Quick Look
Grade Level: 9 (9-12)
Time Required: 1 hours 15 minutes
Expendable Cost/Group: US $5.00
Group Size: 2
Activity Dependency: None
Subject Areas: Chemistry
NGSS Performance Expectations:

| HS-PS1-2 |

Summary
This lab exercise exposes students to a potentially new alternative energy source—hydrogen gas. Student teams are given a hydrogen generator and an oxygen generator. They balance the chemical equation for the combustion of hydrogen gas in the presence of oxygen. Then they analyze what the equation really means. Two hypotheses are given, based on what one might predict upon analyzing the chemical equation. Once students have thought about the process, they are walked through the experiment and shown how to collect the gas in different ratios. By trial and error, students determine the ideal combustion ratio. For both volume of explosion and kick generated by explosion, they qualitatively record results on a 0-4 scale. Then, students evaluate their collected results to see if the hypotheses were correct and how their results match the theoretical equation. Students learn that while hydrogen will most commonly be used for fuel cells (no combustion situation), it has been used in rocket engines (for which a tremendous combustion occurs).Engineering Connection
Many engineered systems have chemical components that react to create new products, release energy or change the state of materials. Engineers who design combustion engines, fuel cells, batteries, medicines, cosmetics, processed foods and many other products, calculate and control chemical reactions as part of their work. As part of their design efforts, chemical engineers, materials engineers, environmental engineers, food engineers, and others, perform chemical reaction calculations and test their correctness in the lab.
Oxygen and hydrogen combine to produce water as well as release energy. Engineers apply this energy-releasing reaction concept to solve real-life problems, such as hydrogen-fueled rockets and fuel cells that generate electrical power for vehicles and homes. Unlike traditional hydrocarbon fuels, hydrogen and oxygen do not produce any polluting byproducts, making it an alternative renewable source of energy. Many engineers are developing technologies to efficiently exploit the potential of hydrogen energy.
Learning Objectives
After this activity, students should be able to:
- Balance chemical equations.
- Safely follow instructions and correctly use laboratory equipment.
- Use qualitative data to explain the validity of a given hypothesis.
- Explain relationships between the tested variables and observed results.
- Explain the importance of following strict laboratory procedures to obtain desired results and keep everyone safe.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-PS1-2. Construct and revise an explanation for the outcome of a simple chemical reaction based on the outermost electron states of atoms, trends in the periodic table, and knowledge of the patterns of chemical properties. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Construct and revise an explanation based on valid and reliable evidence obtained from a variety of sources (including students' own investigations, models, theories, simulations, peer review) and the assumption that theories and laws that describe the natural world operate today as they did in the past and will continue to do so in the future. Alignment agreement: | The periodic table orders elements horizontally by the number of protons in the atom's nucleus and places those with similar chemical properties in columns. The repeating patterns of this table reflect patterns of outer electron states. Alignment agreement: The fact that atoms are conserved, together with knowledge of the chemical properties of the elements involved, can be used to describe and predict chemical reactions.Alignment agreement: | Different patterns may be observed at each of the scales at which a system is studied and can provide evidence for causality in explanations of phenomena. Alignment agreement: |
Common Core State Standards - Math
-
Reason abstractly and quantitatively.
(Grades
K -
12)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Students will develop an understanding of the relationships among technologies and the connections between technology and other fields of study.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Connect technological progress to the advancement of other areas of knowledge and vice versa.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
State Standards
Washington - Math
-
Reason abstractly and quantitatively.
(Grades
K -
12)
More Details
Do you agree with this alignment?
Washington - Science
-
Construct and revise an explanation for the outcome of a simple chemical reaction based on the outermost electron states of atoms, trends in the periodic table, and knowledge of the patterns of chemical properties.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- hydrogen generator, refer to the Hydrogen Generator - Teacher Preparation Instructions for required materials (gas generator kit, chemicals, lab equipment, etc.)
- oxygen generator, refer to the Oxygen Generator - Teacher Preparation Instructions for required materials (gas generator kit, chemicals, lab equipment, etc.)
NOTE: alternatively, prepare a hydrogen bag generator and an oxygen bag generator for the entire class to use; refer to the Gas Bags - Teacher Preparation Instructions for required materials
- 2 pipette bulbs (a plastic pipette with an approximately 15 ml bulb; the plastic stem must be cut and the hole in the bulb must be large enough to fit over the gas outlet tube in the generators)
- 2 x 250 ml beakers filled with water, to hold generators and maintain constant temperature
- book of matches
- short candle
- safety glasses/goggles, pair per person
- chemical-resistant gloves, pair per person
- Microscale Experiment with Hydrogen & Oxygen Combustion Worksheet, one per person
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/wsu_oxygen_hydrogen_activity1] to print or download.Pre-Req Knowledge
Students should be familiar with:
- Balancing chemical equations.
- Measuring and weighing solids and liquids.
- Qualitative and quantitative data.
Introduction/Motivation
Combine two parts hydrogen with one part oxygen and what do you get? (See if students know.) You get water and an energy release (in the form of a loud POW!). Engineers apply this energy-releasing concept to solve real-life problems. Unlike traditional hydrocarbon fuels, hydrogen and oxygen do not produce any polluting byproducts, making it a great alternative renewable source of energy for vehicles and other systems that require energy (fuel or electrical) to operate. The energy-releasing reaction is similar to that used in hydrogen-fueled rockets and fuel cells.
Hydrogen is one of two natural elements that combine to make water. Hydrogen is not an energy source, but an energy carrier because it takes a great deal of energy to extract it from water. Many engineers are developing technologies that can efficiently exploit the potential of this energy-releasing reaction. Hydrogen energy is useful as a compact energy source in fuel cells and batteries.
Have you heard about fuel cells in the news? According to many reports, we may soon be using this new energy-saving technology to generate electrical power for our homes and cars. The technology is extremely interesting to people in all walks of life because it offers a means of making power more efficiently and with less pollution. Let's learn more about how it works. (Proceed to deliver suitable content information to students from the background information provided below.)
Hydrogen-Fueled Rockets
When hydrogen is mixed with oxygen (typically in the form of air), it can burn with a spark ignition. Since hydrogen is a huge storage of energy when it burns (and the chemical bonds are broken) it produces exothermic energy in the form of heat and thrust energy. This thrust energy can be transferred to a rocket to propel it upwards.
Rockets are usually powered by a chemical reaction or explosion within the rocket itself. The first types of rockets were fireworks. Rockets can be powered by different types of fuel. Early Chinese rockets used gunpowder, and later on people used gasoline and other petroleum products to fuel rockets. Rocket fuels often were kept frozen to avoid explosions. Some rockets combine hydrogen and oxygen to create an intense chemical reaction. Some, such as the Russian N1, use kerosene. The Saturn V had three stages and used petroleum fuel in stage 1 and hydrogen fuel in states 2 and 3.
The most powerful rocket ever built was the Saturn V, which was used to deliver a human to the moon. But the most powerful rocket engine ever built is the space shuttle's rocket booster. (The Saturn V was more powerful because it used five engines instead of the two used by the shuttle.) The shuttle boosters are fueled by a solid fuel mixture that is mainly aluminum and ammonium perchlorate.

When most people think about motors or engines, they think of gasoline engines in cars, which produces rotational energy to drive the wheels. Electric motors produce rotational energy to drive fans or spin disks. A steam engine, steam turbine and most gas turbines are used to do the same things.
Rocket engines are fundamentally different. Rocket engines are reaction engines. The basic principle driving a rocket engine is the physical law that "to every action there is an equal and opposite reaction." A rocket engine "throws mass" in one direction and benefits from the reaction that occurs in the other direction. What are some everyday examples of "throwing mass"?
- If you have ever seen a big shotgun fired in the movies or on TV, then you might have noticed the "kick" or "recoil" from the gun being fired. When a person shoots the gun, an exploding force "kicks" the shooter's shoulder back with a great deal of energy. That kick is a reaction. A shotgun shoots about an ounce of metal in one direction at about 700 miles per hour, and the firing person's shoulder gets hit with the reaction. If the person firing the gun was wearing roller skates or standing on a skateboard when the gun was fired, then the gun would seem like a rocket engine because the person would roll in the opposite direction (the reaction).
- Have you ever blown up a balloon and then just let it go? The balloon flies all around the room before running out of air. By doing this, you have created a rocket engine. In this case, what is being thrown is the air molecules inside the balloon. Many people mistakenly believe that air molecules do not weigh anything, but they do. When you "throw them" out the balloon opening, the rest of the balloon reacts by moving in the opposite direction.
- When a big fire hose sprays water, it takes a lot of strength to hold the hose. Sometimes you see two or three firefighters working together to keep the hose under control. The hose is acting like a rocket engine. As the hose "throws water" in one direction, the firefighters use their strength and weight to counteract the reaction. If they let go, the hose thrashes around with tremendous force. If the firefighters were all standing on skateboards, the hose would propel them backwards with great force.
Hydrogen Fuel Cells
Hydrogen is a versatile energy carrier that can be used to power nearly every energy need. A fuel cell is an electrochemical device that converts hydrogen and oxygen into water, producing electricity and heat in the process. It operates at a high level of efficiency (two to three times more efficient than traditional combustion systems) with little noise or air pollution. In principle, a fuel cell operates like a battery. Unlike a battery, a fuel cell does not run down or require recharging. It produces energy in the form of electricity and heat as long as fuel is supplied. Fuel cells can power almost any application that typically uses batteries and also power our modes of transportation, such as personal vehicles, trucks, buses and water vehicles. Hydrogen fuel cells have the potential to play an important role in the future by replacing the use of imported oil and gasoline currently used.
As an example, a conventional power plant typically generates electricity at efficiencies of 33-35%, while fuel cell systems can generate electricity at efficiencies up to 60%. The gasoline engine in a typical car is less than 20% efficient in converting the chemical energy in gasoline into power that moves the vehicle. Hydrogen fuel cell vehicles, which use electric motors, are much more energy efficient and use 40-60% of the fuel's energy. This corresponds to more than a 50% reduction in fuel consumption compared to a typical vehicle with a gasoline ICE (internal combustion engine).
Fuel Cell Design
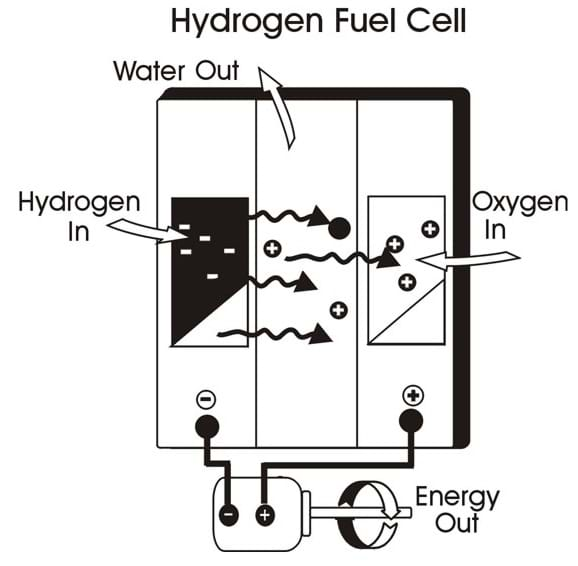
A single fuel cell consists of an electrolyte (a substance containing free ions that make it electrically conductive) sandwiched between two electrodes, an anode and a cathode. Bipolar plates on either side of the cell help distribute gases and serve as current collectors.
Polymer electrolyte membrane (PEM): In a PEM fuel cell, a type of fuel cell, hydrogen gas flows through channels to the anode, where a catalyst causes the hydrogen molecules to separate into protons and electrons. The membrane allows only the protons to pass through it. While the protons are conducted through the membrane to the other side of the cell, the stream of negatively-charged electrons follows an external circuit to the cathode. This flow of electrons is electricity that can be used power a motor. On the other side of the cell, oxygen gas (typically supplied from air that contains approximately 79% N2 and 21% O2), flows through channels to the cathode. The electrons react with oxygen and the hydrogen protons (which have moved through the membrane) at the cathode to form water. This results in an exothermic reaction that generates heat that can be used outside the fuel cell.
Everyday Fuel Cells
Fuel cells have played a role in the space program by supplying electricity to several missions. Fuel cell energy is anticipated to replace traditional power sources in the coming years—from micro fuel cells in cell phones to high-powered fuel cells for stock car racing. Engineers at the major automakers are working to commercialize fuel cell cars. Many uses for fuel cells currently exist; they are powering buses, boats, trains, planes, scooters and even bicycles. We are using fuel cell-powered vending machines, vacuum cleaners and highway road signs. Miniature fuel cells for cellular phones, laptop computers and portable electronics are on their way to market—and the possibilities are endless.
Environmental and Health Impacts
About 25% of all human-generated greenhouse gases are released from powering our many modes of transportation, with more than half of that from light-duty vehicles. Because fuel cells significantly reduce greenhouse gas and other emissions, it is an important technology in our efforts to reduce pollution, which contributes to global warming, smog and other pollution consequences. When hydrogen is carried onboard a fuel-cell powered vehicle, zero emissions result. The use of fuel cells reduces local air and noise pollution and groundwater contamination, and improves public health and safety by reducing citizens' exposure to fuel and emissions dangers. Fuel cells could dramatically reduce urban air pollution, decrease oil imports, reduce the trade deficit and produce jobs because running on hydrogen derived from a renewable source emits nothing but water vapor.
Procedure
Before the Activity
- Gather materials and make copies of the Microscale Experiment with Hydrogen & Oxygen Combustion Worksheet.
- Follow the instructions to build the hydrogen and oxygen generators (or build the hydrogen and oxygen bags).
- Modify the pipette bulbs by cutting the hole in the plastic stem; the hole in the bulb must be large enough to fit over the gas outlet tube in the generator.
- As necessary, conduct the experiment to gain demonstration experience.
With the Students
- Present the Introduction/Motivation content. Briefly talk about developments in hydrogen gas production and its use as rocket fuel and for fuel cells.
- Describe how combustion works and the importance of oxygen.
- Ask students: Do you think that gasoline and methane can burn without oxygen? Looking at the general chemical reaction for combustion, you need fuel +oxidizer +spark in order for combustion to occur.
- Describe the principle behind "throwing mass" to propel an object. This is how a rocket works.
- Describe how hydrogen can be used for energy storage (the principle behind the enormous energy source potential of fuel cells).
- Show students an unbalanced chemical equation (refer to the worksheet and Figure 1). Describe how matter cannot be created or destroyed, and hint that the law of conservation of energy gives them a clue how to balance the chemical equation.
- Have students work together for a few minutes to try to balance the equations.
- Walk through the balancing procedure; if possible, have students do most of the explaining.
![metal + nonmetal > salt (such as 2NA + Cl2 > 2NACl), metal + oxygen (nonmetal) > metal oxide (salt) (such as 4FE + 3O2 > 2Fe2O3); metal oxide + water > base (such as Na2O + H2O > 2NaOH); nonmetal compound + water > acid (SO3 + H2O > H2SO4); acid + base > salt + water (neutralization) (such as 2HCl + Mg(OH)2 > MgCl2 + 2H2O); metal (1) + salt > metal (2) + salt (such as Zn + CuSO4 > Cu + ZnSO4); organic compound + oxygen > carbon dioxide + water (combustion) (such as CH4 + 2O2 > CO2 + 2H2O); monomer (organic compound) + monomer > polymer (polymerization) (such as C2H3Cl [vinyl chloride] + C2H3Cl > -[Ch2CHCl]- (polyvinyl chloride PVC) metal + nonmetal > salt (such as 2NA + Cl2 > 2NACl), metal + oxygen (nonmetal) > metal oxide (salt) (such as 4FE + 3O2 > 2Fe2O3); metal oxide + water > base (such as Na2O + H2O > 2NaOH); nonmetal compound + water > acid (SO3 + H2O > H2SO4); acid + base > salt + water (neutralization) (such as 2HCl + Mg(OH)2 > MgCl2 + 2H2O); metal (1) + salt > metal (2) + salt (such as Zn + CuSO4 > Cu + ZnSO4); organic compound + oxygen > carbon dioxide + water (combustion) (such as CH4 + 2O2 > CO2 + 2H2O); monomer (organic compound) + monomer > polymer (polymerization) (such as C2H3Cl [vinyl chloride] + C2H3Cl > -[Ch2CHCl]- (polyvinyl chloride PVC)](/content/wsu_/activities/wsu_oxygen_hydrogen_activity1/equations.jpg)
- Introduce the concept of qualitative data, which is data not described by specific numbers, but rather by approximate quantities or relationships.
- Divide the class into groups of two students each and have them move to designated stations with the materials needed. Direct them to put on safety equipment and follow the worksheet instructions carefully to ensure safe practices. Put on your own safety lab gear.
- As a class, walk through the worksheet procedures while performing a demonstration.
- Put fresh hydrochloric acid and hydrogen peroxide solutions in group generators.
- As you walk around filling generator stations, ask questions about the results students are getting.
- Expect students to hear explosions as part of the experiment. If no explosion, ask if it was an ineffective combination or a procedural error.
- Have students compare their results with other groups to validate their own.
- Once groups are finished, have them sit down with their results and answer the worksheet post-lab questions about their data. Make sure they make the connections between the theoretical equation and their results by pointing out how the 2:1 ratio of gas in the equation related to the 2:1 gas mixture they just created.
Vocabulary/Definitions
anode: The terminal on a device where current flows in from outside.
balanced chemical reaction: A chemical equation that balances the atoms and molecules on one side with the atoms and molecules on the other side. The reactants on the left side are equal to the products on the right side.
cathode: The terminal on a device where current flows out.
chemical reaction: A process that involves rearrangement of the molecular or ionic structure of a substance, as opposed to a change in physical form or a nuclear reaction.
combustion: A chemical change; the burning of a fuel and oxidant to produce heat and/or work.
electron: A stable subatomic particle with a charge of negative electricity, found in all atoms and acting as the primary carrier of electricity in solids.
kick or recoil: A backward motion resulting in a sudden jerk or jump.
membrane: A pliable sheet-like structure acting as a boundary, lining or partition.
PEM: Acronym for polymer electrolyte membrane. A fuel cell that incorporates a solid polymer membrane used as its electrolyte.
proton: A stable subatomic particle occurring in all atomic nuclei, with a positive electric charge equal in magnitude to that of an electron.
qualitative data: Data that approximates or characterizes, but does not measure the attributes, characteristics, properties, etc., of a thing or phenomenon. Qualitative data describes, whereas quantitative data defines.
quantitative data: Data that can be quantified and verified, and is amenable to statistical manipulation. Quantitative data defines whereas qualitative data describes.
ratio: An expression of the magnitude of one quantity relative to another quantity.
Assessment
Activity Embedded Assessment
Discussions: During the activity, walk around and help students troubleshoot the lab. Ask them questions about the reaction and the results they are getting. Have students from each lab group explain to you the process and results. Make sure all students are participating as equally as possible. Additionally, discuss applications of this type of energy to helping solve real-world problems from an engineering point-of-view.
Post-Activity Assessment
Worksheets: Collect and grade the worksheets. Review their answers to gauge their comprehension.
Concluding Discussion: As a class, share and discuss results and conclusions. Ask the following questions:
- Discuss the post-lab questions as a group. Address common misunderstandings that you observe from grading the worksheets.
- Is a car that runs off of a fuel cell completely emissions free? Why or why not? (students should realize that energy must be invested to generate the hydrogen fuel).
- Now that you have seen the mathematics involved in an explosion, how would you describe it to someone who did not understand how explosions work?
Safety Issues
- Require students to wear safety glasses (or goggles) and chemical resistant gloves during this experiment. Have everyone involved, including the instructor and observers, wear safety glasses.
- Take care when handling open flames.
Troubleshooting Tips
The specified dilutions of hydrochloric acid and hydrogen peroxide solutions used in the gas generators lose their effectiveness in about five minutes, so prepare and give groups fresh solutions right before they are needed. More concentrated solutions may be too dangerous to use at the high school level.
Activity Extensions
In this activity, students balanced chemical equations based on conservation of energy. Now, have students discuss why the formation of H2O makes sense from knowing the outermost electron states of hydrogen and oxygen.
- How many electrons does oxygen need to complete its octet? What can hydrogen do to have a full outer shell of electrons?
- Have students draw an electron diagram for water.
- What are these shared bonds called? (Covalent bonds).
- Given the locations of oxygen and hydrogen on the periodic table, how reactive would we expect these elements to be? Does this make sense given your combustion observations?
Additional Multimedia Support
See a good fuel cell diagram at http://www.blogcdn.com/www.bloggingstocks.com/media/2007/02/fuel-cell01.gif
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

The purpose of this lesson is to teach students how a spacecraft gets from the surface of the Earth to Mars. Students first investigate rockets and how they are able to get us into space. Finally, the nature of an orbit is discussed as well as how orbits enable us to get from planet to planet — spec...

Students acquire a basic understanding of the science and engineering of space travel as well as a brief history of space exploration. They learn about the scientists and engineers who made space travel possible and briefly examine some famous space missions.

Students learn how rocket thrust is generated with propellant. The two types of propellants are discussed—liquid and solid—and their relation to their use on rockets is investigated. Students learn why engineers need to know the different properties of propellants.

Students explore motion, rockets and rocket motion while assisting Spacewoman Tess, Spaceman Rohan and Maya in their explorations. First they learn some basic facts about vehicles, rockets and why we use them. Then, they discover that the motion of all objects—including the flight of a rocket and mo...
Copyright
© 2013 by Regents of the University of Colorado; original © 2010 Board of Regents, Washington State UniversityContributors
Stephen Dent (WSU College of Engineering); Courtney Herring (WSU Gene and Linda Voiland School of Chemical Engineering and Bioengineering)Supporting Program
CREAM GK-12 Program, Engineering Education Research Center, College of Engineering and Architecture, Washington State UniversityAcknowledgements
This content was developed by the Culturally Relevant Engineering Application in Mathematics (CREAM) Program in the Engineering Education Research Center, College of Engineering and Architecture at Washington State University under National Science Foundation GK-12 grant no. DGE 0538652. However, these contents do not necessarily represent the policies of the NSF, and you should not assume endorsement by the federal government.
Last modified: October 3, 2020






User Comments & Tips