
Summary
Students take on the role of engineers designing a “Mars Thermos,” similar to how NASA engineers create insulation systems to protect spacecraft components and scientific samples from extreme temperature changes on Mars. Student teams use the engineering design process and everyday materials to design and build an insulator that keeps a small amount of water from changing more than 5°F over 10 minutes. After conducting a control experiment using uninsulated cups, students investigate the insulating properties of materials to inform their designs. Teams measure temperature at regular intervals, graph and analyze their results, and compare the performance of their designs to the control. Through this process, students explore heat transfer and basic thermodynamics while building scientific inquiry, data collection, and mathematical analysis skills.Engineering Connection
Thermal engineers design and analyze systems that control heat flow to keep equipment, instruments, and astronauts within safe operating temperatures. At NASA, they play a critical role in designing spacecraft, rovers, landers, and habitats for extreme environments like Mars, where temperatures can swing dramatically between day and night. Thermal engineers develop insulation systems, thermal coatings, heaters, radiators, and protective layers that regulate temperature, prevent overheating or freezing, and ensure reliable performance of sensitive electronics and scientific instruments. By applying principles of heat transfer and thermodynamics, thermal engineers help ensure mission success, protect valuable equipment, and enable long-term exploration of Mars and other worlds.
Learning Objectives
After this activity, students should be able to:
- Explain how heat transfer (conduction, convection, and radiation) and thermal equilibrium affect temperature changes in materials and systems.
- Investigate and compare the insulating properties of different materials through controlled experimentation.
- Collect, record, graph, and analyze temperature data to evaluate the effectiveness of design solutions.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-1. Define the criteria and constraints of a design problem with sufficient precision to ensure a successful solution, taking into account relevant scientific principles and potential impacts on people and the natural environment that may limit possible solutions. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Define a design problem that can be solved through the development of an object, tool, process or system and includes multiple criteria and constraints, including scientific knowledge that may limit possible solutions. Alignment agreement: | The more precisely a design task's criteria and constraints can be defined, the more likely it is that the designed solution will be successful. Specification of constraints includes consideration of scientific principles and other relevant knowledge that is likely to limit possible solutions. Alignment agreement: | All human activity draws on natural resources and has both short and long-term consequences, positive as well as negative, for the health of people and the natural environment. Alignment agreement: The uses of technologies and any limitations on their use are driven by individual or societal needs, desires, and values; by the findings of scientific research; and by differences in such factors as climate, natural resources, and economic conditions.Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-2. Evaluate competing design solutions using a systematic process to determine how well they meet the criteria and constraints of the problem. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Evaluate competing design solutions based on jointly developed and agreed-upon design criteria. Alignment agreement: | There are systematic processes for evaluating solutions with respect to how well they meet the criteria and constraints of a problem. Alignment agreement: | |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-3. Analyze data from tests to determine similarities and differences among several design solutions to identify the best characteristics of each that can be combined into a new solution to better meet the criteria for success. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Analyze and interpret data to determine similarities and differences in findings. Alignment agreement: | There are systematic processes for evaluating solutions with respect to how well they meet the criteria and constraints of a problem. Alignment agreement: Sometimes parts of different solutions can be combined to create a solution that is better than any of its predecessors.Alignment agreement: Although one design may not perform the best across all tests, identifying the characteristics of the design that performed the best in each test can provide useful information for the redesign process—that is, some of the characteristics may be incorporated into the new design.Alignment agreement: | |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-PS3-3. Apply scientific principles to design, construct, and test a device that either minimizes or maximizes thermal energy transfer. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Apply scientific ideas or principles to design, construct, and test a design of an object, tool, process or system. Alignment agreement: | Temperature is a measure of the average kinetic energy of particles of matter. The relationship between the temperature and the total energy of a system depends on the types, states, and amounts of matter present. Alignment agreement: Energy is spontaneously transferred out of hotter regions or objects and into colder ones.Alignment agreement: The more precisely a design task's criteria and constraints can be defined, the more likely it is that the designed solution will be successful. Specification of constraints includes consideration of scientific principles and other relevant knowledge that is likely to limit possible solutions.Alignment agreement: A solution needs to be tested, and then modified on the basis of the test results in order to improve it. There are systematic processes for evaluating solutions with respect to how well they meet criteria and constraints of a problem.Alignment agreement: | The transfer of energy can be tracked as energy flows through a designed or natural system. Alignment agreement: |
Common Core State Standards - Math
-
Display numerical data in plots on a number line, including dot plots, histograms, and box plots.
(Grade
6)
More Details
Do you agree with this alignment?
-
Construct and interpret scatter plots for bivariate measurement data to investigate patterns of association between two quantities. Describe patterns such as clustering, outliers, positive or negative association, linear association, and nonlinear association.
(Grade
8)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Apply the technology and engineering design process.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Assess design quality based upon established principles and elements of design.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Explain how knowledge gained from other content areas affects the development of technological products and systems.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- 2 thermometers (inexpensive digital kitchen [meat] thermometers may be purchased online- read reviews for accuracy, durability, and waterproof capabilities- or any classroom-safe thermometers may be used.
- 1 stopwatch
- 1 graduated cylinder
- 2 plastic or hot drink cups (small cappuccino cups or paper cups are about the right size. If there's more than 100 ml of water, the temperature will not change as quickly.)
- writing utensils (1 per student)
- Mars Thermos Worksheet (1 per student)
For the entire class to share:
- various insulating materials (bubble wrap, paper, cloth, sand, water, foil, Styrofoam, etc.)
- tape (masking, packing, or duct)
- paper towels (for cleaning up spills)
- pitcher (for ice water)
- ice
- water
- insulated commercial thermos or carafe (for warm water)
- electric tea kettle or hot plate and pot (for heating water)
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/jpl-3053-mars-thermos-design-heat-transfer] to print or download.Pre-Req Knowledge
Students should have familiarity with:
- Temperature and thermometers, understanding what temperature measures and how to safely and accurately read a thermometer.
- Recording measurements in tables or charts.
- Creating and interpreting basic line graphs.
- Adding, subtracting, and calculating temperature change.
Introduction/Motivation
Today, we are going to do an exciting hands-on activity where you become engineers designing a solution for Mars!
On Mars, temperatures can swing dramatically—sometimes extremely cold at night and warmer during the day. NASA engineers face a big challenge: They must protect important instruments, electronics, and even samples from these extreme temperature changes. If things get too hot or too cold, they could stop working or get damaged. That is where thermal engineers come in. Thermal engineers design insulation and temperature-control systems to keep things safe, so missions to Mars can succeed.
In our activity, you will work in teams to design and build your very own “Mars Thermos” using everyday materials. You will conduct an experiment to see how heat moves between objects and how materials can slow that movement. Your goal is to keep a small amount of water from changing temperature by more than 5 degrees over 10 minutes. First, you will do a test with an uninsulated cup as a control, and then you’ll investigate different materials to see which ones make the best insulation. You’ll collect temperature data, graph your results, and see which designs work best.

This activity will let you practice important skills that engineers use every day: designing solutions, testing ideas, analyzing data, and improving your designs. You’ll also explore science concepts such as heat transfer, thermal equilibrium, and thermodynamics, while using math to record and analyze your results.
Remember, safety is important: Hot water will not be hotter than 120 degrees Fahrenheit, and ice water will be handled carefully. Work with your team, share ideas, and have fun testing and improving your Mars Thermos.
By the end of this activity, you’ll see how engineers think, experiment, and solve problems—just like NASA engineers!
Procedure
Background (Teacher's Guide)
Thermal Concepts
Heat transfer is the movement of thermal energy from a warmer object to a cooler one, and it occurs in three main ways: conduction, which happens through direct contact between materials; convection, which occurs when heat moves through fluids like air or water; and radiation, the transfer of energy through electromagnetic waves, such as sunlight. Thermodynamics is the study of energy and how it moves and changes form, including the rules that govern heat flow. One important concept is thermal equilibrium, which occurs when two objects of different temperatures come into contact and heat flows from the warmer object to the cooler one until their temperatures are equal. In this activity, students observe these principles in action as heat moves into or out of water and explore how insulation slows that process, helping to maintain the water’s temperature over time.
Thermal and Aerospace Engineering
This activity connects directly to real-world engineering, particularly thermal and aerospace engineering. Engineers at NASA designing spacecraft, rovers, and landers for Mars must protect sensitive instruments and collected samples from extreme temperature swings, which can damage electronics or compromise experiments. To solve this, they create insulation systems, thermal coatings, and temperature-control components that keep equipment within safe operating ranges. In this activity, students simulate this process by acting as engineers: They test different materials, build prototypes, collect temperature data, and iteratively improve their designs. Understanding this connection allows you to present the activity as a meaningful, hands-on example of how engineers solve real-world problems in extreme environments.

Before the Activity (Preparation)
- Make copies of the Mars Thermos Worksheet (1 per student)
- Gather materials
- Collect various insulating materials (bubble wrap, paper, cloth, sand, water, foil, Styrofoam, etc.)
- Heat water to no warmer than 120°F and place it in a well-insulated commercial thermos or carafe.
- Fill the pitcher with ice and pour water over the ice to fill the pitcher. Mix and allow the water to become ice cold.
- Have paper towels readily accessible to all teams in case of spills.
- Optional: To expedite the experiment, set up one control (un-insulated) set of cups for which you (or a designated student) will record temperatures.
During the Activity
- Introduce the activity by explaining the objective: Teams must keep water from changing temperature more than 5°F in 10 minutes.

- Quick concept check: Ask students to answer the following questions individually or in pairs:
- What is the difference between heat and temperature? (Potential answers: Heat is the transfer of energy from a warmer object to a cooler object; temperature is a measure of how hot or cold an object is. Heat is energy in motion, while temperature tells us how much energy the particles in an object have.)
- Name two ways heat can move from one object to another. (Potential answers: Conduction is when heat moves through direct contact between materials (e.g., a metal spoon in hot water); convection is when heat moves through fluids like liquids or gases as warmer particles rise and cooler particles sink (e.g., warm water circulating in a cup); radiation is when heat moves as electromagnetic waves, without needing direct contact (e.g., sunlight warming your skin).)
- What do you think will happen to hot water if it is left in an uninsulated cup at room temperature? Why? (Potential answers: The hot water will cool down until it reaches the same temperature as the room (thermal equilibrium). This happens because heat flows from the warmer water to the cooler air around it.)
- What materials do you think could keep water hot or cold longer? Why? (Potential answers: Materials that slow heat transfer work best, such as foam, plastic, bubble wrap, or cloth. These materials trap air, resist conduction, and act as insulators, keeping hot water hot and cold water cold for a longer time.)
- As a class, discuss the definitions and real-world applications of heat, temperature, thermal equilibrium, and thermal energy transfer:
- Heat
- Definition: The transfer of thermal energy from a warmer object to a cooler one.
- Real-world application: Engineers design insulation in spacecrafts, buildings, and thermoses to control heat flow and keep objects or environments at safe temperatures.
- Temperature
- Definition: A measure of how hot or cold an object is; it reflects the average energy of particles in a substance.
- Real-world application: Thermometers measure temperature to monitor weather, cooking, body health, and scientific experiments. NASA engineers monitor spacecraft temperatures to ensure instruments operate safely.
- Equilibrium
- Definition: The state in which two objects in contact reach the same temperature and heat no longer flows between them.
- Real-world application: Hot coffee cools to room temperature over time because it eventually reaches thermal equilibrium with the surrounding air. Engineers use thermal equilibrium calculations to design temperature-controlled systems for electronics or spacecraft.
- Thermal Energy Transfer
- Definition: The movement of energy from one object or system to another due to a temperature difference.
- Real-world application: Thermal energy transfer explains why ice melts in warm water or why buildings use insulation. NASA engineers design spacecraft to manage thermal energy transfer to protect instruments from Mars’ extreme temperatures.
- Distribute one Mars Thermos Worksheet to each student.
- State the activity challenge: Today you will conduct an experiment to demonstrate the movement of atoms and molecules through temperature measurements and engineer a design that can slow down that movement.
- Give students 5 minutes to write definitions of the above terms in their own words on their copies of the Mars Thermos Worksheet.
- Ask students to thoroughly read through the procedure on their Mars Thermos Worksheet (also listed below.)
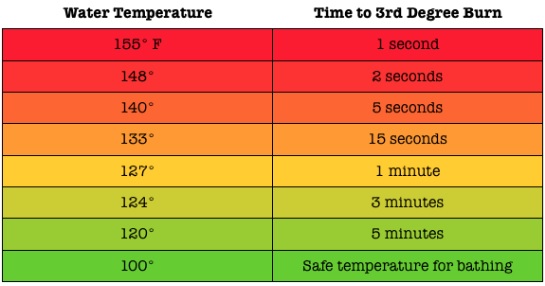
- Emphasize hot water safety protocols. (Reference Image 6 for the Time/Temperature Relationship in Scalds.)
- Before beginning, ask students to predict how the temperature will change in each cup of water (cold and hot) and then, using the vocabulary defined previously, write a hypothesis about what they think will happen in the experiment.
- Optional: Instruct students on the use of thermometers and have them announce, compare, and record the temperature of the room in degrees Fahrenheit.
- Ask each group to decide how much water they should use in each cup. Ask them to write their justification for their chosen amount. Students should use their prior knowledge and thought processes to consider that a greater amount of water will change temperature more slowly than a smaller amount of water.
- Ask each group to decide how often they will record the temperature for each cup of water. More frequent measurements will yield more accurate graphs but may not be feasible. A reasonable compromise should be reached that will allow an accurate graph without undue burden of manual data collection.
- Have student teams use graduated cylinders to obtain ice water (with no ice) and hot water (no warmer than 120°F) samples, place the samples in their control cups, and perform the control experiment.
- Have students investigate the insulative properties of the materials available and then design and build a Mars Thermos for the cold and hot cup. Remind them to make sure they are able to easily obtain temperature measurements.
- Have teams perform the experiment with their Mars Thermos designs, then graph their data.
- Circulate the classroom during the activity asking questions about students’ designs.
- Optional: Have teams design a second set of Mars Thermoses, improving upon their original design, and then redo the experiment.
- Have teams share their data with the class. Note: A successful Mars Thermos prevents water from changing temperature more than 5°F in 10 minutes. An accurate graph and explanation for the design should accompany each Mars Thermos. (See the Engineering Challenge Rubric.)
- Wrap up the activity with the following questions:
- How did the temperature of the hot water change? Cold water?
- How do your experiment’s data compare to the control experiment?
- Do you think the temperatures in the cup will reach equilibrium with the air in the room? If so, predict how long this would take.
Vocabulary/Definitions
conduction: Heat transfer that occurs through direct contact between materials.
control (experiment): A standard setup used for comparison that does not include the experimental variable.
convection: Heat transfer that occurs through fluids (liquids or gases) as warmer particles rise and cooler particles sink.
heat: The transfer of thermal energy from a warmer object to a cooler one.
insulation: A material or system that slows down the transfer of heat.
radiation: Heat transfer through electromagnetic waves without direct contact (e.g., sunlight).
temperature: A measure of how hot or cold an object is; reflects the average energy of its particles.
thermal energy transfer: The movement of energy from one object or system to another due to a temperature difference.
thermal equilibrium: The state in which two objects in contact reach the same temperature and heat no longer flows between them.
Assessment
Pre-Activity Assessment
Quick Concept Check: Students are asked a few short questions individually or in pairs before the activity starts. Student answers show you their baseline understanding of heat transfer, insulation, and everyday materials.
- What is the difference between heat and temperature? (Potential answers: Heat is the transfer of energy from a warmer object to a cooler object; temperature is a measure of how hot or cold an object is. Heat is energy in motion, while temperature tells us how much energy the particles in an object have.)
- Name two ways heat can move from one object to another. (Potential answers: Conduction is when heat moves through direct contact between materials (e.g., a metal spoon in hot water); convection is when heat moves through fluids like liquids or gases as warmer particles rise and cooler particles sink (e.g., warm water circulating in a cup); radiation is when heat moves as electromagnetic waves, without needing direct contact (e.g., sunlight warming your skin).)
- What do you think will happen to hot water if it is left in an uninsulated cup at room temperature? Why? (Potential answers: The hot water will cool down until it reaches the same temperature as the room (thermal equilibrium). This happens because heat flows from the warmer water to the cooler air around it.)
- What materials do you think could keep water hot or cold longer? Why? (Potential answers: Materials that slow heat transfer work best, such as foam, plastic, bubble wrap, or cloth. These materials trap air, resist conduction, and act as insulators, keeping hot water hot and cold water cold for a longer time.)
Activity Embedded (Formative) Assessment
Activity Observations: The formative assessment for this activity is ongoing and occurs as you observe and question students while they predict, design, test, collect data, graph results, and reflect on their Mars Thermos performance. Check for understanding of heat transfer concepts, proper use of scientific reasoning, accurate data collection, and the ability to draw evidence-based conclusions.
Data Analysis and Graphing: Students graph their temperature data and compare it to the control, demonstrating their ability to organize and visualize results. Assess the accuracy of their data collection and their ability to identify trends and draw evidence-based conclusions.
Post-Activity (Summative) Assessment
Class Discussion and Reflection: Each team shares their results and discusses how their designs performed, explaining their reasoning and observations. Listen for correct use of vocabulary and concepts and ask reflective questions, such as why one design worked better or how changes in insulation or water volume could affect the outcome.
Wrap Up Discussion Questions: Wrap up the activity with the following questions:
- How did the temperature of the hot water change? Cold water?
- How do your experiment’s data compare to the control experiment?
- Do you think the temperatures in the cup will reach equilibrium with the air in the room? If so, predict how long this would take.
Safety Issues
- Water should NEVER be more than 120°F for students. See scald table below.
- Scalds can cause serious injury. If in doubt about your students' ability to follow proper safety precautions, perform the cold-water portion of the experiment only.
- Use an electric teapot to heat water safely away from students and mix with cooler water in a commercial insulated thermos to the appropriate temperature (120°F) and then pour for students. Note: BE CAUTIOUS of using a microwave oven to heat the water—scalds can happen very easily by way of super-heating.
Troubleshooting Tips
- This experiment works best when the temperature in your classroom is moderate (i.e., not on a cold winter day when the heater is broken).
- Use ice to pre-cool the water only. DO NOT transfer any ice to student cups.
- If there's more than 100 ml of hot or cold water, the temperature will not change as quickly.
- Instruct students to start their measurements immediately upon receiving their water samples.
Activity Extensions
- Have students vary the amount of water used in the two designs to determine the impact the amount of water has on the experiment.
- Have students answer the following questions and support their reasoning with facts:
- Would the water change temperature more slowly or more rapidly on Mars?
- Does it matter whether the application of keeping my soup warm until lunch is as effective as keeping my body at roughly “body temperature” when on Mars?
- In what situations on Earth are insulation and limiting thermal transfer important?
Activity Scaling
For lower grades:
- Use fewer materials or just one cup per team.
- Reduce data collection frequency (e.g., only record start, middle, and end temperatures).
- Skip redesign/iteration to shorten the activity.
- Focus on one temperature condition (hot or cold) instead of both.
- Use pre-filled water cups rather than having students measure, to save time.
For upper grades or advanced students:
- Add multiple variables to test, such as water volume, cup material, or insulation layers.
- Require students to graph and analyze data in more detail, including slope of temperature change or rate of heat transfer.
- Include a second round of redesign with constraints such as limited materials or weight limits.
- Introduce thermodynamics calculations, such as estimating heat lost or gained using specific heat equations.
- Have students compare their designs to real NASA thermal protection systems and discuss engineering tradeoffs.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

In this lesson and its associated activity, students learn about aerogel, the silicon-based solid with a sponge-like structure. Students also learn about density and how aerogel is 99.8% air by volume, making it the lightest solid known to humans!

Students learn about the definition of heat as a form of energy and how it exists in everyday life. They learn about the three types of heat transfer—conduction, convection and radiation—as well as the connection between heat and insulation.

Students are introduced to various types of energy with a focus on thermal energy and types of heat transfer as they are challenged to design a better travel thermos that is cost efficient, aesthetically pleasing and meets the design objective of keeping liquids hot.
References
JPL Activity: https://www.jpl.nasa.gov/edu/resources/lesson-plan/mars-thermos
Other Related Information
Modified from https://www.jpl.nasa.gov/edu/resources/lesson-plan/mars-thermos
Copyright
© 2026 by Regents of the University of Colorado; original © 2025 Jet Propulsion LaboratorySupporting Program
NASA Jet Propulsion Laboratory (JPL)Last modified: April 28, 2026







User Comments & Tips