Quick Look
Grade Level: 9 (9-12)
Time Required: 2 hours
(can be split into two 60-minute sessions)
Expendable Cost/Group: US $40.00 This activity also uses some non-expendable (reusable) materials such as a water quality testing kit.
Group Size: 3
Activity Dependency: None
Subject Areas: Life Science, Measurement, Problem Solving, Science and Technology
NGSS Performance Expectations:

| HS-ESS3-4 |
| HS-ETS1-3 |
| HS-LS2-7 |
Summary
Student teams design and then create small-size models of working filter systems to simulate multi-stage wastewater treatment plants. Drawing from assorted provided materials (gravel, pebbles, sand, activated charcoal, algae, coffee filters, cloth) and staying within a (hypothetical) budget, teams create filter systems within 2-liter plastic bottles to clean the teacher-made simulated wastewater (soap, oil, sand, fertilizer, coffee grounds, beads). They aim to remove the water contaminants while reclaiming the waste material as valuable resources. They design and build the filtering systems, redesigning for improvement, and then measuring and comparing results (across teams): reclaimed quantities, water quality tests, costs, experiences and best practices. They conduct common water quality tests (such as turbidity, pH, etc., as determined by the teacher) to check the water quality before and after treatment.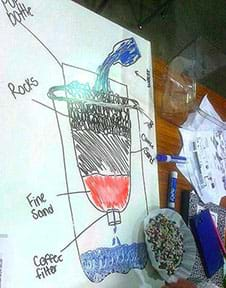
Engineering Connection
Environmental engineers design wastewater treatment plants to clean water before discharging into rivers, streams, lakes and seas. They use a variety of stages and processes to remove solids and contaminants from wastewater, including biological and mechanical processes. Filters and settling are common mechanical processes used to remove large particles from raw wastewater. The removal of plastic from a water source is a new challenge for engineers, given the fact that plastic is not soluble nor magnetic and varies in its size and decomposition state.
Learning Objectives
After this activity, students should be able to:
- Develop and test a model for an innovative and efficient water filtration system, and recover resources.
- Conduct water quality tests.
- Provide qualitative and quantitative results that describe the success (or failure) of a model filtering system in meeting the project objectives.
- Understand the steps of the engineering design process.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-ESS3-4. Evaluate or refine a technological solution that reduces impacts of human activities on natural systems. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Design or refine a solution to a complex real-world problem, based on scientific knowledge, student-generated sources of evidence, prioritized criteria, and tradeoff considerations. Alignment agreement: | Scientists and engineers can make major contributions by developing technologies that produce less pollution and waste and that preclude ecosystem degradation. Alignment agreement: When evaluating solutions it is important to take into account a range of constraints including cost, safety, reliability and aesthetics and to consider social, cultural and environmental impacts.Alignment agreement: | Feedback (negative or positive) can stabilize or destabilize a system. Alignment agreement: Engineers continuously modify these technological systems by applying scientific knowledge and engineering design practices to increase benefits while decreasing costs and risks.Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-ETS1-3. Evaluate a solution to a complex real-world problem based on prioritized criteria and trade-offs that account for a range of constraints, including cost, safety, reliability, and aesthetics, as well as possible social, cultural, and environmental impacts. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Evaluate a solution to a complex real-world problem, based on scientific knowledge, student-generated sources of evidence, prioritized criteria, and tradeoff considerations. Alignment agreement: | When evaluating solutions it is important to take into account a range of constraints including cost, safety, reliability and aesthetics and to consider social, cultural and environmental impacts. Alignment agreement: | New technologies can have deep impacts on society and the environment, including some that were not anticipated. Analysis of costs and benefits is a critical aspect of decisions about technology. Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-LS2-7. Design, evaluate, and refine a solution for reducing the impacts of human activities on the environment and biodiversity. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Design, evaluate, and refine a solution to a complex real-world problem, based on scientific knowledge, student-generated sources of evidence, prioritized criteria, and tradeoff considerations. Alignment agreement: | Moreover, anthropogenic changes (induced by human activity) in the environment—including habitat destruction, pollution, introduction of invasive species, overexploitation, and climate change—can disrupt an ecosystem and threaten the survival of some species. Alignment agreement: Biodiversity is increased by the formation of new species (speciation) and decreased by the loss of species (extinction).Alignment agreement: Humans depend on the living world for the resources and other benefits provided by biodiversity. But human activity is also having adverse impacts on biodiversity through overpopulation, overexploitation, habitat destruction, pollution, introduction of invasive species, and climate change. Thus sustaining biodiversity so that ecosystem functioning and productivity are maintained is essential to supporting and enhancing life on Earth. Sustaining biodiversity also aids humanity by preserving landscapes of recreational or inspirational value.Alignment agreement: When evaluating solutions it is important to take into account a range of constraints including cost, safety, reliability and aesthetics and to consider social, cultural and environmental impacts.Alignment agreement: | Much of science deals with constructing explanations of how things change and how they remain stable. Alignment agreement: |
Common Core State Standards - Math
-
Summarize, represent, and interpret data on a single count or measurement variable
(Grades
9 -
12)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Students will develop an understanding of the effects of technology on the environment.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop an understanding of the role of troubleshooting, research and development, invention and innovation, and experimentation in problem solving.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop an understanding of the attributes of design.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop an understanding of engineering design.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop abilities to apply the design process.
(Grades
K -
12)
More Details
Do you agree with this alignment?
State Standards
Kansas - Math
-
Summarize, represent, and interpret data on a single count or measurement variable.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
Kansas - Science
-
Design, evaluate, and refine a solution for reducing the impacts of human activities on the environment and biodiversity.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Evaluate or refine a technological solution that reduces impacts of human activities on natural systems.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Evaluate a solution to a complex real-world problem based on prioritized criteria and trade-offs that account for a range of constraints, including cost, safety, reliability, and aesthetics, as well as possible social, cultural, and environmental impacts.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- 2-liter clear plastic bottle
- 2 liters of simulated wastewater containing a predetermined volume of these (or other) contaminants-pollutants; the following mix of materials added to 1.5 liters of water yields a challenging wastewater for students:
- 5 grams (~1 tablespoon) coffee grounds
- 40 grams (~2 tablespoons) sand
- 15 grams (~1 tablespoon) vegetable oil
- 1 ounce (~2 tablespoons) liquid soap, such as hand or dish soap
- 30 grams (~2 tablespoons) fertilizer, such as 1.5 lbs of Miracle-Gro all-purpose plant food at Walmart
- 4 grams of small, brightly colored plastic beads (or similar), such as from a 100-g box of ~6,000 2-mm glass seed beads at Amazon
- safety goggles, one pair per student
- rubber gloves, one pair per student
- paper and pencils, for sketching, planning, data collection, calculating, analysis, reflection
- Wastewater Design Project Info Sheet, edit as needed to reflect budget, costs and simulated wastewater composition
To share with the entire class:
- 1-liter stream, lake, pond or lagoon water, for Introduction/Motivation demonstration; alternatively, show a liter amount of the simulated wastewater
- materials for water quality testing (as determined by the instructor), such as turbidity, pH, dissolved oxygen, nitrogen, phosphorus, conductivity or any other available tests
- a water quality testing kit such as the Earth Force Low-Cost Water Quality Monitoring Kit at Carolina.com, which contains multiple tests and student-friendly instructions
- scale, to measure in milligrams
- fine-grain sand, ~45 lbs provides ~2 kg per group for a class of 30; such as “scenic sand” at Amazon
- large gravel, ~45 lbs provides ~2 kg per group for a class of 30; such as 5 lbs “exotic 3/8-inch pebbles polished mixed gravel” for at Amazon
- small pebbles, ~45 lbs provides ~2 kg per group for a class of 30; aquarium gravel from pet stores works well, such as a 5-lb bag for at Amazon
- activated charcoal, ~45 lbs provides ~2 kg per group for a class of 30; available in pet stores, such as a 10-lb bag at Amazon
- algae, such as 50 ml of spirulina culture at Algae Research and Supply
- coffee filters, such as a pack 100 unbleached #4 size from Walmart
- cotton mesh/cheese cloth, such as 1.5-square yards at Amazon
- water and sink
- (optional) computers with Internet access, for student research
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/uok-2216-wastewater-treatment-plant-model-water-quality] to print or download.Pre-Req Knowledge
It is recommended that students complete the associated lesson prior to conducting this activity.
Introduction/Motivation
As wastewater is deposited into a river, downstream at the next town, drinking water is being pulled from the same river. So, even though drinking water is treated before it enters our tap, wastewater must also be treated and cleaned before it is returned to a river.

Let’s take a look at a sample of river water [or show some of the teacher-made “dirty water] and discuss its cleanliness. (Show students the liter of stream, lake, pond or lagoon water. Give them time to discuss among themselves. Facilitate the discussion with the following prompts.) Would you drink it? Is it safe to drink? How is this dirty water related to your drinking water? Do they come from the same place? How would you clean up the dirty water?
Just like you have been doing, environmental engineers ask themselves these questions when they design wastewater and drinking water treatment plants. How do we clean up the dirty water?
Today, you will work in teams to design a way to clean wastewater. Your engineering challenge is to use the engineering design process to clean the water as efficiently and inexpensively as possible while also reclaiming contaminants in the water—such as fertilizer and plastics—for reuse.
Each group gets a (hypothetical) budget of $1,000, which mirrors what real-life engineers encounter. They must design and build their projects within a cost constraint. To help with your overall budget, you can sell back any reclaimed materials! Let’s get started!
Procedure
Background
Similar to the Earth’s natural water cycle—composed mainly of the evaporation, condensation and precipitation processes—urban water goes through its own water cycle (see Figure 1). Water is withdrawn from surface and groundwater sources and sent to water treatment plants. After purification, the water is distributed to homes, businesses and schools. Then, no longer clean, it flows to centralized wastewater treatment plants. At these plants, contaminants and pollutants are removed from the water before discharging the now-clean water into the environment—where it may be collected by another water treatment plant downstream to serve as another community’s drinking water supply. In this cyclical process, all water is reclaimed water.
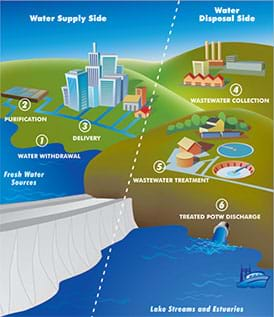
Wastewater treatment plants vary in their configuration, depending on the contaminants present in the water and applicable treatment regulations. Typically, most wastewater treatment plants are designed with the steps illustrated in Figure 2.
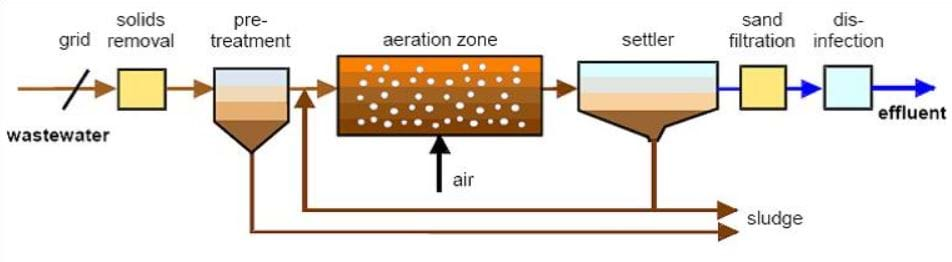
Most inorganic larger solids (such as trash from runoff) are removed in the grid and solids removal steps while the pre-treatment stage removes larger organic solids. The aeration zone is a form of biological treatment in which carbon and nutrients such as nitrogen and phosphorus are removed and become solids, which separate from the water in the settler. Sometimes, water goes through an additional sand filtration process before finally being disinfected and released as clean effluent. Sludge from pre-treatment and the settler is collected and dried. The resulting sludge may be reused as fertilizer or sent to a landfill.
In this activity, students are challenged to create filtration systems to clean the provided wastewater sample. They focus on the grid, solids removal, and pre-treatment steps.
Before the Activity
- Gather materials and make copies of the Wastewater Design Project Info Sheet. Before printing, edit as necessary to make sure the list of wastewater contaminants matches the simulated wastewater you have created (see next) and the hypothetical budget amount you are imposing as a project constraint.
- Create samples of simulated wastewater, one per group. The suggested mix (or similar) below makes the activity sufficiently challenging for students. To a 2-liter bottle containing 1.5-liters of water, add:
- 5 grams (~1 tablespoon) coffee grounds
- 40 grams (~2 tablespoons) sand
- 15 grams (~1 tablespoon) vegetable oil
- 1 ounce (~2 tablespoons) liquid soap, such as hand or dish soap
- 30 grams (~2 tablespoons) fertilizer
- 4 grams of small, brightly colored plastic beads (or similar)
- Set out filter materials (gravel, sand, etc.) in a centralized location in the classroom so that students may observe and collect them as needed throughout the activity.
- Determine which water quality tests you want students to conduct on the simulated wastewater, and have available the necessary tools and supplies. Possible tests are for turbidity, pH, nitrogen, phosphorus, dissolved oxygen and conductivity. Modify this list as you desire, depending on tests and materials available.
- Assign the pre-activity reading/recap assessment as a group homework assignment to introduce students to the wastewater topic, as described in the Assessment section. Alternatively, conduct in class if time permits.
With the Students: Project Kickoff, Research and Design
- Lead students through the Introduction/Motivation discussion, which presents the engineering challenge. Provide 5-10 minutes for students to also share and discuss their pre-activity group research findings. Explain that their pre-activity homework was step 2 of the engineering design process: researching the problem.
- Divide the class into groups of three to five students each. Have each team obtain a 2-liter sample of the simulated wastewater.
- Hand out a water quality kit/tests to each group. Direct students to test the water using the teacher-chosen water quality tests.
- How do we clean the water? Direct the groups to move into step 3 engineering design process: imagine possible solutions for how they might go about cleaning the dirty water. Provide students with information from the Teacher Background and Concepts section. Encourage students to use the Internet and other resources to come up with ideas. Hand out the info sheet, which includes a list of the simulated wastewater contaminants.
- Ask students to observe the filter material resources the teacher has made available to purify and extract contaminants from the water. Remind students of the goal: To clean the water as effectively and inexpensively as possible while also reclaiming contaminants for reuse.
- Refer to the budget information on the info sheet and quickly review it as a class. Make sure students understand that each team has a budget ($1,000), each filtering method/material has a cost, and that filtering materials and contaminants also have a post-extraction value for reuse or resale. The costs and resale values are reproduced below.
Filtration Material/Method Costs
fine-grain sand = $400/kg
large gravel = $200/kg
small pebbles = $300/kg
activated charcoal = $1,000/kg
algae = $1/mg
coffee filters = $50 each
cotton mesh/cheese cloth $300/m2
Reuse/Resale Value
clean fine-grain sand = $400/kg
clean large gravel = $200/kg
clean small pebbles = $300/kg
activated charcoal = $400/kg
fertilizer= $20/g
plastic = $10/g
- Direct the teams complete step 4 of the engineering design process: to each develop a plan to submit to the instructor in the form of a diagram and budget of how the group intends to clean the water. Expect the plan to stay under the budget set by the instructor and use the provided materials. Remind students to consider how much of the materials can be reused. As students are designing their filters, ask them the Investigating Questions.
With the Students: Step 5, 6, and 7 of the Engineering Design Process: Create, Test, Improve
- Based on the approved plan and budget, groups collect materials and construct their water treatment apparatuses. Through at least one build-test-redesign iteration, the aim is to develop a successful filter for wastewater treatment and resource reclamation.
- Once the initial water treatment system is built, students begin running the contaminated water through it. Advise students to take time to discuss their observations and make modifications to their designs, as needed. Remind students that any improvements/redesigns must stay within the budget and that unused materials may be sold back to offset expenses.
- Students conduct water quality tests on the filtered water using the same available measures and methods used for the initial contaminated wastewater. They collect and weigh the plastic beads to determine how much of the initial 4 grams they reclaimed. They measure the amount of clean water they reclaimed.
- Have teams prepare the reclaimed used materials and pollutants for instructor evaluation (and resale) by making small piles of each material.
- Direct students to clean up other supplies and turn them in to the instructor.
With the Students: Results and Evaluation
- Have students calculate the reclaimed water and plastic beads as a proportion of the initial amounts put in the simulated wastewater.
- As a class, have students compare their results about the reclaimed quantities, water quality tests, final costs and value of remaining resources across groups. Also have them share their experiences and best practices.
- To conclude, assign students to reflect on their results via the post-activity assessment, as described in the Assessment section.
Vocabulary/Definitions
dissolved oxygen : The amount of free oxygen in solution in water or wastewater effluent. Adequate concentrations of dissolved oxygen are necessary for fish and other aquatic organisms to live and to prevent offensive odors. Abbreviated as DO.
effluent: The outflowing liquid from a treatment plant to a natural body of water after completion of the treatment process.
eutrophication: The process by which a reservoir, pond or lake becomes enriched with nutrients, especially nitrogen and phosphorus, that support the excess production of algae and other aquatic plant life.
flocculation: The process by which clumps of solids in sewage are made to increase in size by chemical action.
influent: The water, wastewater or other liquid that flows into a reservoir, basin or treatment plant.
organic chemicals: A broad class of compounds that contain carbon. Smaller molecules of this class are primary components of ecological systems. Larger compounds are commonly used to create fuels, solvents, pesticides, plastics and many industrial processes.
pathogen: A disease-causing microorganism, including pathogenic bacteria, viruses, helminths and protozoans.
sedimentation: A water-treatment process during which floating wastes are skimmed off and settled solids are removed for disposal or recycling.
sludge: A thick, soft, wet mud or a similar viscous mixture of liquid and solid components, such as the product of a wastewater treatment, industrial or refining process.
valorization: To give a value to, especially a higher value.
wastewater treatment: When water used by cities or industry is sent through a series of tanks, screens, filters and other treatment actions during which pollutants are removed.
WRRRF: Acronym for water recycling and resource recovery facility. A centralized plant where water used by the people in cities or industrial processes is sent through a series of tanks, screens, filters and other treatment actions for the purpose of removing, valorizing and reusing materials.
Assessment
Pre-Activity Homework
Wastewater Background: As a homework assignment, have groups of three to five students each read one section from the EPA Primer for Municipal Wastewater Treatment Systems and recap their findings, which they share with the class to kick off the activity. Recommended distribution of sections:
- Requirements, Needs and Challenges (pages 4-6)
- Collecting and Treating Wastewater (pages 6-7, 9)
- Pollutants (page 8)
- Wastewater Treatment (pages 9-16); consider further dividing this section into Primary Treatment and Basic Wastewater Treatment Processes, Secondary Treatment and Land Treatment, and Constructed Wetlands and Disinfection
- Pretreatment and Advanced Methods of Wastewater Treatment (pages 16-19)
- The Use or Disposal of Wastewater Residuals and Biosolids (pages 19-21)
- Decentralized (Onsite or Cluster) Systems (pages 21-24)
- Asset Management (pages 24-25)
Activity Embedded Assessment
Design Filters: Observe student teams as they design successful (!) model filters for wastewater treatment and resource reclamation. Make sure teams remain within the budget and that each student contributes to the design and build.
Post-Activity Assessment
Group/Self Evaluations: Have students explain the following through discussion and/or writing. The same reflection questions are provided on the Wastewater Design Project Info Sheet.
- How effective at cleaning the water is your filter device? Be descriptive!
- Provide evidence that your device is cleaning the water. Use test data to back up your claims.
- Is this water safe to drink? Why or why not?
- How would you change your device to make it more effective?
- How could you have worked more effectively as a group to obtain higher-quality products?
Making Sense: Have students reflect on the science concepts they explored and/or the science and engineering skills they used by completing the Making Sense Assessment.
Investigating Questions
- How can we filter out resources and reuse materials from polluted water to retain optimum value? (Possible answer: Use the gravel to filter out large materials, use the coffee filter to catch small materials, etc.)
- Why do we want to separate materials for reuse? Why not just filter out and dispose everything? (Possible answer: Reasons to reuse materials are to save money by selling them to others or reusing the materials in order to offset the treatment process expenses; it often costs less to reuse materials than to obtain the same materials that have never been used before—especially if transportation costs are factored in; to be environmentally responsible, to aim for sustainability, etc.)
- Real-world wastewater treatment plants have operating costs from pumping and renewing materials; how would these costs affect your budget? How would they affect your design? (Possible answers: Operating costs take additional funds from the budget, which might force your team to choose less-expensive materials, use fewer materials, or choose higher-quality materials that last longer so new materials do not need to be purchased as often, etc.)
- Are the materials good for one use only? (Possible answers: The gravel could be used multiple times, sand might need to be cleaned before being used again, etc.)
- Which pollutants and filtering resources can be reused and resold? (Answer: Sand, large gravel, small pebbles, activated charcoal, fertilizer and plastic.)
- What methods can be used to minimize waste and retain value? (Possible answers: Use multiple filtering materials such as gravel to catch large pollutants and sand to catch small pollutants, use a coffee filter at the bottom of the filter to prevent particulates and plastics from getting through the filter, etc.)
Safety Issues
- Require the use of safety goggles.
- Wastewater that has set for more than 24 hours may become septic and require the use of gloves and more extensive sanitation measures.
- Have paper towels handy in case of spills.
Activity Extensions
Testing, redesign and revision of student filtration systems could be iterated for improvement over several days. It is up to the instructor to decide how long to carry on the project. Consider implementing a project-based approach in which students research and develop their model devices, build and test them, revise them and test again, and eventually present finished products (model processes) to industry professionals or other expert personnel outside of the school setting.
Activity Scaling
- For beginning courses, have students carry out simple water tests such as mass of sediment, pH and turbidity measured qualitatively. Help younger students with the water quality tests or conduct tests as a teacher demonstration.
- For advanced courses, have students carry out more analytical testing procedures such as conductivity and turbidity using probes and chemical assessment of nitrogen and phosphorus using photometric methods.
Additional Multimedia Support
Primer for Municipal Wastewater Treatment Systems by the U.S. Environmental Protection Agency, 2004, at https://www.epa.gov/sites/production/files/2015-09/documents/primer.pdf
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn how plastics in the human trash stream end up as microplastic particles entering the food chains via polluted water, harming animals and people. They think of ways to reuse or replace the common plastic items they discard daily. They learn how microplastics persist in the environment ...
Copyright
© 2018 by Regents of the University of Colorado; original © 2017 University of KansasContributors
David Bennett; Sara Hettenbach; William WelchSupporting Program
SHIFTED RET Program, University of Kansas; Southeast Kansas Education Service Center-GreenbushAcknowledgements
This curriculum was based upon work supported by the National Science Foundation under RET grant no. EEC 1301051—Shaping Inquiry from Feedstock to Tailpipe with Education Development (SHIFTED) through the Center for Environmentally Beneficial Catalysis, hosted by the University of Kansas Lawrence and the Southeast Kansas Education Service Center (referred to as Greenbush). Any opinions, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation.
Special thanks to Belinda Sturm, Samik Bagchi, Robert Everhart and Rachel Bowes.
Last modified: August 16, 2023




User Comments & Tips