Quick Look
Grade Level: 5 (4-6)
Time Required: 45 minutes
Expendable Cost/Group: US $3.00
Group Size: 4
Activity Dependency: None
Subject Areas: Algebra
NGSS Performance Expectations:

| MS-ESS2-5 |

Summary
Students use a sponge and water model to explore the concept of relative humidity and create a percent scale. Through this experiment, teams collect data, make calculations and draw conclusions. They gain clarification in the difference between relative and absolute humidity. They also learn real-world reasons why understanding the concept of humidity is important for engineers. A student activity worksheet is provided.Engineering Connection
Understanding the science of humidity is important to many engineers. Environmental engineers consider the specific weather and atmospheric conditions of an area when researching air pollutant transport and concentrations. Industrial engineers find that the amount of water in the air affects how difficult or easy it is to remove pollutants from the air. Civil engineers carefully control indoor humidity levels, as needed; high humidity can affect electronics equipment, paper (starts to curl), food storage and lab experiments (mold growth), in addition to creating an uncomfortable work environment and moist conditions that cultivate poor air quality.
Learning Objectives
After this activity, students should be able to:
- Define and explain the terms humidity and saturation.
- Build a simple humidity/saturation model.
- Use data from a model to create a percent scale.
- Explain how meteorologists and engineers use humidity information.
- Explain how humidity affects local weather conditions.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ESS2-5. Collect data to provide evidence for how the motions and complex interactions of air masses results in changes in weather conditions. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Collect data to produce data to serve as the basis for evidence to answer scientific questions or test design solutions under a range of conditions. Alignment agreement: | The complex patterns of the changes and the movement of water in the atmosphere, determined by winds, landforms, and ocean temperatures and currents, are major determinants of local weather patterns. Alignment agreement: Because these patterns are so complex, weather can only be predicted probabilistically.Alignment agreement: | Cause and effect relationships may be used to predict phenomena in natural or designed systems. Alignment agreement: |
Common Core State Standards - Math
-
Represent real world and mathematical problems by graphing points in the first quadrant of the coordinate plane, and interpret coordinate values of points in the context of the situation.
(Grade
5)
More Details
Do you agree with this alignment?
-
Find a percent of a quantity as a rate per 100 (e.g., 30% of a quantity means 30/100 times the quantity); solve problems involving finding the whole, given a part and the percent.
(Grade
6)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Explain how various relationships can exist between technology and engineering and other content areas.
(Grades
3 -
5)
More Details
Do you agree with this alignment?
State Standards
Colorado - Math
-
Visual displays are used to interpret data.
(Grade
5)
More Details
Do you agree with this alignment?
-
Find a percent of a quantity as a rate per 100.
(Grade
6)
More Details
Do you agree with this alignment?
Colorado - Science
-
Describe weather conditions based on data collected using a variety of weather tools
(Grade
5)
More Details
Do you agree with this alignment?
-
Use data collection tools and measuring devices to gather, organize, and analyze data such as temperature, air pressure, wind, and humidity in relation to daily weather conditions
(Grade
5)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- 1 dry sponge
- 1 plate, plastic or aluminum
- 1 plastic spoon
- a cup
- water, 1 cup
- paper and pencil, to keep a tally
- Sponge Saturation Worksheet, one per student
- Relative Humidity Graph, as a handout for each group or each student, or as an overhead projector transparency to show the class
- (optional, if students are graphing data) graph paper, one sheet per student
For the entire class to share:
- the day's humidity, such as from the newspaper weather page, television or radio weather broadcast, or Internet weather website
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/cub_air_lesson04_activity3] to print or download.Introduction/Motivation
Under usual Earth conditions, water exists in any of three physical states: solid (ice, snow), liquid (which we typically call water) or gas (humidity, water vapor [steam]). According to meteorologists, humidity can be absolute or relative, giving us the terms absolute humidity and relative humidity. In common usage, the adjective is often dropped, and the speaker (or writer) just says "humidity," expecting us to know of which type they are speaking. Even some television and radio weathercasters, including those formally trained in meteorology, often do not clarify the difference when reporting the weather conditions.
Absolute humidity is defined as the ratio of the mass of water vapor contained per volume of moist air. Relative humidity, technically, is the ratio between the partial pressure of water in the air and the maximum possible vapor pressure of water at a particular temperature. It is dimensionless. Relative humidity is usually what the media announcers mean when they say "humidity," and it is useful in determining conditions for human comfort. But, it can be confusing because its value varies with air temperature. For example, the morning may have a relative humidity of 78%, which by afternoon drops to 53% as the air temperature rises. (Note: the absolute humidity for that day remained essentially constant, but the vapor pressure of water increased with the temperature). Similarly in winter, the outdoor relative humidity may be 63%, but when outdoor air permeates our warm homes and offices, the relative humidity level may drop to 35% or lower. (In this example, the absolute humidity is quite low in the outdoor air, but the vapor pressure of water at cold temperatures is also low, thus, the outdoor air is more humid, relatively speaking.) See Figure 1.
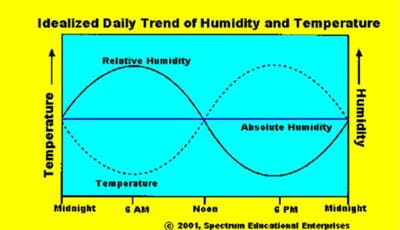
Understanding the concept of saturation is important to understanding relative humidity. Saturation is defined as the condition in which air at a specific temperature contains all the water vapor it can hold; 100% relative humidity. At saturation, the partial pressure of water vapor in the atmosphere is at its maximum level for the existing ambient temperature and pressure. At saturation, equilibrium exists between water vapor and liquid water, and there is no net evaporation or condensation. Above saturation, some of the water in the air condenses to form droplets or clouds (collections of droplets). By better understanding humidity, one can better understand local weather. Given the temperature of a volume of air and its pressure, we can determine a saturation value. We can saturate a parcel of air by adding more water vapor to it (through evaporation or mixing with another parcel of more humid air), or by cooling the parcel down to its saturation temperature. Both processes are at work continually in the atmosphere, but the latter is more familiar to us as it forms fog or dew (or frost if cold enough). In this activity, we talk about the temperature of the air affecting the vapor pressure, but it is technically the temperature of the water molecules that determine vapor pressure. For a good discussion of this, see Professor Alistair Fraser's comments at http://www.ems.psu.edu/~fraser/Bad/BadClouds.html.
The saturation temperature of the ambient air is commonly called the dew point temperature or simply the dew point (or frost point if it is below freezing). You might hear a weathercaster or meteorologist discuss the dew point of a particular air mass. Dew point, like absolute humidity, varies little within an air mass. When the ambient air temperature equals the dew point, the relative humidity is 100% and the air is saturated. If the air temperature falls lower, water vapor begins to condense into very small liquid droplets to form clouds or fog (if it is near the ground). If the surface temperature of an object (vegetation, rooftop or car exterior) falls below the dew point while most of the surrounding air remains above it, dew forms through the condensation of water vapor onto that surface (or ice crystals, we call it frost, if it is below freezing).
Procedure
Before the Activity
- Gather materials and make copies of the Sponge Saturation Worksheet.
- Find the day's humidity measurement.
With the Students
- Ask students: What is humidity? How does it affect the weather? Have students brainstorm and write their ideas on the board.
- Divide the class into groups of four students each. Distribute supplies (1 dry sponge on a plate, a cup of water, 1 plastic spoon and a copy of the worksheet; see Figure 2).
- Have students squeeze the sponges to verify that they contain no water. Ask: How much water is in the sponge? (Answer: None)
- One spoonful at a time, direct students to slowly and carefully pour water onto the sponges. Have them count how many spoonfuls of water are being added; it helps to have one group member keep a tally (see Figure 1).

- After a few spoonfuls, stop the students and ask them the following questions: What is happening to the water? Where is it going? What do you think is going to happen as you keep filling the sponge with water? Can you put water into this sponge forever? Will we be counting forever?
- Have students resume adding water to the sponge by the spoonful until the sponge starts to drip water (reaches saturation). Ask the students to explain what has happened to the sponge. Why is water dripping from it?
- Explain that the sponge is like the air. Ask: How does the water dripping from the sponge act like rain or a cloud? Explain that air can "hold" water, too, similar to the sponge. Ask if anyone knows another name that means something is full (of water, like the air or the sponge). Share the word "saturated." Explain that when something (air or the sponge) is full (of water), we say that it is saturated. Write the following statement on the board:
sponge full (100% saturated) = ___ spoonfuls of water
- Ask students to share how many spoonfuls it took to reach 100% saturation of their team's sponge. (If differences among the group data; lead a discussion about why the differences exist. Does it have to do with experimental apparatus, experimental procedure, etc.?) Have students record on their worksheets the number of spoonfuls to reach 100% saturation.
- Ask students if they know how many tablespoons of water had been poured into the sponge when it was halfway full of water? Write the following statement on the board:
sponge half full (50% saturated) = ___ spoonfuls of water
- Ask students how many spoonfuls of water were in the sponge when the sponge was empty? Write the following statement on the board:
sponge empty (0% saturated) = 0 spoonfuls of water
- Have students begin to complete their worksheet data tables with 0 spoonfuls of water and 0% saturation.
- Ask them to write the number of spoonfuls (up to the 100% saturation number) and calculate each of the corresponding % saturations. (If you want the students to graph the data, have them do it now).
- Ask students to refer to their worksheet data tables (and/or graphs) to answer the following questions:
- How many tablespoons of water would be in the sponge if the sponge were nearly ready to drip, but not dripping yet?
- How many tablespoons of water would be in the sponge if it were nearly empty, but not totally empty?
- Ask students to make other predictions from their data/graphs (such as, how many spoonfuls are required to reach 40% saturation? What would happen if you added more spoonfuls than are required to reach 100% saturation? etc.)
- Explain that they just used a scale to measure/predict how much water is in their sponges and how close they are to dripping. Meteorologists can measure how close is it to raining by using a humidity scale for water present in the air.
- Display the Relative Humidity Graph either by handout or overhead projector.
- Have students make comparisons to the data (and graph) just collected. (Note that the graph handout is much more complex and is dependent on temperature.)
- In conclusion, ask the students:
- If it is almost ready to rain, what is the humidity? (Expect them to inquire: Are you talking about relative or absolute humidity? Answer: Close to 100% relative humidity.)
- If the air is nearly, but not completely dry, what is the humidity? (Answer: Lower, but it depends on the temperature.)
- If the air was halfway full of water or halfway saturated, what is the humidity? (Answer: Encourage them to look along the 50% relative humidity line and see how it changes with temperature. Notice the dew points at each of these temperatures.)
- Why is humidity important to engineers? (Answer: Because it helps to predict the weather, which affects pollutant transport and concentrations, and the amount of water in the air affects how difficult/easy it is to remove pollutants from the air. Engineers are also concerned with humidity levels indoors. Buildings with high humidity are not good for storing food or electronics equipment; even copier paper starts to curl. Laboratories also control humidity levels so that the moisture does not affect their experiments. High humidity levels also cause bacteria and molds to grow, which can affect indoor air quality.)
Vocabulary/Definitions
absolute humidity: The ratio of the mass of water vapor contained per volume of moist air.
ambient: Surrounding or encircling, such as ambient air.
dew point: The saturation temperature of ambient air. Also called dew point temperature.
humidity: Dampness; the amount of water vapor in the air.
meteorologist: A scientist who studies meteorology (the atmosphere, weather and weather forecasting).
partial pressure: The pressure that one component of a mixture of gases would exert if it were alone in a container.
relative humidity: The ratio between the partial pressure of water in the air and the maximum possible vapor pressure of water at a particular temperature; it is dimensionless.
saturation: The condition in which air at a specific temperature contains all the water vapor it can hold; 100% relative humidity. The condition when the partial pressure of water vapor in the atmosphere is at its maximum level for the existing ambient temperature and pressure. At saturation, equilibrium exists between water vapor and liquid water, and there is no net evaporation or condensation.
Assessment
Pre-Activity Assessment
Brainstorming: As a class, have students engage in open discussion. Remind them that no idea or suggestion is "silly." All ideas should be respectfully heard. Write their ideas on the board. Ask the students:
- What is humidity?
- How does it affect the weather?
- How are clouds formed? (Answer: Warm air near the land (or water) surface rises. This rising, and subsequent pressure drop, results in the air expanding, thereby using some energy of the molecules, and cooling the air. At these lower temperatures, some of the water vapor in the air condenses to form clouds or rain. See Figure 3 for more information.)
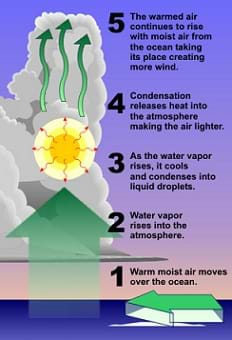
Figure 3. The process of cloud formation.
Activity Embedded Assessment
Worksheets: Have students use the Sponge Saturaion Worksheet and Relative Humidity Graph, as directed in the Procedure section, to follow along with the activity. Review their data and calculations (and graphs) to gauge their engagement and comprehension.
Post-Activity Assessment
Question/Answer: Ask students the questions at the end of the Procedure section, and discuss as a class.
Safety Issues
To prevent slipping, minimize water dripping onto floors and keep paper towels handy. Warn students that the floors may be slippery when wet.
Troubleshooting Tips
Try the activity in advance, so that you have a general idea of how many spoonfuls of water it takes to saturate the sponges. To speed up the activity, cut the sponges in half.
Activity Extensions
Have students make graphs of the sponge saturation data. Ask them to make predictions from the graph.
Consider using a simple computer application spreadsheet with students sot they can quickly enter data, calucate percentages, and create graph.
Gather humidity data from different areas around the globe (or perhaps a specific area the class is studying in social studies) and discuss/compare the information to the activity data.
Have students draw the mechanisms of cloud formation and how air behaves as it rises.
Activity Scaling
- For grades 3 and 4, help students calculate percentages (especially if they are rounding them).
- For grades 1 and 2, students should be able to collect data and create graphs without discussing percentages.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!References
Heidorn, Keith C. Laying Some Groundwork-2: Humidity, Science of the Sky. Suite101.com. Accessed August 11, 2004. http://www.suite101.com/article.cfm/13646/82259
Fraser, Alistair B. "Bad Clouds, Bad Meteorology: The reason clouds form when air cools is because cold air cannot hold as much water vapor as warm air." Updated November 27, 1994. The College of Earth and Mineral Sciences, Pennsylvania State University. Accessed August 11, 2004. http://www.ems.psu.edu/~fraser/Bad/BadClouds.html
Copyright
© 2004 by Regents of the University of ColoradoContributors
Amy Kolenbrander; Daria Kotys-Schwartz; Janet Yowell; Natalie Mach; Malinda Schaefer Zarske; Denise W. CarlsonSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under grants from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and National Science Foundation (GK-12 grant no. 0338326). However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: May 25, 2017


User Comments & Tips