a percent of a quantity as a rate per 100 (e.g., 30% of a quantity means 30/100 times the quantity); solve problems involving finding the whole, given a part and the percent.
(Grade
6)
More Details
Do you agree with this alignment?
-
Summarize numerical data sets in relation to their context, such as by:
(Grade
6)
More Details
Do you agree with this alignment?
-
Reporting the number of observations.
(Grade
6)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Students will develop an understanding of the relationships among technologies and the connections between technology and other fields of study.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Advances and innovations in medical technologies are used to improve healthcare.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
State Standards
Colorado - Math
-
Fluently add, subtract, multiply, and divide multidigit decimals using standard algorithms for each operation.
(Grade
6)
More Details
Do you agree with this alignment?
-
Fluently divide multi-digit numbers using standard algorithms.
(Grade
6)
More Details
Do you agree with this alignment?
-
Find a percent of a quantity as a rate per 100.
(Grade
6)
More Details
Do you agree with this alignment?
-
Report the number of observations.
(Grade
6)
More Details
Do you agree with this alignment?
-
Summarize numerical data sets in relation to their context.
(Grade
6)
More Details
Do you agree with this alignment?
Colorado - Science
-
Gather, analyze, and interpret data and models on the different types of cells, their structures, components and functions
(Grade
7)
More Details
Do you agree with this alignment?
-
Employ tools to gather, view, analyze, and report results for the scientific investigations of cells
(Grade
7)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- log sheet
- 2 sterile cotton swabs
- 2 Petri dishes with nutrient media (Note: the Petri dishes are sold in packs of 10 through Carolina Biological. If you have access to an autoclave (a machine that sterilizes solutions and tools using high temperature and pressure), it is less expensive to make your own media. However, it is much easier and less time consuming to buy the already-prepared nutrient media.)
- Sudsy Cells Worksheet
To share with the entire class:
- bar soap (any type; least expensive is okay)
- liquid hand soap (any type; least expensive is okay)
- liquid dish soap (any type; least expensive is okay)
- anti-bacterial soap (any type; least expensive is okay)
- hand sanitizer (any type; least expensive is okay)
- magnifying glass (optional)
- bowl of water (for teacher demonstration)
- pepper shaker (for teacher demonstration)
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/cub_cells_lesson01_activity1] to print or download.Pre-Req Knowledge
The students should know the parts of a prokaryotic cell. They should also be able to calculate averages and percentages.
Introduction/Motivation
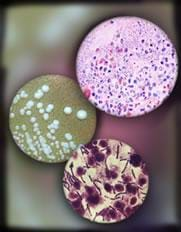
Today we're going to learn more about one way that engineers help protect humans and the environment from harmful bacteria. Why do we wash our hands? We wash our hands to get rid of bacteria that we may have picked up that could make us sick. What do we use to wash our hands? Soap! What type of soap do you use? Many different types of soap are available: bar, liquid, anti-bacterial, and even special soaps for medical people to use before they perform surgery. Engineers are involved in designing the soap products that we use every day. Chemical engineers look at different mixtures of chemical compounds, called surfactants, and design products that are suited for different situations, from hand washing to medical cleansers to washing the oil out of the ocean after an oil spill.
Surfactants, or surface acting agents, are compounds that reduce the interface tension between water and an organic compound, such as oil. Basically, adding a surfactant to a solution of oil and water, breaks down the barriers between the oil and the water, allowing them to mix. Surfactants have very special chemical properties that help them combine with both oil and water. Think about when you wash dishes that have grimy grease or heavy oil on them. Dishwasher soap (a surfactant) breaks down the grease or oil on the dishes into the water, so you can to clean the dish. Surfactants are used in many ways, including dishwashing detergents, shampoos and conditioners, paints, adhesives, ski and snowboard wax, fire extinguishers, sanitizers, medical cleansers, and hand soaps, among many other uses. Surfactants are also used in environmental engineering to clean up oil spills and pollution in water and soil. Engineers design different surfactants depending on what purpose they want the design to serve. As with all designs, they need to keep in mind design criteria and constraints.
To understand why surfactants such as soap are effective at killing bacteria, we need to know all the parts of a target cell. Let's build a bacteria cell on the chalk board together.
Bacteria are what kind of cells? Right, prokaryotes. Who can tell me a part of a prokaryotic cell? (Answer: The parts are cell wall, cell membrane, cytoplasm, nucleolis and ribosomes.) When we wash our hands, the soap attacks one part of the cell and kills it. Can anyone guess what part of the cell the soap destroys? Soap attacks the cell wall of a prokaryotic cell. So, when we wash our hands, the soap destroys the cell wall of the bacteria, spilling out all of the cell contents. The cell is no longer able to function properly — to eat, breathe and reproduce without a cell wall holding its parts together. Basically, the cell is inactivated. The reason why soap destroys the cell wall is because the cell wall is made of fat molecules (an organic compound), and soap (a surfactant) has the chemical properties that break down fats.
(Teacher demonstration using a bowl of water, pepper and liquid dish soap.) Here is a short demonstration to show how a surfactant changes the surface of water. This is a bowl of water (show water to students). Now I'm going to shake some pepper onto the surface of the water. See how it is evenly spread across the surface (walk around the students' desks showing the bowl of peppered water). Now watch as I release one drop of liquid soap on the top of the water. See how the pepper moved to the side. This is because the soap broke the surface tension of the water just like it breaks the cell wall when we wash our hands.
Today, we are going to work as engineers and test some surfactants by culturing bacteria that comes from our hands. Bacteria culturing is a technique that scientists and engineers use to study cells. The number of cell colonies on a culture plate tells us the effects that different surfactants have on the number of bacteria on our hands, and allows us to evaluate different soap designs. To culture bacteria cells, we must make sure that the culture media contains all of the food, vitamins and physical characteristics that the cells need to survive. For this experiment we are using a prepared nutrient-rich media, but some types of cells (especially those found in the natural environment) do not grow well on these types of media; they need a media that only has a small amount of nutrients. For today's activity, we will look at the effects of different surfactants on bacteria and develop a design for an improved surfactant to use in a variety of environmental situations.
Procedure
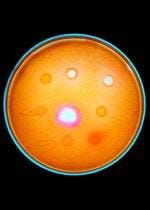
Background
It is important to keep the cultures sterile so that the bacteria that grow on the Petri dish only represent the bacteria from the students' hands, not those on the desks, for example. Discuss why it is important to have controls and the how the experimental process works. There are some assumptions that we make when using culturing as a technique, including: each colony represents one cell from the swab. and all of the cells on the swab were able to live off of the chosen media. The media for this experiment was chosen to grow bacteria, but there are many other types of media available for growing different types of microorganisms.
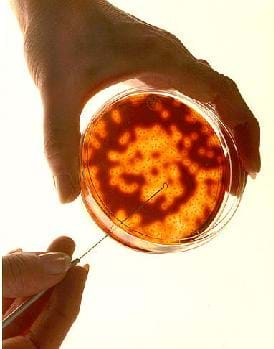
Before the Activity
- Gather materials and make copies of the Sudsy Cells Worksheet.
- If you are preparing the Petri dishes with media rather than buying them, do this a day in advance so the media has time to cool completely.
- Assign each student group one type of soap to use. Student groups should be counted off with the first five groups each using a different type of soap (bar soap vs. liquid hand soap, etc) and then assigning the next five groups the five different types of soaps, etc.
With the Students
- Go over the procedures for the activity with the class. Remind students about the basics of what a cell needs to survive and reproduce (food, room to grow, air to breathe). Talk about how the nutrient media meets the needs of the cells and that each cell should reproduce enough to form a visible colony by the end of the incubation period (2 days).
- Divide the class into groups of two students each. One is the washer, the other the recorder.
- Have students discuss a hypothesis and record it on their worksheet. The hypothesis should be an assertion of what the experiment will tell us about the specific surfactant (soap) and the number of cells on the hands washed with it. An example is: The hand washed with liquid soap will have half as much bacteria on it as the unwashed hand. Have each group tell the class their hypothesis.
- Have students answer the second pre-lab questions on their worksheets. Give the groups time to answer the question and then discuss the right answers as a class. Make sure that the students understand the idea of a control and how important it is for quality scientific experimentation, and a fair evaluation of designs. In this experiment, the unwashed hands are the controls. The effectiveness of the soaps will be determined by comparing the number of colonies grown on the unwashed hand (the control) to the number of colonies on the washed hands.
- Next, have groups gather the materials they need for the experiment. Each group needs two Petri dishes, two sterile swabs, and one type of surfactant (soap).
- Have groups decide who will wash his/her hands with the soap for the group. This student must swab her hand and wipe the swab on the first Petri dish before washing her hand with the assigned soap and swabbing a sample for the second Petri dish. Note: If students need to leave the classroom to wash their hands, make sure the other group member goes with him/her to open doors and make sure the first student does not touch anything on the way back.
- Put all the Petri dishes in a warm dark place to be incubated for 48 hours.
- When it is time to analyze the results of the experiment, have groups retrieve their Petri dishes and bring them back to their desks. The first thing the groups should do is draw on their log sheets what the Petri dishes look like.
- Have the second student (recorder) count the number of colonies on each Petri dish. Each colony represents a cell cultured from the student's hand. Students should fill in the appropriate spots in the table on the log sheet.
- Have students compare the number of colonies on the control (unwashed hand) and the number of colonies on an experimental dish. Have them write their group's answers on the board.
- Have students complete the chart on their worksheet.
- As a class, have students determine which soap design best decreases the amount of bacteria on dirty hands.
- Have the students discuss what the design criteria would be for the different soaps. For instance, the number of cells that must be removed as a percentage. Ask the students if the design criteria might change depending on the application (hospital soaps vs. soaps in your house).
- Have students think about designing a new surfactant for various applications. Remind students that this is similar to what engineers might do when designing a solution to an organic compound (like bacteria or oil) problem in people or the environment. Engineers study the ingredients in different surfactants (soaps) and design a surfactant product that would remove the offensive bacteria or organic compound. (Note: A next step in the design process [that they will not do in this activity] might be to test their new surfactant design on cultures of the organic compound for that situation.
Vocabulary/Definitions
cell colony: When bacteria reproduce they form colonies — groups of identical cells from the same parent cell.
culture media: Contains all of the nutrients and food necessary for bacteria to grow and reproduce.
inactivate: To lose disease causing capabilities, to render non-functional; that is, the cells are no longer dangerous and cannot make us sick.
soap: Soap is a chemical that breaks up cell membranes and also acts as a surfactant.
surfactant: Surfactants can attach to both polar and non-polar molecules making them good at dissolving non-polar molecules (such as oil), in polar liquids (such as water); soap is a type of surfactant.
Assessment
Pre-Activity Assessment
Prediction: Have students predict the outcome of the activity before the activity is performed.
- Which soap will destroy the most bacteria during this activity?
Practice Math Calculations: Have students answer the following word problems to prepare for the activity.
- Samantha, Bridget and Tony counted three cells on their plate after culturing a hand washed with bar soap, while Pete, Eric and Shelley counted five cells on their plate after culturing a hand washed with the same soap. What is the class average of the two trials? (Answer: Find the mean by adding the total cells counted for all trials [5+3] and then dividing by the number of trials [2]: [5 + 3] / 2 = 4.)
- The class average was 12 cell colonies on the Petri dish after culturing an unwashed hand and three cell colonies on the Petri dish after culturing a hand washed with anti-bacterial soap. What percentage of bacteria was removed by the antibacterial soap? (Answer: Find the difference between the control and the soap that is tested [12-3], and divide that by the number of colonies in the control [12] and then multiply by 100% to turn it into a percentage: [12-3] / 12 x 100% = 75%)
Activity Embedded Assessment
Worksheet: Have students complete the Sudsy Cells Worksheet, which asks them to draw pictures of the experiment as well as analyze the results using averages and percentages. Have them check with their groups to see if they got the right answers. Review their answers to gauge their mastery of the subject.
Post-Activity Assessment
Engineering Surfactant Impacts: Have students think about the impacts of engineering surfactants on individuals, society and the environment. Some surfactants are known to be toxic to the environment, other animals and humans. For example, industrial cleansers may remove harmful pollutants from entering the air stream but can be harmful to the environment if waste products are dumped into local waterways. Have students pick one of the following scenarios and discuss what impacts need to be considered in the design of a surfactant for that application.
- A large laundromat that cleans sheets and towels for a local hospital.
- An airport that removes airplane oil spills from the tarmac.
- A farm that removes weeds from their crop using an herbicide (surfactant).
- For recycling purposes, a factory that removes ink from old newspapers using a deinker (surfactant).
- A cleaning company that cleans the playground equipment at a school.
Troubleshooting Tips
If the cells are clumped together, the students could calculate the area covered by placing the Petri dishes over graph paper and the number of squares covered by bacteria can be used instead of counting individual colonies.
Also, if the colonies seem very small, they can be incubated longer than 48 hours or the students can use a magnifying glass to count them.
Activity Extensions
If microscopes are available, have students make slides with the cultured bacteria. See if different kinds of bacteria are present in the cultures and determine which soaps worked best with different bacteria. If possible, find out the types of bacteria by using an identification key. To extend the activity to oil spills, have students try out their soap design on some vegetable oil poured into a small bowl of water. Which soap design worked the best at breaking up the oil in the bowl?
Activity Scaling
- For upper grades, engage students in a more rigorous discussion of the factors affecting cell growth. Also, bring up the idea of antibiotic resistance and how microorganisms can adapt to our anti-bacterial products, rendering them ineffective.
- For lower grades, have students analyze results in a more qualitative way instead of calculating averages and percentages.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn about the basics of molecules and how they interact with each other. They learn about the idea of polar and non-polar molecules and how they act with other fluids and surfaces. Students acquire a conceptual understanding of surfactant molecules and how they work on a molecular level. ...

Student teams are challenged to evaluate the design of several liquid soaps to answer the question, “Which soap is the best?” Through two simple teacher class demonstrations and the activity investigation, students learn about surface tension and how it is measured, the properties of surfactants (so...

Students look at the components of cells and their functions. The lesson focuses on the difference between prokaryotic and eukaryotic cells.

In a very hands-on activity, students observe and feel the differences between two cleaning methods, with and without hand soap, using coffee grounds to represent "dirt."
References
Centers for Disease Control and Prevention, Emergency Preparedness and Response, "Plague," February 12, 2007, accessed September 29, 2008. <http://www.bt.cdc.gov/agent/plague/trainingmodule/images/Bio_Agents.jpg>
Pacific Northwest National Laboratory, Battelle for the U.S. Department of Energy, PictureThis, June 2008, accessed September 29, 2008. <http://picturethis.pnl.gov/im2/8208417-5cn0/8208417-5cn.jpg>
U.S. Department of the Interior, U.S. Geological Survey, Center for Coastal and Watershed Studies, Coral Microbial Ecology, "Microbial Wars: Mucus-Associated Bacteria Fend off Coral Pathogens," February 9, 2006, accessed September 29, 2008. < http://coastal.er.usgs.gov/coral-microbes/wars.html>
Copyright
© 2008 by Regents of the University of Colorado.Contributors
Kaelin Cawley; Malinda Schaefer Zarske; Janet YowellSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under a grant from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and National Science Foundation GK-12 grant no. 0338326. However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: March 8, 2021





User Comments & Tips