Quick Look
Grade Level: 8 (6-8)
Time Required: 45 minutes
Expendable Cost/Group: US $5.00
Group Size: 3
Activity Dependency: None
Subject Areas: Earth and Space
Summary
Students learn how to prevent exposure to the sun's ultraviolet rays. Students systematically test various sunscreens to determine the relationship between SPF (sun protection factor) value and sun exposure. At activity end, students are asked to consider how their investigation results could be used to help them design a new sunscreen.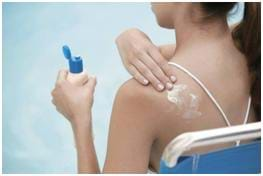
Engineering Connection
Engineers play a vital role in the design of sunscreen and other protective equipment that shield us from the harmful rays of the sun. Chemical engineers test and decide what chemicals best make up sunscreen products in order to achieve the desired ultraviolet (UV) protection. In addition, engineers contribute to the design of materials that are used for sunglasses and clothing that blocks the sun, as well as medical technologies used to treat skin cancer, including medicines and equipment.
Learning Objectives
After this activity, students should be able to:
- List and describe the types of UV radiation.
- Describe the importance of sunscreen and other protective equipment to block the sun's rays.
- Correlate sun exposure to SPF value of sunscreen.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
-
Conduct an investigation to produce data to serve as the basis for evidence that meet the goals of an investigation.
(Grades 6 - 8)
More Details
Do you agree with this alignment?
-
Construct and interpret graphical displays of data to identify linear and nonlinear relationships.
(Grades 6 - 8)
More Details
Do you agree with this alignment?
Common Core State Standards - Math
-
Reason abstractly and quantitatively.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Display numerical data in plots on a number line, including dot plots, histograms, and box plots.
(Grade
6)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Advances and innovations in medical technologies are used to improve healthcare.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
State Standards
Colorado - Math
-
Display numerical data in plots on a number line, including dot plots, histograms, and box plots.
(Grade
6)
More Details
Do you agree with this alignment?
-
Direct and indirect measurement can be used to describe and make comparisons.
(Grade
8)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- 1 square of UV photo-sensitive paper, such as Sun Print UV paper, approximately 4" x 6", which is a special paper that changes color in sunlight depending on how much UV light to which it is exposed; it is sold in 8" x 12" sheets, so you can cut them in half; available at https://www.amazon.com/SunPrint-W330-Paper-Kit/dp/B001KOGY3M
- 1 piece of glass or transparent plastic that is at least as big as the UV paper; overhead transparencies and plastic wrap work well
- 1 strip of opaque paper that is 1" x 6"; 4" x 6" index cards work well
- 1 office file folder, to keep the photographic paper in before exposing it to sunlight
- 2-3 cotton balls
- 1 small strip of index card to evenly spread the sunscreen; a straw works also
- pan of water, big enough to hold the square of photo-sensitive paper
Materials that may be shared with the entire class:
- 4 (or more) different SPF value sunscreens of a broad range; ideally, the SPF values increase in equal increments to facilitate a quantitative analysis at the end (for example: 2, 4, 6, 8 or 5, 10, 15, 20); lower SPF values for sunscreens and tanning oils work best
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/cub_sun_lesson01_activity1] to print or download.Pre-Req Knowledge
A basic understanding of creating bar graphs.
Introduction/Motivation
In the middle of a blizzard, most of us would not consider leaving the house without warm protective clothing. Why? Well, because we could get frostbite very quickly. When the weather is sunny, we still need to be careful to protect ourselves from harm. Sunlight exposure is important and necessary, because it helps your skin make vitamin D, but too much sun can be a bad thing! Raise your hand if you put sunscreen on before you head out into the sun.
What specifically does sunscreen guard against? It shields us from the sun's ultraviolet (UV) light rays. Much of the sun's UV light is absorbed or reflected by our atmosphere—more specifically, our ozone layer. The three three types of UV radiation are UVA, UVB and UVC. UVB and UVC are mostly absorbed by the ozone layer, although some still gets through. UVA is not absorbed at all. The UV radiation that penetrates the ozone layer can cause long-term problems such as skin cancer or premature aging. Of course it can also cause the short-term, very immediate problem of an uncomfortable burn.
Have any of you had a sunburn so badly that it caused your skin to peel or blister? Ouch, that hurts! Fortunately, we have ways to prevent too much sun exposure. First, we want to avoid things that increase our exposure. What are some factors that increase the likelihood that you will get burned? (Write student ideas on the board. Add any of the following that are not suggested: time of day, time of year, location compared to the Earth's equator, altitude, cloud cover). We can best protect ourselves by avoiding prolonged sun exposure, especially during peak hours (~noon to 3PM). Remember, even on cloudy or hazy days, your skin still can get burned.
If you must be in the sun, it is easy enough to cover up with clothing and hats. But if you are swimming, you can also apply sunscreen every few hours, as the product instructions suggest. Sunscreen acts in a similar fashion to the Earth's atmosphere; it helps block the UVB from reaching our skin. UVB is generally known to cause sun burns. What about UVA rays? Researchers do not yet know what causes skin cancer, but research points to UVA rays, which sunscreen generally does not block. What does SPF mean? (Answer: sun protection factor) The higher the SPF value, the more protection the sunscreen offers. But, that does not mean you should stay out in the sun longer or use less sunscreen!
Engineers play an important role in the design and production of sunscreen. Chemical engineers work together with researchers to determine the best compounds to use in the sunscreen to protect skin from too much sun exposure. For quality control, engineers test their products to determine how well they work. If the tests reveal that they do not work as well as expected, then engineers redesign them to improve them. We are going to see how well the SPF rating works in sunscreen. Will greater SPF values do a better job protecting the skin from harmful rays than lower ones? Let's test it out!
Procedure
Before the Activity
- In a dark room, prepare the materials by cutting the UV paper and the 1" strips down to size for each group. Place each paper in a file folder to keep the paper unexposed until the students are ready to collect their data.
- If possible, arrange to have the students complete this activity in the middle of the day, when the sun is at its highest point.
With the Students
- Have each group collect its materials (except the UV paper). Direct students to use a cotton ball to deposit a 1-2" diameter circle of each of the various types of sunscreen or tanning oils onto the plastic. Use the small piece of index card to spread the sunscreen into an even layer. You can fit 4-5 different sunscreens on the same piece of plastic. Refer to the diagram on the worksheet.
- Ask students to write down a hypothesis about which area (that is, which sunscreened spot) they expect will be the most protected.
- Move the entire class outside into a sunny area. Give each group a folder containing one piece of UV paper. Remind students to keep the UV paper in the folder until you tell them to take it out. Then they should carry their plastic piece on top of the file folder, holding it level as they walk.
- Explain this next step in advance, so that students can complete it correctly. It might be worthwhile to practice the steps with a blank sheet of paper, so that mistakes are minimized once the UV paper is exposed. Have students refer to the diagram on the worksheet.
- When ready, remove the UV paper and place the plastic piece directly on top of it. Place them in direct sunlight. The dark side of the paper should be the side that makes contact with the plastic sheet. The paper should change color or intensity as it is exposed to light. Expose the paper for 5 minutes.
- During these same 5 minutes, perform a control test. Take an opaque slip of paper that covers the top of the UV paper. Every minute, move the slip of paper about 1 inch to the right. This exposes the paper in a systematic way, giving students comparison sunscreen samples.
- Once the exposure is complete, move the project back inside. To stop further exposure of the UV paper, place it in a pan of water (so that it is completely submerged). The image may completely disappear and then reappear. Depending on the type of paper, the image will be completely inverted after soaking in the water. Leave the image in the water for 3 to 5 minutes. Remove the UV paper from the water and let it dry.
- Ask students to record observations as to the color differences between areas protected by the various lotions/oils. Then have them make a bar graph of the results. It may help to do make the first bar on the graph as a class. Have students look at their lowest SPF value. How does that exposed area compare to the control strip on top? Is it closest to the 1 min exposure, or somewhere else? Ask them to estimate about how long of an exposure to which it is equivalent. Once they have identified the equivalent exposure time, have them make a vertical bar on their graph to that point.
- Ask students to evaluate the quality of their data, as the last step on the worksheet.
Assessment
Pre-Activity Assessment
Prediction: Have students predict the outcome of the activity before the activity is performed. Ask them to make their hypotheses quantitative, for example: SPF 8 sunscreen with will have twice the amount of protection as SPF 4 sunscreen.
Activity Embedded Assessment
Worksheet: Have students record measurements and follow along with the activity on their worksheets. After students have finished their worksheets, have them compare answers with their peers.
Post-Activity Assessment
Prediction Analysis: Have students compare their initial predictions with their test results, as recorded on the worksheets.
Informal Discussion: Solicit, integrate and summarize student responses.
- Check for reliability of the experiment by comparing the results from each group. Ask students to explain why we would expect the results to be the same. (Answer: Research experiments should be repeatable, meaning that the result should be the same if the methods are identical.)
- Ask students to imagine that they are engineers designing a new sunscreen. Ask them to determine several ways that the results from their SPF experiment could be used in the design process.
Activity Extensions
- Have students design a new investigation related to sunscreen. For example, they could study the effect of sunscreen brand or expiration date. They would repeat the experiment above, but instead of varying the SPF value, they vary the brand or expiration date. After conducting the experiment, have students prepare a poster that compares the different brands. The poster could also include a comparison of their findings to background information that they have researched online.
- Complete a small research project (short paper or presentation) summarizing the experimental findings. Include information on skin cancer, how to prevent it, and the need to protect yourself while out in the sun.
Activity Scaling
For advanced students, have them try to develop a quantitative relationship between sunscreen SPF value and exposure time. Ask them to read an article about this relationship (http://www.fda.gov/Drugs/ResourcesForYou/Consumers/BuyingUsingMedicineSafely/UnderstandingOver-the-CounterMedicines/ucm239463.htm) and explain why exposure time is not directly related to SPF value.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

After seeing ultraviolet-sensitive beads change color and learning how they work, students learn about skin anatomy and the effects of ultraviolet radiation on human skin, pollution's damaging effect on the ozone layer that can lead to increases in skin cancer, the UV index, types of skin cancer, AB...

Students learn about the electromagnetic spectrum, ultraviolet radiation (including UVA, UVB and UVC rays), photon energy, the relationship between wave frequency and energy (c = λν), as well as about the Earth's ozone-layer protection and that nanoparticles are being used for medical applications

Students design beach clothing that is both fashionable and functional, meeting two key criteria: the clothing stays cool (considering factors such as fabric weight and thickness) and effectively shields against ultraviolet (UV) radiation. By the end of this activity, students gain a deeper understa...

Students explore the causes and effects of the Earth's ozone holes through discussion and an interactive simulation. In an associated literacy activity, students learn how to tell a story in order to make a complex topic (such as global warming or ozone holes) easier for a reader to grasp.
References
U.S. Environmental Protection Agency, SunWise Protection, "UV Radiation," January 3, 2008, accessed March 16, 2009. http://www.epa.gov/sunwise/uvradiation.html
Medical College of Wisconsin, HealthLink, "Sunscreen, Skin Cancer, and UVA," July 26, 2000, accessed March 16, 2009. http://healthlink.mcw.edu/article/964647970.html
U.S. Department of Veterans Affairs, VA Pacific Islands Health Care System, February 25, 2009, accessed March 17, 2009. http://www.va.gov/hawaii/images/clip_image002_0000.jpg
Copyright
© 2009 by Regents of the University of ColoradoContributors
Brian Kay; Karen King; Janet YowellSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
This activity was developed by the Integrated Teaching and Learning Program under National Science Foundation GK-12 grant no. 0338326. However, these contents do not necessarily represent the policies of the National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: October 23, 2025






User Comments & Tips