Quick Look
Grade Level: 4 (3-5)
Time Required: 2 hours 45 minutes
(five 35-minute sessions)
Expendable Cost/Group: US $0.00
Group Size: 3
Activity Dependency: None
Subject Areas: Biology, Chemistry, Measurement, Problem Solving
NGSS Performance Expectations:

| 3-5-ETS1-1 |
| 3-5-ETS1-3 |
| 4-LS1-1 |

Summary
Students use gummy bears and their own gummy prototype to simulate a cell membrane as they learn how osmosis works in a cell and how water moves down the concentration gradient. As they learn more about cell membrane function, they better understand the concepts of cell membranes, osmosis, and concentration gradient.Engineering Connection
Biomedical engineers design tools and systems to model how the human body works so they can study it in a safe and controlled way. By learning how different parts of the body interact, they can create new ways to deliver medicine, prevent and treat diseases, and build helpful devices like artificial joints and organs. They also improve technologies such as MRI machines and develop tools for robotic surgery. All of this work helps doctors take better care of people and improves our quality of life.
Learning Objectives
After this activity, students should be able to:
- Describe how osmosis is the movement of water across a semipermeable membrane from an area of high-water concentration to an area of low-water concentration (gradient).
- Discuss the effects that different solutions (water, sugar, salt) can have on the gummy bear and prototype due to their varying concentrations and properties.
- Record the changes in gummy bear and prototype length and weight, analyze the data, and explain their observations in terms of osmosis.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
3-5-ETS1-1. Define a simple design problem reflecting a need or a want that includes specified criteria for success and constraints on materials, time, or cost. (Grades 3 - 5) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Define a simple design problem that can be solved through the development of an object, tool, process, or system and includes several criteria for success and constraints on materials, time, or cost. Alignment agreement: | Possible solutions to a problem are limited by available materials and resources (constraints). The success of a designed solution is determined by considering the desired features of a solution (criteria). Different proposals for solutions can be compared on the basis of how well each one meets the specified criteria for success or how well each takes the constraints into account. Alignment agreement: | People's needs and wants change over time, as do their demands for new and improved technologies. Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
3-5-ETS1-3. Plan and carry out fair tests in which variables are controlled and failure points are considered to identify aspects of a model or prototype that can be improved. (Grades 3 - 5) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Plan and conduct an investigation collaboratively to produce data to serve as the basis for evidence, using fair tests in which variables are controlled and the number of trials considered. Alignment agreement: | Tests are often designed to identify failure points or difficulties, which suggest the elements of the design that need to be improved. Alignment agreement: Different solutions need to be tested in order to determine which of them best solves the problem, given the criteria and the constraints.Alignment agreement: | |
| NGSS Performance Expectation | ||
|---|---|---|
|
4-LS1-1. Construct an argument that plants and animals have internal and external structures that function to support survival, growth, behavior, and reproduction. (Grade 4) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Construct an argument with evidence, data, and/or a model. Alignment agreement: | Plants and animals have both internal and external structures that serve various functions in growth, survival, behavior, and reproduction. Alignment agreement: | A system can be described in terms of its components and their interactions. Alignment agreement: |
Common Core State Standards - Math
-
Make a line plot to display a data set of measurements in fractions of a unit (1/2, 1/4, 1/8). Solve problems involving addition and subtraction of fractions by using information presented in line plots.
(Grade
4)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Evaluate designs based on criteria, constraints, and standards.
(Grades
3 -
5)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- 9 clear plastic 9 oz cups
- 1 permanent marker
- 1 ruler (cm units)
- 50 mL each of water, salt water solution, and sugar water solution (see below)
- 1 graduated cylinder or measuring cup (to measure 50 mL accurately, if not pre-poured)
- 4 gummy bears (select same color in a group)
- 1 gram scale
- 3-5 plastic spoons (to remove gummies from cups)
- 3 teacher-made gummy bears (one salt water, one sugar water, and one water)
- Gummy Bear Research Worksheet (1 per student)
- Gummy Prototype Investigation Worksheet (1 per student)
- Control and Prototype Data Analysis Worksheet (1 per student)
- paper towels (for spills and cleanup)
For the entire class to share:
- 1 laptop or tablet with access to the internet and projector (for YouTube videos and the What? How? Presentation)
- 3 1 L pitchers with:
- 1 L water1 L salt solution (see below)
- 1 L sugar solution (see below)
For the teacher:
To make salt and sugar solutions:
- access to the Solution and Gummy Bear Recipes
- 1 hotplate
- 1 stirring rod or spoon
- 2,000 mL distilled or tap water
- 35 g table salt (NaCl)
- 250 g sugar
- 2 1,000 mL beakers or heat-safe containers
- safety goggles and heat-resistant gloves
To make the gummy prototypes:
- access to the Solution and Gummy Bear Recipes
- 3 silicone molds such as mini ice cube trays, pop it fidgets, or gummy bear molds with at least 30 cavities each (use same size for all gummy prototypes to reduce variability)
- 9 oz total, 3 oz each of 3 different flavored gelatin mixes (1 flavor/color per batch
- 1.5 oz total, 0.25 oz each packet, of unflavored powder (6 envelopes at 0.25 oz each; use 1.5 envelopes per batch)
- 1/3 cup each of water, salt solution, and sugar solution
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/uot-3040-cell-membranes-osmosis-gummy-bears] to print or download.Introduction/Motivation
Let’s start at the beginning, with the basic building block of all living things: the cell. A cell is the smallest unit of life. It is like a tiny building block that makes up all living things. Every living organism, including plants, animals, and bacteria, is made of one or more cells. Cells do important jobs to keep living things alive, like getting energy, growing, and making more cells.
(Show What? How? Presentation Slide 1.)
I present to you a stunning image of human cells! What do you observe about these cells? Do you notice any structures (parts) inside or on them? (Let students offer observations.)
Now, let’s look at some other images of cells in action. As you look, think: What is happening? And how did this happen to it? (Show Slides 2-7 in the What? How? Presentation, pausing for think time for each slide.)
Cells have a special outer layer called a cell membrane that helps them carry out important functions. One of these important functions is osmosis.
Let’s see this in action, using the help of something you might have seen before: an onion. (Show Red Onion Osmosis - Plasmolysis - Microscope YouTube video, 2:54 minutes). After watching the video, how did your understanding of cells and their parts (structures) change? What do you understand about osmosis? (Let students offer their thoughts.)
Scientists use a word called “osmolarity” to describe how much “stuff,” like salt or sugar, is dissolved in water.
Water moves toward the side that has more “stuff.” So, if there is more “stuff” outside the cell, water moves out. If there is less “stuff” outside, water moves in.
Osmosis is when water moves into or out of a cell through the cell membrane to keep things balanced, meaning not too full and not too empty. This movement depends on osmolarity: how much “stuff” is dissolved inside and outside the cell. In the video, when salt water was added, water moved out of the cells, causing them to shrink. When fresh water was added, water moved back into the cells, and they expanded again. This shows how cells try to stay balanced. Let’s learn even more about cells and osmosis by watching this video. (Show What is Osmosis? YouTube video, 2:01 minutes.)
Now let’s think about what we’ve learned: Why do cells need cell membranes? (Let students offer answers. Answer: Cells need a membrane because it protects the cell and controls what goes in and out, like nutrients and waste.)
How is the function of osmosis important in a cell? (Let students offer answers. Answer: Osmosis helps move water in and out of the cell, so it doesn’t get too full or too empty. This helps the cell stay healthy and do its job properly.)
Now let’s do a quick poll. Can we use an object to simulate a cell conducting osmosis? (Let students raise their hands for yes and no. Tally their responses on chart paper.)
Biomedical engineers study cells and design ways to help our bodies work better. They create things like medicines and medical tools that help treat diseases and keep us healthy.
This week, we are going to be biomedical engineers and learn more about how osmosis works in cell membranes. We are also going to develop our own cell membrane prototypes that simulate a real cell. Do you think you can engineer this? Let’s get started!
Procedure
Background
Osmosis is the movement of water across a barrier (like a cell membrane) from an area where there is more water to an area where there is less water. It doesn’t require energy, and it helps cells stay balanced. If too much water enters a cell, it can swell; if too much water leaves, it can shrink. This balance is important so cells can function properly.
Osmolarity is a way to describe how much dissolved stuff (like salt or sugar) is in a solution. A solution with high osmolarity has lots of dissolved particles, while one with low osmolarity has fewer. Osmolarity helps determine where water will move during osmosis. Note: Water will move toward the side with higher osmolarity. This is why cells may shrink in a salty (hypertonic) environment or swell in a low-salt (hypotonic) environment.
In this activity, gummy bears and gelatin are used as models of cells because they can absorb and release water, just like real cells respond to osmosis. Gelatin is made of long polymer chains that form a flexible structure with tiny spaces that can trap water. When placed in different solutions, gummy bears change size as water moves in or out, helping students see how osmosis and osmolarity work in a real, visible way. Although they are not true cells, these models help us understand how real cells react to different environments.
Before the Activity
- Gather group materials.
- Make one copy per student of each of the following documents:
- Gummy Bear Research Worksheet
- Gummy Prototype Investigation Worksheet
- Control and Prototype Data Analysis Worksheet
- Create the salt and sugar solutions with 1,000 mL water to 35 g (approximately 2 Tbs) salt and 1,000 mL water to 250 g (approximately 1 cup) sugar, respectively). See Solution and Gummy Bears Recipes.
- Prepare three different types of gummy prototypes prior to Day 3: water, salt, and sugar. Follow the procedure in Solution and Gummy Bears Recipes. Note: Each group gets one of each gummy prototype.

During the Activity
Day 1: Introduction, Ask and Research (35 minutes)
- Introduce the activity by reading through the Introduction and Motivation section, which includes going through the What? How? Presentation and watching two YouTube videos.
- Optional: Review the engineering design process.
- Define the engineering problem: How can we design a gummy model of a cell that controls water movement so the cell stays balanced in different liquids?
- Divide the students into groups of 4.
- Distribute one Gummy Bear Research Worksheet to each student.
- Explain that students will explore osmosis and how gelatin behaves in different solutions to gather evidence for designing their own cell model later. They will observe what happens to gummy bears in water, sugar water, and salt water.
- Have students hypothesize what they think will happen to the gummy bear in each solution. (Optional sentence frame: “I think the gummy bear in ___ will ___ because ___.”)
- Distribute materials to each group: 3 9 oz clear plastic cups, 50 mL of water, salt water solution, and sugar water solution, 4 gummy bears (3 for testing and one as a control), 1 ruler, 1 permanent marker, and 1 scale.
- Verbally explain the procedure to the class. Each group will:
- Label their plastic cups water, salt, and sugar, respectively.
- Add 50 mL of the corresponding solution into each cup.
- Add one gummy bear to each cup.
- Measure the length (in cm) and weight (in g) of the control gummy bear (this gummy bear WILL NOT be placed in any solution) and record these values as the “Before” measurements in the Gummy Bear Research Data table.
- Allow all three cups to remain undisturbed for 24 hours.

- Give groups time to complete the procedure.
- Have students complete the first side of the Gummy Bear Research Worksheet.
Day 2: Research and Imagine (35 minutes)
- Have each group obtain their three gummy bear solutions from the previous day.
- Remind students that they now need to observe their gummy bears after 24 hours to see how they changed (i.e., how much they swelled and/or shrank).
- Have students carefully remove each gummy bear from its solution using a clean plastic spoon.
- Instruct students to measure the length and weight of each gummy bear and record those values in on the Gummy Bear Research Data table of their Gummy Bear Research Worksheet.


- Ask each team to discuss their results and make conclusions about the gummy bears and osmolarity based on the data they gathered on the length and weight of their gummy bears (e.g., which solution best maintained osmolarity).
- Optional: Pose the following questions if students need guidance:
- Which solution kept the gummy bear closest to its original size?
- What does that tell you about osmolarity?
- Instruct students to write their conclusions in the indicated section of the Gummy Bear Research Worksheet.
- Facilitate a brief class discussion to compare results. Guide students to recognize that the salt solution caused the least change for most groups, meaning it maintained balance (osmolarity). This means water did not move in or out as much, so the inside and outside were more balanced.
- Optional: Check that each student has completed their Gummy Bear Research Worksheet.
- Hand out a Gummy Prototype Investigation Worksheet to each student.
- Remind students of the challenge: How can we design a gummy model of a cell that helps control water movement so the cell stays balanced in different liquids?
- Explain that each team should use their findings from the gummy bear research to brainstorm and plan their own gummy model based on evidence from their data. Teams should consider:
- Which type of gummy (water-based, sugar-based, or salt-based).
- Which solution (water, sugar water, or salt water).
- Explain to the students that plain and flavored gelatin are provided as the solute base of their “recipe” and that they will be able to select water, sugar solution, and salt solution as the liquid in which to dissolve the chosen solute.
- Give each group time to brainstorm and plan their gummies and the solutions they will use to test their model.
Day 3: Plan and Create (35 minutes)
- Encourage each group to use their brainstormed ideas from the previous day to create a finalized plan for the gummies and solutions they will use to test their model.
- Prompt students to complete the following: “We will test a ___ gummy in ___ solution because ___.”
- Optional: Have each group briefly share their plan with you for approval (circulate to sign off).
- Remind students to conduct the same procedure as done with the initial gummy bear research. Each group will:
- Label their plastic cups appropriately.
- Add 50 mL of the chosen solution(s) into each cup.
- Add the appropriate gummy bear to each cup.
- Measure the length (in cm) and weight (in g) of the control gummy bear (this gummy bear WILL NOT be placed in any solution) and record these values.
- Allow all three cups to remain undisturbed for 24 hours.
Day 4: Test and Analyze (35 minutes)
- Have each group obtain their gummy bears and solutions from the previous day.
- Instruct students to measure the length and weight of each gummy bear and record those values in the Gummy Prototype Data table of their Gummy Prototype Investigation Worksheet.
- Distribute one Control and Prototype Data Analysis Worksheet to each student.
- Instruct teams to organize and analyze their data by:
- Comparing gummy measurements to the control.
- Identifying which gummy changed the most and least.
- Have students create a simple data representation (e.g., bar graphs showing gummy length and mass compared to the control).
- Have groups compare their data analysis with another group and discuss similarities and differences.
- Ensure all students/groups have time to finish the worksheet.
- Based on their data, guide students to write conclusions in the Control and Prototype Data Analysis Worksheet. Students should think about what worked in their model, what didn’t work, and what they would change in their model to make it better.
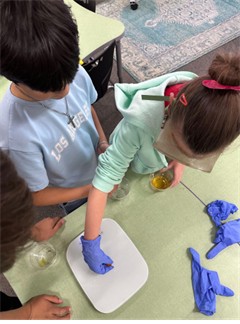
- Optional: If time and resources allow, give teams time to improve and retest their model.
Day 5: Communicate Results (35 minutes)
- Optional: If groups improved their model the previous day, give them time to measure the length and weight of each of their improved gummy bears.
- Have each group present their findings (i.e., which solute and solution combination were used and what data they measured, mass and length).
- Facilitate a class discussion to help students relate their results to the key concepts/terms:
- Osmosis is the movement of water into or out of the gummy prototype. We can see this by changes in mass and size (length).
- Osmolarity describes how much dissolved “stuff” (like salt or sugar) is in a solution. When a gummy shows little or no change, it means the inside and outside environments were more balanced.
- The next two concepts are at a metacognitive level:
- Cells in plants and animals—for example, grapes drying and shrinking into raisins due to water leaving the cells.
- Biomedical engineers study how cells respond to different environments, such as high-salt conditions in the body, to help design medical treatments and technologies.
Vocabulary/Definitions
balanced osmolarity: Balanced concentration between solution and molecule.
cell: The basic unit of a living organism.
cell membrane: The semipermeable membrane that surrounds a cell.
gradient: The rate a physical quantity increases or decreases.
osmosis: The movement of water from a low solute concentration to a high.
semipermeable: Allows some things in but not others.
solute: Able to be dissolved.
Assessment
Pre-Activity Assessment
Brainstorming: Define a cell for the class. In small groups, have students engage in open discussion about ordinary things that exist that are like a cell. As a class, record their ideas on chart paper.
Quick Poll: Define osmosis for the class. Before the activity begins, ask the class a question and tally their responses on chart paper. Ask: Can we use an object to simulate a cell conducting osmosis?
Activity Embedded (Formative) Assessment
Research Worksheet: Have students work individually or in groups on the Gummy Bear Research Worksheet. After they finish, have them compare answers with their group, giving all students time to finish the worksheet.
Hypothesize: Ask each group what would happen if we placed a gummy with a different composition in one of the solutions? (Answer: This is dependent on what the gummy composition is and what type of solution it is being placed in. The result will be that water either moves out or in to balance the concentration of sugar or salt. The gummy will also either increase or decrease in volume and mass.)
Post-Activity (Summative) Assessment
Investigation Worksheet: Have students work individually or in groups on the Gummy Prototype Investigation Worksheet. After they finish, have them compare answers with their own or another group, giving all students time to finish the worksheet.
Data Analysis Worksheet: Have students work individually or in groups on the Control and Prototype Data Analysis Worksheet. After they finish, have them compare answers with their own or another group, giving all students time to finish the worksheet.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn about the different structures that comprise cell membranes, fulfilling part of the Research and Revise stages of the legacy cycle. They view online animations of cell membrane dynamics (links provided).

Students learn that engineers develop different polymers to serve various functions and are introduced to selectively permeable membranes. In the main activity, student pairs test and compare the selective permeability of everyday polymer materials engineered for food storage (including plastic groc...

Students explore the structure and function of cell membranes. As they study the ingress and egress of particles through membranes, students learn about quantum dots and biotechnology through the concept of intracellular engineering.
Copyright
© 2026 by Regents of the University of Colorado; original © 2025 University of Texas at AustinContributors
Monica WarrSupporting Program
Research Experience for Teachers (RET), The Material Science and Engineering Department at the University of Texas, AustinAcknowledgements
This curriculum was developed under National Science Foundation through the Center for Dynamics and Control of Materials: an NSF MRSEC under Cooperative Agreement number DMR-1720595. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation.
Last modified: May 8, 2026





User Comments & Tips