Quick Look

Summary
Students explore the natural phenomenon of surface tension and learn how it is applied in industry, such as food science, and in medical contexts, including disease progression from vaping-related lung injury. Students design and test candy-coating techniques as a model to investigate how surfactants affect liquid spreading and adhesion. They then connect this model to biology by examining how natural lung surfactants function in respiration and how vaping additives can disrupt alveolar coating efficiency, linking engineering design to real-world health challenges.Engineering Connection
Chemical and biomedical engineers use science, math, and engineering to design solutions that improve products, processes, and human health. Chemical engineers develop and optimize materials, coatings, and additives used in industries such as food science, medicine, and manufacturing, while biomedical engineers apply these principles to study how materials and chemicals interact with the human body. Together, they investigate how surfactants control liquid behavior, support lung function, and influence medical treatments, including understanding and preventing lung injuries caused by harmful substances like vaping additives.
Learning Objectives
After this activity, students should be able to:
- Describe how surfactants affect the surface tension of liquids and influence fluid behavior.
- Explain the role of surfactants in the lungs and how they support normal breathing.
- Investigate how additives influence surface tension and coating performance using a physical model.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-ETS1-2. Design a solution to a complex real-world problem by breaking it down into smaller, more manageable problems that can be solved through engineering. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Design a solution to a complex real-world problem, based on scientific knowledge, student-generated sources of evidence, prioritized criteria, and tradeoff considerations. Alignment agreement: | Criteria may need to be broken down into simpler ones that can be approached systematically, and decisions about the priority of certain criteria over others (trade-offs) may be needed. Alignment agreement: | |
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-ETS1-3. Evaluate a solution to a complex real-world problem based on prioritized criteria and trade-offs that account for a range of constraints, including cost, safety, reliability, and aesthetics, as well as possible social, cultural, and environmental impacts. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Evaluate a solution to a complex real-world problem, based on scientific knowledge, student-generated sources of evidence, prioritized criteria, and tradeoff considerations. Alignment agreement: | When evaluating solutions it is important to take into account a range of constraints including cost, safety, reliability and aesthetics and to consider social, cultural and environmental impacts. Alignment agreement: | New technologies can have deep impacts on society and the environment, including some that were not anticipated. Analysis of costs and benefits is a critical aspect of decisions about technology. Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-LS1-2. Develop and use a model to illustrate the hierarchical organization of interacting systems that provide specific functions within multicellular organisms. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Develop and use a model based on evidence to illustrate the relationships between systems or between components of a system. Alignment agreement: | Multicellular organisms have a hierarchical structural organization, in which any one system is made up of numerous parts and is itself a component of the next level. Alignment agreement: | Models (e.g., physical, mathematical, computer models) can be used to simulate systems and interactions—including energy, matter, and information flows—within and between systems at different scales. Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-PS2-6. Communicate scientific and technical information about why the molecular-level structure is important in the functioning of designed materials. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Communicate scientific and technical information (e.g. about the process of development and the design and performance of a proposed process or system) in multiple formats (including orally, graphically, textually, and mathematically). Alignment agreement: | Attraction and repulsion between electric charges at the atomic scale explain the structure, properties, and transformations of matter, as well as the contact forces between material objects. Alignment agreement: | Investigating or designing new systems or structures requires a detailed examination of the properties of different materials, the structures of different components, and connections of components to reveal its function and/or solve a problem. Alignment agreement: |
Common Core State Standards - Math
-
Reason abstractly and quantitatively.
(Grades
K -
12)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Synthesize data and analyze trends to make decisions about technological products, systems, or processes.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Apply principles of human-centered design.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Evaluate ways that technology can impact individuals, society, and the environment.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Develop a plan that incorporates knowledge from science, mathematics, and other disciplines to design or improve a technological product or system.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
Soap Motor Activity
- Soap Motor Phenomenon Worksheet (1 per student)
- 1 tray or wide container
- 1 cotton swab
- 1 pair of scissors (to cut out the boat, if not already cut out)
- 1 boat or small leaf (from Boat Template sheet)
- 1 container (e.g., beaker or graduated cylinder or measuring cup) to hold water
- 2-4 drops of liquid soap
- colored water (enough to cover the bottom of the tray/wide container)
Hydro Dipping Activity
- Student Worksheet (1 per student)
- Real-World Connection Worksheet (1 per student)
- 2-3 pre-made cake pops, marshmallows, or fun-sized candy bars (at least 1 per person)
- 1-2 toothpicks or skewers (for holding/dipping treats; at least 1 per person)
- 1 small plate or wax paper (for drying the treat after dipping; at least 1 per person)
- 1 dipping trough/small container (~6–8 cm deep to fully submerge treat; could be a small Tupperware or similar)
- 100-150 mL water/corn syrup bath (assigned concentration: 33% or 66% or 100% corn syrup)
- ~50 g candy melts of varying colors
- 1-2 tsp additives (coconut oil, food-grade lecithin, or vegetable oil)
- 1 drizzle spoon, teaspoon or a pipette
- 1 scale/balance
- 5-10 food safe weigh boats/paper
- 1 graduated cylinder (for measuring the volume of candy melt the spoon can hold)
- 1 spoon
- food-safe gloves (1 per student)
- (optional) 1 food thermometer and heat source (if candy melts need melting)
- (optional) apron (1 per student)
For the class to share:
- 1 laptop or tablet with projector and internet access (to show YouTube videos)
- paper towels to clean up spills
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/uok-3030-sweet-surfactants-surface-tension-activity] to print or download.Pre-Req Knowledge
Students should have:
- A basic understanding of molecules.
- A basic understanding of how to measure volume and mass.
- The ability to do computational work (adding, multiplying, calculating percentages).
- Familiarity with best practices for collecting data, both qualitative and quantitative.
Introduction/Motivation
Take a look at this image. (Project Image 1 or another similar image of an insect or arachnid on the surface of water for the class to see.) What do you notice about this image? What do you wonder about this image? List some qualitative data and some quantitative data about the image. (Allow students 1-2 minutes to discuss with their partners/groups.)

Let’s share some ideas with the class. Who wants to give one qualitative observation? (Possible qualitative responses: The spider looks as if it is walking on the water. The water looks like it is bending where the spider legs touch the water.) Now, who has a quantitative observation? (Possible quantitative response: The spider has 6 of its 8 legs on the surface of the water but is not sinking.)
What you’re seeing is called surface tension. Water molecules at the surface hold tightly to each other, forming a kind of invisible skin. This is why small insects can walk on water without sinking. Surface tension is not just a neat trick in nature; it’s important in many systems, including biology and medicine.
However, surface tension can be broken easily with a common material: dish soap! Soap disrupts the forces holding the water molecules together, reducing the surface tension. Soap is a surfactant, short for surface-active agent, which means it is composed of molecules that reduce the surface tension of a liquid. Why are we interested in this? In our lungs, there’s a similar process. Tiny air sacs called alveoli are coated with a thin layer of fluid. To prevent these sacs from collapsing, our bodies produce surfactants that allow the alveoli to stay open and make breathing easier.
It turns out that some additives, such as those found in vaping products, can interfere with lung surfactants. When surfactants don’t work properly, the alveoli are more likely to collapse. This makes breathing harder and can contribute to lung injury resulting in a condition called EVALI (e-cigarette or vaping associated lung injury).
Today, we are going to start an activity that investigates surface tension and surfactants. We’ll use a hands-on model with candy melts to understand how surfactants coat surfaces, and how additives can affect them, just like what can happen in the lungs with vaping.
Let’s get started with our Sweet Surfactants activity!
Procedure
Background
Surface tension is a fundamental physical phenomenon that arises from cohesive forces between liquid molecules at the interface between a liquid and another medium, typically air. These intermolecular attractions create a “skin-like” surface that resists external forces, allowing liquids to form droplets, support lightweight objects, and maintain their shape in small-scale systems. Surface tension plays a critical role in both natural and engineered processes, governing capillary action in plants and soil, enabling insects such as water striders to walk on water, and controlling the behavior of bubbles, droplets, and thin liquid films. At small scales, surface forces dominate over gravity, making surface tension especially important in applications such as microfluidics, drug delivery, coating technologies, and biomedical engineering. Understanding surface tension allows students to connect molecular-scale interactions to observable, real-world phenomena.
Surfactants are substances that reduce surface tension by disrupting the cohesive forces between liquid molecules. These molecules contain both hydrophilic (water-attracting) and hydrophobic (water-repelling) regions, enabling them to position themselves at liquid interfaces and modify fluid behavior. In biological systems, surfactants play a vital role. In the lungs, pulmonary surfactant coats the alveoli—tiny air sacs responsible for gas exchange—and reduces the surface tension of the thin water layer lining their surfaces. Without this surfactant, alveoli would collapse during exhalation, making breathing extremely difficult. By lowering surface tension, lung surfactants stabilize alveoli, decrease the energy required for breathing, and support efficient oxygen transfer into the bloodstream.
Vaping is currently the most common form of tobacco product use among middle and high school students and is considered a gateway to traditional cigarette smoking. Many vaping products contain additives such as CBD, vitamin E acetate, and THC, which have been linked to cases of e-cigarette or vaping-associated lung injury (EVALI). Ongoing research is investigating how these additives interact with lung surfactants and affect respiratory function. Early findings suggest that some additives may interfere with the ability of pulmonary surfactants to reduce surface tension in the alveolar fluid, which can impair normal breathing and contribute to lung injury.
In this activity, students investigate surface tension and surfactant behavior using candy melts and syrup–water baths as a physical analogy for lung surfactants. Candy melts, which are primarily composed of fats and oils, function similarly to surfactants by influencing how liquids spread, adhere, and transfer onto solid surfaces. Through hydro-dipping treats, students directly observe how variations in fluid composition, viscosity, and additive concentration affect coating quality. In the extension portion of the lesson, students modify the concentration of additives such as coconut oil, vegetable oil, and lecithin in the candy melts to optimize coating performance. This models how chemical additives in vaping liquids may alter lung surfactant function, providing a meaningful connection between engineering design, chemistry, biology, and public health.
Before the Activity
For Part 1:
- Make copies of:
- Soap Motor Phenomenon Worksheet (1 per student).
- Boat Templates (2 for the entire class).
- Gather materials for the Soap Motor Phenomenon Activity.
- Set up laptop/tablet to show three YouTube videos:
- "Surface Tension of Water Explained” (https://www.youtube.com/watch?v=i2TguDlE680; 3:36 minutes)
- “Surfactants Mechanism of Action” (https://www.youtube.com/watch?v=UU9ij_z6jXQ, 3:42 minutes)
- “Surfactant and Surface Tension in Respiration” (https://www.youtube.com/watch?si=gTt_NjR69DUQSEyG&v=7ZMweT5o3Io&feature=youtu.be, 4:21 minutes)
For Part 2:
- Make copies of the Student Worksheet (1 per student).
- Gather materials for the Sweet Surfactants Activity.
- Prepare enough pre-made treats: cake pops, marshmallows, fun-sized candy bars, etc.
- Prepare corn syrup/water baths at assigned concentrations in separate containers. See Teacher Tips.
- 33% syrup: 33 mL corn syrup + 67 mL water
- 66% syrup: 66 mL corn syrup + 34 mL water
- 100% syrup: 100 mL corn syrup only
- Prepare candy melts in various colors. See Teacher Tips.
- Prepare additives: coconut oil, vegetable oil, lecithin.
- Prepare Class Data Sheet to record group data.
- Optional: Prepare any demonstration materials or sample setups for teacher-led demos.

For Part 3:
- Make copies of the Real-World Connection Worksheet (1 per student).
- Prepare additional candy melts and treats for repeated testing/revisions.
- Set up laptop/tablet to show YouTube video: “What Vaping Does to Your Body” (https://www.youtube.com/watch?v=IaVaoja2Uk4, 14:47 minutes). Cue to show video from minutes 3:30 –9:00.
During the Activity
Part 1: Introduction and Soap Motor Phenomenon Activity (~ 60 minutes)
- Introduce the activity with the Introduction and Motivation section.
- Organize students into groups of 2–4, depending on class size.
- Give one Soap Motor Phenomenon Worksheet to each student.
- Instruct students to follow the instructions (listed below) on the sheet up to the “Pause Here” instruction.
- Students gather materials for the activity.
- Students set up the activity and make their observations of the boat with and without the soap.
- Students record their observations in their Soap Motor Phenomenon Worksheet.
- Students clean up their materials.
- Students write down three questions they have about the boat before and after the soap.
- Have students share their findings with their groups and the class with the following prompts:
- What did you observe about the boat on the water?
- What happened when you put the soap behind the boat?
- Why do you think the boat acted the way it did?
- What do you think is happening at the molecular level in the water when the soap touches it?
- Instruct students to get ready to take notes as you show videos on surface tension, surfactants, and respiration.
- Play the following YouTube videos:
- "Surface Tension of Water Explained” (https://www.youtube.com/watch?v=i2TguDlE680; 3:36 minutes)
- “Surfactants Mechanism of Action” (https://www.youtube.com/watch?v=UU9ij_z6jXQ, 3:42 minutes)
- “Surfactant and Surface Tension in Respiration” (https://www.youtube.com/watch?si=gTt_NjR69DUQSEyG&v=7ZMweT5o3Io&feature=youtu.be, 4:21 minutes)
- Lead a class discussion summarizing key points of the three videos. For example:
- “Surface Tension of Water Explained”
- Water molecules attract each other through cohesive forces, especially hydrogen bonds.
- Molecules at the surface are pulled inward, creating a “tight skin” effect.
- Surface tension allows water to form rounded droplets.
- Small objects denser than water can sometimes float if they don’t break the surface layer.
- “Surfactants Mechanism of Action”
- Surfactants are molecules with two ends: one attracted to water, and one repelled by water.
- Surfactants insert themselves at the water surface.
- They disrupt cohesive forces between water molecules.
- This reduces surface tension, making liquids spread more easily.
- Surfactants are responsible for better interaction between liquids and solids in experiments.
- "Surfactant and Surface Tension in Respiration”
- Alveoli in the lungs are lined with water, which has high surface tension.
- High surface tension would make alveoli collapse and harder to inflate.
- The lungs produce a natural surfactant to lower surface tension.
- Lower surface tension helps alveoli remain open and makes breathing easier.
Part 2: Sweet Surfactants Activity (~ 60 minutes)
- Give one Student Worksheet to each student.
- State the objective: You will investigate how additives affect surface tension using two liquids interacting with a solid to coat your delectable treat.
- Read through the Materials and Procedure of the worksheet with the students.
- Have students record the following in their worksheet data table:
- Hypothesis
- Assigned corn syrup bath %
- Assigned candy melt mix
- Chosen technique for dipping
- Dispense materials to each group.

- Remind students to record their initial masses.
- Optional: Demonstrate how to drizzle the candy melts on the corn syrup bath.

- Let students follow the directions in the worksheet to dip and set their treats before measuring mass again.
- Instruct students to clean up their group workspace. (Note: Do not put candy melts in or down the drain).
- Display the Class Data Sheet, where each group can share their data with the class.
- As a class, discuss the groups’ findings. Potential findings include:
- Surface tension: Lower values mean better spread.
- Spread diameter: Wider means better wetting.
- Coating mass: Higher mass means better adhesion.
- Drips/Smears: Fewer drips/smears means cleaner coating.
- Percent coverage: Higher means better transfer.
- Higher syrup concentration has higher density and viscosity.
- Higher syrup concentration improves the support of the candy melt.
- Additive: Oil
- Lowers the candy melt surface tension
- Improves spread
- May make the coating thin
- Additive: Lecithin
- Improves interaction
- Improves adhesion
- Increases coating uniformity
- State the challenge: Design and optimize a hydro-dipping coating system that produces the most uniform, clean, and effective candy coating.
- Remind students to use their understanding of surface tension, surfactants, and fluid interactions to optimize their design.
- Emphasize success criteria: uniform coating, minimal drips, optimal spread, and effective adhesion.
- Give groups time to brainstorm and plan design variations to optimize their coating system for the challenge.
Part 3: Hydro-Dipped Design Challenge and Relating to Biology (~60 minutes)
- Have the students test their candy-coating design. Students should:
- Dip their treat(s) using their updated technique and/or additive combination.
- Observe how the coating behaves: Does it spread more evenly? Stick better? Drip less?
- Record data for each revision in their worksheet: mass, percent coverage, spread diameter, drips/smears, etc.

- Encourage groups to compare their results to the first trial and note improvements or unexpected outcomes.
- Circulate and ask guiding questions:
- What changed in your coating compared to the previous attempt?
- Which additive or technique seems most effective, and why?
- How does surface tension or fluid interaction explain these results?
- Optional: Let students improve their design and retest.
- Relate the candy coating back to surfactants coating the alveoli by leading a short discussion connecting the activity to biology:
- Ask students to recall the “Surfactant and Surface Tension in Respiration” video.
- Draw the analogy: Just like surfactants help liquid coat alveoli evenly and prevent collapse, the additives in candy melts help the coating spread and adhere uniformly.
- Discuss why uniform coatings are important in real-world applications, e.g., medical, industrial, or food science.
- Hand out the Real-World Connection Sheet and explain that this sheet links the activity to health and engineering applications.
- Have students complete the sheet individually or in pairs, answering questions such as:
- How does understanding surface tension and surfactants help engineers and scientists?
- What parallels exist between coating candy and coating biological tissues?
- How might these principles apply to other industries (pharmaceutical, aerospace, food, etc.)?
- Show the video clip from “What Vaping Does to Your Body” (https://www.youtube.com/watch?v=IaVaoja2Uk4, 14:47 minutes) Note: Play the clip from minutes 3:30–9:00, noting that it contains a cadaver sagittal section of the head and lungs.
- While watching, have students take notes on how surfactants in the lungs are affected by vaping, and any consequences for respiratory function.
- After the clip, facilitate a discussion:
- How does vaping disrupt natural surfactant function?
- How might this relate to what they observed in their candy-coating lab?
- Why is understanding surface tension and surfactants important in medicine and public health?
- Ask each group to summarize their findings:
- What worked best in their candy-coating revision, and why?
- How does this connect to surfactants in the lungs?
- What surprised them about the interaction of liquids with solids?
Vocabulary/Definitions
additive: A substance added to a product to change or improve its properties.
adhesion: The attraction between different substances, such as liquid sticking to a solid surface.
alveoli: Tiny air sacs in the lungs where oxygen and carbon dioxide are exchanged.
coating: A layer of material applied to cover or protect a surface.
cohesion: The attraction between molecules of the same substance, such as water molecules sticking together.
hydro-dipping: A coating process where an object is dipped into a liquid to evenly coat its surface.
hydrogen bonding: A weak type of attraction between a hydrogen atom in one molecule and a highly electronegative atom (such as oxygen or nitrogen) in another molecule; important for water’s properties.
interface: The boundary where two different materials, substances, or phases meet and interact.
lung surfactant: A natural substance in the lungs that reduces surface tension, preventing alveoli from collapsing and making breathing easier.
respiration: The process of breathing and exchanging oxygen and carbon dioxide in the body.
surface tension: The force that causes the surface of a liquid to act like a stretched elastic skin due to attraction between molecules.
surfactant: A substance that reduces surface tension, allowing liquids to spread more easily and interact better with solids or other liquids.
viscosity: A measure of how thick or resistant to flow a liquid is.
wetting: The ability of a liquid to spread across and stick to a solid surface.
Assessment
Pre-Activity Assessment
Phenomenon Activity: Students complete the Soap Motor Phenomenon Worksheet so you can assess their background knowledge of surface tension and surfactants.
Activity Embedded (Formative) Assessment
Data Collection and Sharing: During the activity, students record observations and measurements (e.g., candy coating mass, coverage, spread, drips) in their worksheets. They then share their data with the class, allowing you to monitor understanding, provide feedback, and guide discussion on trends, patterns, and scientific reasoning.
Post-Activity (Summative) Assessment
Real-World Connections: Students complete the Real-World Connection Sheet, which allows you to evaluate their understanding, application, and reasoning skills.
Safety Issues
- If you are letting your students eat the treats, be sure to use food-safe equipment.
- Check for any food allergies among your students before doing this activity and supply nut-free alternatives for treats.
Troubleshooting Tips
- If the soap doesn’t make the boat move:
- Check that the water surface is clean and free of debris or oils.
- Ensure the boat is light enough to float and not waterlogged.
- Use the correct amount of soap (2–4 drops) behind the boat.
- If candy melts don’t spread evenly:
- Adjust the dipping technique (speed, angle, or swirling motion).
- Check the syrup bath concentration—higher concentrations may better support coating.
- Mix in small amounts of additives (oil or lecithin) to improve spread or adhesion.
- If treats break or fall off skewers:
- Make sure the skewer is properly inserted and stable.
- Avoid dipping treats that are too soft or fragile.
- If coating drips or smears:
- Reduce the amount of candy melt applied at once.
- Let excess coating drip back into the container before placing on drying surface.
- If data seems inconsistent:
- Double-check measurements (mass, volume, spread diameter).
- Confirm all groups are using the same syrup concentration and candy melt type.
- Refer to the Teacher Tips for further trouble shooting.
Activity Extensions
- Challenge them to prepare their own corn syrup baths by calculating concentrations using:
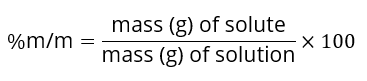
- Allow students to experiment with multiple syrup concentrations (e.g., 33%, 66%, 100%) and different additives simultaneously.
- Have students compare how combinations of concentration and additives affect spread, adhesion, and coating uniformity.
- Have students measure and record mass, spread diameter, percent coverage, and drips/smears.
- Have them present their optimized design and reasoning, explaining trade-offs or limitations.
- Encourage students to graph their results (bar graphs, scatter plots) and analyze trends or correlations between variables.
Activity Scaling
To modify for lower-level students:
- Skip detailed biology or vaping discussions; just introduce that surfactants help lungs stay open.
- Have students test fewer variables.
- Use pre-measured candy melts and syrup baths to reduce spills.
- Prepare corn syrup baths at 80–90% for best support, and have students only test different additive concentrations.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students explore the concept of surface tension and how additives such as surfactants can alter it. Students investigate how substances like surfactants change the surface tension of water and relate this to lung function. Students will then connect their observations to real-world concerns, examini...

Students learn about the parts of the human respiratory system and the gas exchange process that occurs in the lungs. They also learn about the changes in the respiratory system that occur during spaceflight, such as decreased lung capacity.

Students learn about the basics of molecules and how they interact with each other. They learn about the idea of polar and non-polar molecules and how they act with other fluids and surfaces. Students acquire a conceptual understanding of surfactant molecules and how they work on a molecular level. ...

Student teams are challenged to evaluate the design of several liquid soaps to answer the question, “Which soap is the best?” Through two simple teacher class demonstrations and the activity investigation, students learn about surface tension and how it is measured, the properties of surfactants (so...
References
[1] Jamal A, Park-Lee E, Birdsey J, et al. Tobacco Product Use Among Middle and High School Students — National Youth Tobacco Survey, United States, 2024. MMWR Morb Mortal Wkly Rep 2024;73:917–924. DOI: http://dx.doi.org/10.15585/mmwr.mm7341a2
Copyright
© 2026 by Regents of the University of Colorado; original © 2025 University of KansasContributors
Cara Snyder, Nicholas Anderson, Soraiya Coffman, Dr. Prajna Dhar, Meagan PattersonSupporting Program
Inquiry-Driven Engineering Activities using Bioengineering (IDEA-BioE), University of KansasAcknowledgements
This material is based upon work supported by the National Science Foundation under grant no. ECC-2055716 - a Research Experience for Teachers program titled Inquiry-Driven Engineering Activities using Bioengineering (IDEA-BioE) at the University of Kansas. Any opinions, findings and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation.
Last modified: March 25, 2026







User Comments & Tips