
Summary
Towards finding a solution to the unit's Grand Challenge Question about using nanoparticles to detect, treat and protect against skin cancer, students continue the research phase in order to answer the next research questions: What is the structure and function of skin? How does UV radiation affect chemical reactions in the skin? After seeing ultraviolet-sensitive beads change color and learning how they work, students learn about skin anatomy and the effects of ultraviolet radiation on human skin, pollution's damaging effect on the ozone layer that can lead to increases in skin cancer, the UV index, types of skin cancer, ABCDEs of mole and lesion evaluation, and the sun protection factor (SPF) rating system for sunscreens. This prepares students to conduct the associated activity, in which they design quality-control experiments to test SPF substances.Engineering Connection
Environmental and chemical engineers examine the chemical reactions that are involved when chlorofluorocarbons and other human-generated pollutants react to break down the ozone layer, permitting more UV radiation to penetrate through the Earth's atmosphere and putting people at higher risk of UV damage. Biomedical engineers understand that skin is the body's first defense against all outside intrusion as they research ways to protect against, detect and treat skin cancer.
Learning Objectives
After this lesson, students should be able to:
- Identify the main layers of human skin.
- Explain how UV radiation can cause skin cancer.
- Explain how biomedical engineers can begin to design solutions that prevent skin cancer if they know more about the causes.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-ESS3-1. Construct an explanation based on evidence for how the availability of natural resources, occurrence of natural hazards, and changes in climate have influenced human activity. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This lesson focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Construct an explanation based on valid and reliable evidence obtained from a variety of sources (including students' own investigations, models, theories, simulations, peer review) and the assumption that theories and laws that describe the natural world operate today as they did in the past and will continue to do so in the future. Alignment agreement: | Resource availability has guided the development of human society. Alignment agreement: Natural hazards and other geologic events have shaped the course of human history; [they] have significantly altered the sizes of human populations and have driven human migrations.Alignment agreement: | Empirical evidence is required to differentiate between cause and correlation and make claims about specific causes and effects. Alignment agreement: Modern civilization depends on major technological systems.Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-ETS1-1. Analyze a major global challenge to specify qualitative and quantitative criteria and constraints for solutions that account for societal needs and wants. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This lesson focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Analyze complex real-world problems by specifying criteria and constraints for successful solutions. Alignment agreement: | Criteria and constraints also include satisfying any requirements set by society, such as taking issues of risk mitigation into account, and they should be quantified to the extent possible and stated in such a way that one can tell if a given design meets them. Alignment agreement: Humanity faces major global challenges today, such as the need for supplies of clean water and food or for energy sources that minimize pollution, which can be addressed through engineering. These global challenges also may have manifestations in local communities.Alignment agreement: | New technologies can have deep impacts on society and the environment, including some that were not anticipated. Analysis of costs and benefits is a critical aspect of decisions about technology. Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-ETS1-2. Design a solution to a complex real-world problem by breaking it down into smaller, more manageable problems that can be solved through engineering. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This lesson focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Design a solution to a complex real-world problem, based on scientific knowledge, student-generated sources of evidence, prioritized criteria, and tradeoff considerations. Alignment agreement: | Criteria may need to be broken down into simpler ones that can be approached systematically, and decisions about the priority of certain criteria over others (trade-offs) may be needed. Alignment agreement: | |
International Technology and Engineering Educators Association - Technology
-
Medical technologies include prevention and rehabilitation, vaccines and pharmaceuticals, medical and surgical procedures, genetic engineering, and the systems within which health is protected and maintained.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Illustrate principles, elements, and factors of design.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
State Standards
Tennessee - Science
-
Recognize that science is a progressive endeavor that reevaluates and extends what is already accepted.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Explore the impact of technology on social, political, and economic systems.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Explain the relationship between the properties of a material and the use of the material in the application of a technology.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Describe the dynamic interplay among science, technology, and engineering within living, earth-space, and physical systems.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Identify structures of the integumentary, skeletal, and muscular systems and show the relationship between these structures and their functions.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Investigate physiological mechanisms that allow the integumentary, skeletal, and muscular systems to function.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Identify the structures of the skin and explain their role in protecting the body and maintaining homeostasis.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Explain the physiological processes involved in healing the skin and disorders of the skin.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
Worksheets and Attachments
Visit [www.teachengineering.org/lessons/view/van_nanoparticles_lesson02] to print or download.Introduction/Motivation
(For a class demo, have handy an ultraviolet-sensitive bead and access to a window with sunlight coming in. UV-detecting beads can be purchased from Flinn Scientific at https://www.flinnsci.com/ultraviolet-detecting-beads/fb1147/ and are used in the associated activity.)
In today's lesson, we will build on what we learned in the first lesson in this unit by taking a closer look at ultraviolet (UV) radiation. We will also learn how human-generated pollution can destroy the ozone layer and thus lead to greater UV intensity, and how overexposure to UV radiation can have detrimental effects to our overall health. Remember, environmental engineers study how people impact the environment and how the environment can affect us. So, studying how UV radiation can be intensified by pollution is a good example of an environmental engineering problem.
(Near a closed window, show students some UV-sensitive beads.) These are ultraviolet-detecting beads. Observe them in the sunlight coming through this window. (Let students watch. Expect the beads to begin changing color immediately when exposed to UV light.) Notice that this is happening through a closed window, through the glass. You can see how it is possible to get a sunburn even while driving in a car on a long trip.
UV-sensitive beads are typically constructed of two large planar conjugated molecules. Before exposure to UV light, these molecules are not bonded and appear colorless. Upon exposure to UV light, the two molecules bond to form a larger complex that absorbs a certain wavelength of light, resulting in a visible color. The longer the conjugated molecule, the longer the wavelength of light absorbed; thus, different colors of beads can be produced by using different molecules in the manufacture of the beads. When placed in the dark, the beads return to their colorless appearance, indicating that this is a reversible reaction in the absence of the UV light.
Chemical, biomedical and environmental engineering researchers are involved in addressing the problems that can result from too much UV radiation on human skin by studying the breakdown of the Earth's protective ozone layer and working to develop detection procedures and skin cancer treatments and cures. Understanding how these beads work as indicators can help engineers design better detection systems to help prevent skin cancer caused by UV radiation.
Lesson Background and Concepts for Teachers
Legacy Cycle Information for this Lesson
(Generate Ideas) Ask students the following research questions:
- What is the structure and function of skin?
- How does UV radiation affect chemical reactions in the skin?
In class, have students write journal entries to answer the following question: What are your initial ideas about how to answer these two questions?
(Multiple Perspectives) As a class, have students share ideas from their journals. Record their ideas on the classroom board.
(Research and Revise) Show students an ultraviolet-sensitive bead so they see that UV radiation, although unseen, has the capability to cause chemical reactions. Present to students the Introduction/Motivation information to explain how UV beads work. Then deliver the background lecture information on skin anatomy and the effect of ultraviolet radiation on it, pollution such as chlorfluorocarbons and its effect on the ozone layer, the UV index, types of skin cancer, ABCDEs of mole and lesion evaluation, and the sun protection factor (SPF) rating system for sunscreens.
(Test Your Mettle) Conduct the How Effective Is Your Sunscreen? associated activity.
(Multiple Perspectives extension) Conclude the lesson by showing the Using Lasers to Detect Skin Cancer video clip (2:13 minutes) on laser skin treatment with Dr. Anita Jansen, professor of biomedical engineering at Vanderbilt University.
Lecture Information
UVA and UVB radiation is radiant energy of wavelength 280 nm – 400 nm. Ultraviolet radiation has the capability to cause certain chemical reactions including several within human skin. Human skin is a key component in the body's integumentary system, which is the organ system that protects the body from external damage, such as water loss or abrasion. Skin consists of three main layers: epidermis, dermis and hypodermis.
- The epidermis, composed of the outermost layers of skin, consists of four layers in most areas of the body and five layers on areas of the body that have thick skin, such as the soles of the feet. These layers, from outermost to innermost, are the stratum corneum, stratum lucidum (only present in thicker skin), stratum granulosum, stratum spinosum and stratum basale.
- The dermis, located beneath the epidermis, consists of two layers: superficial papillary layer and deeper reticular layer. The dermis is strong and elastic in part due to the collagen and elastic fibers dispersed throughout. The breaking down of these collagen fibers after overexposure to UV light can lead to the loss of elasticity of the skin and premature aging.
- The hypodermis or subcutis, which lies below the dermis, is essentially adipose (fat) tissue.
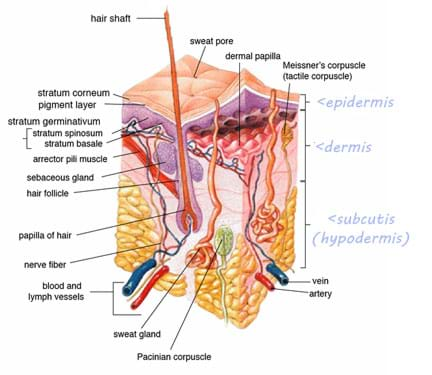
A look inside human skin.
The epidermis only has one active dividing cell layer, the stratum basale. The stratum basale is supplied with nutrients through diffusion from the vascularized dermis. The epidermis gets its tough protective characteristics from a type of cell called a keratinocyte. Keratinocytes die and change shape as they are pushed from the stratum basale to the stratum corneum, where they eventually slough or rub off. Melanocytes are also contained in the stratum basale. These cells produce melanin, which is taken in by the keratinocytes in order to protect the nucleus from UV damage. Some amount of UV damage can be repaired naturally by the body's enzyme system, and UV exposure is essential for the production of vitamin D. The molecule 7-dehydrocholesterol produces vitamin D3 when skin is exposed to ultraviolet light in an environment in which the UV index is 3 or greater. Vitamin D3 is a precursor to active vitamin D, which is essential in calcium metabolism.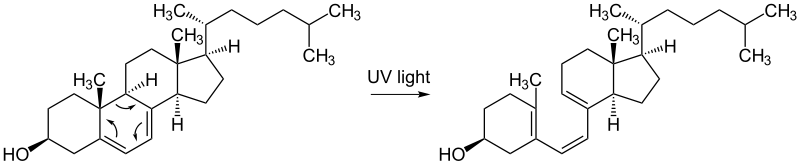
Ultraviolet light is also essential in producing the ozone layer that is our environment's natural way of shielding us from UV light.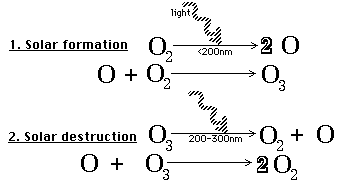
The formation and destruction of ozone, when held in balance, helps to create the ozone layer, which serves as our skin's first line of defense from ultraviolet radiation exposure from the sun. In certain areas of the world, the ozone layer has been depleted and an increase in the incidence of human skin cancer in those areas has been observed.
The human-generated pollution includes substances such as chlorfluorocarbons. As the following series of reactions show, chlorofluorcarbons react with ultraviolet light to produce Cl (chlorine free radical), which in turn reacts to break down O3 (ozone). To contribute to the problem, ClO (chlorine monoxide free radical), which is a product of the Cl and O3 reaction, reacts with O (oxygen), thereby removing an oxygen that could have been used in the further production of ozone.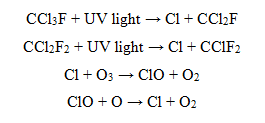
Over the country of Australia, ozone depletion due to pollution has caused dramatic health effects; statistically, one of every three people is expected to get skin cancer. The Australian government is implementing a plan to restore the ozone layer and educate its citizens on the danger of overexposure to ultraviolet radiation. In Australia and many other places in the world, UV index ratings are posted to inform people about the local UV intensity for particular months, days and sometimes certain times of the day. The typical global UV index scale is a color-coded number range from 1 to 11+ to indicate the human risk of exposure, such as this UV index scale used in the U.S.: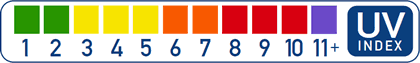
Explanation of the UV index scale:
- 2 or lower means low danger from the sun's UV rays for the average person.
- 3–5 means moderate risk of harm from unprotected sun exposure.
- 6–7 means high risk of harm from unprotected sun exposure. Apply a sunscreen with a SPF of at least 15. Wear a wide-brim hat and sunglasses to protect the eyes.
- 8–10 means very high risk of harm from unprotected sun exposure. Minimize sun exposure during midday hours, from 10 AM to 4 PM. Protect skin by liberally applying a sunscreen with an SPF of at least 15. Wear protective clothing and sunglasses to protect the eyes.
- 11 or higher means extreme risk of harm from unprotected sun exposure. Try to avoid sun exposure during midday hours, from 10 AM to 4 PM. Apply sunscreen with an SPF of at least 15 liberally every two hours.
UV radiation rays, which are more energetic than visible light, have the potential to damage skin cell DNA and lead to skin cancer. UVA has the ability to penetrate through the epidermis, dermis and enter the hypodermis. UVB penetrates completely through the epidermis and slightly into the dermis. UV exposure may damage skin cells, leading to DNA defects in which adjacent thymine bases become covalently linked, creating thymine dimers. Thymine dimers have the potential to cause buckling in the strand and create misreading during DNA replication.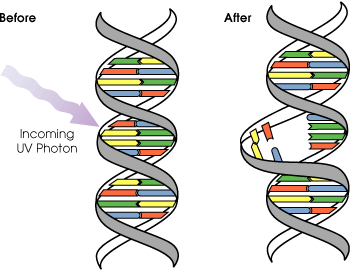
On a small scale, the body has its own system to correct damaged DNA through a process called nucleotide excision repair. In this type of repair, the damaged portion of DNA is cut out of the strand by an enzyme called a nuclease. Then, the space left behind is filled with the correct bases to fit the complimentary DNA strand. DNA polymerase and DNA ligase are two of the enzymes involved in filling the strand. However, with enough overexposure from the sun or artificial UV sources, such as tanning beds, the damage may be too great for repair, so skin cancer results.
The three main types of skin cancer:
- Basal cell carcinoma is the least fatal of the three, with 99% of people surviving if the cancer is treated. In basal cell carcinoma, shiny lesions appear, typically on the face. In this type of cancer, stratum basale cells lose the ability to form keratin and the boundary between the epidermis and dermis is not observed. This occurs slowly, which is why treatment is typically successful.
- Squamous cell carcinoma originates from the cells of the stratum spinosum. This type of skin cancer grows more rapidly than the basal cell carcinoma, but has a fairly good cure rate if removed and treated early with radiation. The lesions appear scaly and usually produce an ulcer.
- Malignant melanoma is skin cancer affecting melanocytes. This type grows rapidly and metastasizes quickly. The survival rate is ~50%. Usually, this type spreads as a black or brown lesion with irregular borders.
To help with early recognition of skin cancer, the medical community suggests looking for these warning signs in any moles and lesions, which they call the ABCDEs of melanoma:
A. Asymmetry: If the two sides of the lesion do not match.
B. Border irregularity: If the borders of the lesion have indentations.
C. Color: If the lesion contains different colors or if it is extremely dark.
D. Diameter: If the lesion is larger in diameter than 6 millimeters.
E. Evolving: If any changes are seen in size, shape, color, elevation or other traits.
Sunscreen is designed to protect skin from overexposure from ultraviolet light. Sunscreen products use a sun protection factor (SPF) rating system that is based on multiples of the number of minutes the average person could be exposed to the sun without burning. If the average time is 15 minutes, then use of an SPF 15 sunscreen theoretically enables the person to be in the sun for 225 minutes without burning. Some sunscreens protect against both UVA and UVB, so it is important to check before purchasing them. Some products—sunscreens—contain molecules that filter UV light, while other products—sunblocks—contain chemicals designed to block UV light all together. The next lesson, Nanotechnology and Cancer Treatment (the third lesson in this unit), describes ways nanotechnology is being used for the detection and treatment of cancer.
Associated Activities
- How Effective Is Your Sunscreen? - Student groups design their own quality-control experiments to test the effectiveness of current UV safety products such as sunscreens, sunblock and sunglasses. They are provided with a number of materials and tools that can be used, including UV-detecting beads, and must select one variable to test. They collect data, graph results, write lab reports and share their findings with the class.
Vocabulary/Definitions
ABCDEs of melanoma: Medical guidelines to screen moles and lesions for potential skin cancer.
collagen: The fibrous constituent of connective tissue within the dermis of the skin; provides skin with elasticity.
dermis: The inner layer of skin, also known as the hide. Consist of a reticular and papillary layer.
enzyme: A substance that speeds up the rate of or facilitates a chemical reaction.
epidermis: The most superficial layer of the skin.
hypodermis: The deepest layer of skin composed of adipose (fat) tissue.
keratinocyte: A skin cell that produce keratin.
laser: Acronym for "light amplification through stimulated emission of radiation."
melanocyte: A skin cell that produces melanin.
skin: The body's outer covering that serves to maintain borders and first line of defense for human immunity.
SPF: Acronym for sun protection factor, a standardized rating for sunscreen effectiveness in filtering or blocking ultraviolet light.
Assessment
Pre-Lesson Assessment
Research Questions: As part of the Generate Ideas phase, pose the following questions to students:
- What is the structure and function of skin?
- How does UV radiation affect chemical reactions in the skin?
Then have students individually write journal entries to answer the question:
- What are your initial ideas about how to answer these two questions?
To fulfill the Multiple Perspectives phase, have students share their ideas from their journals as a class, writing them on the classroom board.
Lesson Summary Assessment
Review and Revise: Have students re-read their initial journal entries from the first lesson in this unit, Electromagnetic Radiation, and correct any misconceptions. Have them share with the class the corrections they made.
Additional Multimedia Support
Find out the daily UV index for your U.S. zip code using the EPA's SunWise utility at http://www2.epa.gov/sunwise/uv-index#lookup.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn how to prevent exposure to the sun's ultraviolet rays. Students systematically test various sunscreens to determine the relationship between SPF (sun protection factor) value and sun exposure.

This unit on nanoparticles engages students with a hypothetical Grand Challenge Question that asks about the skin cancer risk for someone living in Australia, given the local UV index and the condition of the region's ozone layer. Through three lessons, students learn about the science of electromag...

Students learn about the electromagnetic spectrum, ultraviolet radiation (including UVA, UVB and UVC rays), photon energy, the relationship between wave frequency and energy (c = λν), as well as about the Earth's ozone-layer protection and that nanoparticles are being used for medical applications

Students explore the causes and effects of the Earth's ozone holes through discussion and an interactive simulation. In an associated literacy activity, students learn how to tell a story in order to make a complex topic (such as global warming or ozone holes) easier for a reader to grasp.
References
Marieb, Elaine N. Essentials of Human Anatomy and Physiology. Seventh edition. San Francisco, CA: Benjamin Cummings, 2003.
Copyright
© 2013 by Regents of the University of Colorado; original © 2010 Vanderbilt UniversityContributors
Michelle Bell, Amber SpolarichSupporting Program
VU Bioengineering RET Program, School of Engineering, Vanderbilt UniversityAcknowledgements
The contents of this digital library curriculum were developed under National Science Foundation RET grant nos. 0338092 and 0742871. However, these contents do not necessarily represent the policies of the NSF, and you should not assume endorsement by the federal government.
Last modified: July 17, 2018







User Comments & Tips