Quick Look
Grade Level: 8 (8-10)
Time Required: 2 hours 30 minutes
(three 50-minute periods; can be over three days)
Lesson Dependency: None
Subject Areas: Physical Science, Science and Technology
NGSS Performance Expectations:

| MS-PS4-2 |

Summary
Students learn about the basic properties of light and how light interacts with objects. They are introduced to the additive and subtractive color systems, and the phenomena of refraction. Students further explore the differences between the additive and subtractive color systems via predictions, observations and analysis during three demonstrations. These topics help students gain a better understanding of how light is connected to color, bringing them closer to answering an overarching engineering challenge question.Engineering Connection
A deep understanding and exploration of the properties of light has enabled engineers and scientists to illuminate our world with everything from computer monitors, TV screens, and lasers to fiber optic cables and medical devices. For example, fiber optic cables give us the ability to communicate and transmit data over long distances at high rates by guiding light through refraction. Since first demonstrated in the 1840s, engineers and scientists have adapted fiber optics to many applications, including communication and medical imaging. Portable glucose monitors are one example of medical devices that use color change as a way to detect body chemicals. With this device, a diabetes patient can independently monitor his/her glucose levels, greatly increasing his/her quality of life. Before designing a solution to a challenge, engineers conduct research and gather information as a key step in the engineering design process. Through this legacy cycle lesson, students continue to gather the knowledge necessary to devise solutions to the engineering challenge outlined in lesson 1 of this unit.
Learning Objectives
After this lesson, students should be able to:
- Explain how objects appear to be a certain color.
- Explain how monitors and TVs display color.
- Describe opaque, translucent and transparent objects.
- Identify additive and subtractive color phenomena.
- Explain the difference between additive and subtractive color.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-PS4-2. Develop and use a model to describe that waves are reflected, absorbed, or transmitted through various materials. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This lesson focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Develop and use a model to describe phenomena. Alignment agreement: Ask questions that can be investigated within the scope of the classroom, outdoor environment, and museums and other public facilities with available resources and, when appropriate, frame a hypothesis based on observations and scientific principles.Alignment agreement: Construct an explanation that includes qualitative or quantitative relationships between variables that describe phenomena.Alignment agreement: | When light shines on an object, it is reflected, absorbed, or transmitted through the object, depending on the object's material and the frequency (color) of the light. Alignment agreement: The path that light travels can be traced as straight lines, except at surfaces between different transparent materials (e.g., air and water, air and glass) where the light path bends.Alignment agreement: A wave model of light is useful for explaining brightness, color, and the frequency-dependent bending of light at a surface between media.Alignment agreement: However, because light can travel through space, it cannot be a matter wave, like sound or water waves.Alignment agreement: | Structures can be designed to serve particular functions by taking into account properties of different materials, and how materials can be shaped and used. Alignment agreement: |
International Technology and Engineering Educators Association - Technology
-
Students will develop an understanding of the role of troubleshooting, research and development, invention and innovation, and experimentation in problem solving.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop an understanding of the relationships among technologies and the connections between technology and other fields of study.
(Grades
K -
12)
More Details
Do you agree with this alignment?
State Standards
South Carolina - Science
-
Waves (including sound and seismic waves, waves on water, and light waves) have energy and transfer energy when they interact with matter. Waves are a repeating pattern of motion that transfers energy from place to place without overall displacement of matter. All types of waves have some features in common. When waves interact, they superimpose upon or interfere with each other resulting in changes to the amplitude. Major modern technologies are based on waves and their interactions with matter.
(Grade
8)
More Details
Do you agree with this alignment?
-
The student will demonstrate an understanding of the properties and behaviors of waves.
(Grade
8)
More Details
Do you agree with this alignment?
-
Construct explanations for how humans see color as a result of the transmission, absorption, and reflection of light waves by various materials.
(Grade
8)
More Details
Do you agree with this alignment?
-
Summarize the behaviors of waves (including refraction, reflection, transmission, and absorption).
(Grade
8)
More Details
Do you agree with this alignment?
-
Explain how the absorption and reflection of light waves by various materials result in the human perception of color.
(Grade
8)
More Details
Do you agree with this alignment?
Worksheets and Attachments
Visit [www.teachengineering.org/lessons/view/clem_waves_lesson03] to print or download.Introduction/Motivation
(In advance, make copies of the All About Light—Notes Outline and Color Magic Demo Worksheet, one each per student, and have ready the demo supplies and preparation described in the Background section. Also [optional], prepare to show students the attached 15-slide Properties of Light Presentation to accompany the lesson introduction. The slides are "animated" so you can click to show the next item when ready.)
Today we are going to begin exploring the properties of light! We will start by talking about some of the basic properties of light and what it does when it strikes an object. Then, we will explore the characteristics of white light and learn what causes rainbows. We will also explore what it is about the properties of light that allows us to see a lemon as yellow and a strawberry as red. Finally, we will talk about how the properties of light are connected to communicating over the Internet. Let's get started!
(At this point, hand out the notes outlines and present the lecture material provided in the Background section, in tandem with the slides.)
(Next, so students can apply what they just learned, move to conduct the demos, starting with the following introduction.)
Who remembers what gives something color? Why do some objects appear red and other purple? (Listen to student responses.) As we just learned, it is because of a phenomenon called subtractive color. A red ball really has no color or light energy of its own. It reflects the wavelengths of white light that cause us to see red and absorbs all the other wavelengths. This results in the visual sensation of red by the viewer.
You were also just introduced to a phenomenon called additive color, which involves light emitted directly from a source. What are some examples? (Television and computer monitors create color by using the primary colors of light.)
Now, you are going to apply what you know during three demonstrations that will show us the phenomena of subtractive color and additive color. Here is a worksheet for you to fill out during the demonstrations. (Hand out the worksheets.)
For these color magic demonstrations, write down your initial observations, your predications about what you think will happen, and your final observations. Then, explain what happened and why. Make sure to note which demonstrations represented the additive and subtractive color system.
(Conduct the three demos, as described in the Background section.)
(After the demos, conclude by asking students which demos they thought represented additive and subtractive color phenomenon. Ask other questions, as provided in the Assessment section and Lesson Closure.)
Lesson Background and Concepts for Teachers
Legacy Cycle Information: This lesson falls into the research and revise phase of the legacy cycle. During this phase, students learn the basic concepts required to design solutions to the engineering challenge presented in lesson 1 of this unit. After lesson 3, students should be able to revise their initial thoughts, forming new ones that will help solve the challenge.
Properties of Light
(The following lecture material aligns with the slides.)
We use light everyday—to read books, light rooms and search for the unknown. Light is a small part of the electromagnetic spectrum. So, what are some properties of light?
How fast does it go? The speed of light! What is the speed of light? It is 300,000 km/s in space. That's pretty fast! In glass, light slows down to 197,000 km/s.
How big are light waves? They range from about 400 nm to about 700 nm in length. What is a nm? It is 1 x 10-9 meter, which is one billionth of a meter. This means light waves are very small.
What happens to light when it strikes an object? It does one of several things:
- It can be absorbed— it is transferred to the object (mainly as heat).
- It can be reflected—meaning it bounces off the object (for example, a mirror).
- It can be transmitted—or go through the object (for example, glass).
We classify objects according to what they do to light:
- Opaque objects do not allow light to pass through; they absorb or reflect all light.
- Translucent objects can be seen through, but not clearly; they absorb, reflect and transmit light (such as waxed paper or frosted glass).
- Transparent objects allow almost all of the light to pass through, so they can be seen through clearly (for example, window glass or clear plastic).
White light is made of all the colors of the rainbow. How do we know? Because a prism splits the white light into its component colors (see Figure 1).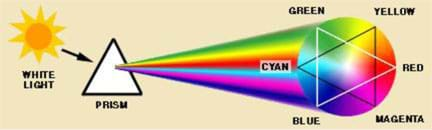
So, what do you think causes a rainbow? Well, raindrops in the air act like tiny prisms. Light enters the raindrop, reflects off the side and then exits the drop. During this process, the light is broken into a spectrum, just like it is in a triangular glass prism.
So, what makes a red car appear red and a blue car appear blue? Objects create color by subtracting or absorbing certain wavelengths of color while reflecting other wavelengths back to the viewer. This phenomenon is called subtractive color (see Figure 2). So back to the red car, the car really has no color; it reflects the wavelengths of white light that cause us to see red and absorbs most of the other wavelengths, giving us the visual sensation of red.
The subtractive color system uses colorants and reflected light. You start with an object that reflects light and use colorants, such as dyes or pigments, to subtract portions of the white light that is shining on the object. The primary colors are cyan, magenta and yellow.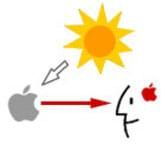
How do monitors and TVs display color? These produce color based on the additive color system, which involves light emitted directly from a source before an object reflects light. Think about it this way, you can watch TV or play on a computer without a light on in your room and still see color, but you need a light on in your room to see color in photographs on your wall. A TV screen or computer monitor creates color using the primary colors of light: red, blue and green. From these three colors a wide range of colors can be produced. When you turn on your TV or computer monitor, thousands of red, green and blue phosphor dots make up the images you see. The phosphor dots emit light when activated electronically. The combination of different intensities of these three colors produces all the colors on a video monitor. We cannot see the dots individually because of how close they are to one another.
Light and Reflection
The two types of light reflection are "regular" and "diffuse." The "regular" type occurs when light strikes a smooth surface, so that you can see an image on the surface because most or all of the reflected light reaches your eyes. A mirror is a good example of regular reflection. Plane (flat) mirrors reflect upright, same-size images, while curved mirrors change the shapes of images. "Diffuse" reflection occurs when a rough surface scatters the light in many different directions so that not all of it reaches your eyes and you do not see a reflection. (Note: Rough is a relative term; even a sheet of paper is usually "rough" enough so that images are not reflected.)
Light and Refraction
Light slows down as it goes from space into air. It slows down even more in water, and even slower in solids (glass). Why? Because the atoms get in the way.
We can use lenses to curve light for specific purposes. A lens is a clear, curved transparent object used to bend light. Convex (or positive) lenses converge (or focus) light and can form images. Concave (or negative) lenses diverge (or spread out) light rays. (See Figure 3.)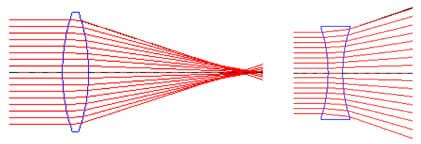
Practical Use: Fiber Optics
When light strikes a boundary between two transparent materials at the correct angle, all the light gets reflected. This is called total internal reflection and it is how fiber optics works. Total internal reflection allows the transmission of light-carrying information to travel great distances over curved paths.
Total internal reflection confines light within optical fibers (similar to looking down a mirror made in the shape of a long tube. Because the cladding has a lower refractive index, light rays reflect back into the core if they encounter the cladding at a shallow angle (red lines in Figure 4). A ray that exceeds a certain "critical" angle escapes from the fiber (yellow line in Figure 4).
Laser is an acronym for "light amplification by stimulated emission of radiation." Lasers use one wavelength of light so that all the crests and troughs line up (they are "in step"). Because they are all lined up, they do not interfere with each other and spread the light out like white light does (such as a flashlight). Lasers are used for welding, cutting materials (including biological tissue), reading and writing CDs, transmitting information through space or optical fibers, or simply as pointers.
Overview of Three "Color Magic" Demonstrations
Demo 1: pH Change and Color-Red Cabbage Indicator Demo (subtractive color system)
- Vinegar contains acetic acid and turns reddish when cabbage extract is added.
- Ammonia is a base and turns greenish when cabbage extract is added.
Demo 2: Goldenrod Paper Demo (subtractive color system)
- The paper changes to a red color because the dye in the paper is sensitive to bases.
Demo 3: Luminol Demo (additive color system)
- Luminol loses nitrogen and gains oxygen in an oxidation reaction. This causes the luminol to become excited. When it goes back to the ground state, it emits a photon of blue light. Bleach or hydrogen peroxide are required to cause the oxidation reaction.
Materials List for Demonstrations
Demo 1:
- half-filled glass of water
- half-filled glass of vinegar
- half-filled glass of general cleaning ammonia (available at most grocery stores)
- red cabbage extract (see preparation instructions, below)
Demo 2:
- sheet of goldenrod paper
- wax
- spray bottle of window cleaner containing ammonia
Demo 3:
- 0.23 g luminol (available at any chemistry supply store)
- 500 ml 0.1M sodium hydroxide, NaOH (available at any chemistry supply store)
- 50 ml bleach in 500 ml water or 25 ml of 3% hydrogen peroxide in 50 ml water
- 400 ml beaker
Demonstrations Preparations Before Students Arrive
Demo 1:
- Prepare red cabbage extract by these steps:
- Shred some red cabbage into a bowl and pour in enough boiling water to cover the cabbage.
- Let it sit for 10 minutes or longer (the longer it sits the better).
- Strain out the cabbage and keep the liquid.
- Fill three glasses each halfway with water, vinegar and ammonia.
Demo 2:
- Write a message on the goldenrod paper using wax (such as paraffin).
Demo 3:
- Mix 0.23 g luminol in 500 ml 0.1M sodium hydroxide (NaOH)
- Make either of the following two solutions and set aside 100 ml of it.
- bleach solution: 50 ml bleach to 500 ml water OR
- hydrogen peroxide solution: 25 ml 3% hydrogen peroxide in 50 ml water
- Put 100 ml of the luminol solution in a 400 ml beaker
With the Students
Demo 1: Color Indicator
- Ask students to make predictions on their worksheets about what they think will happen when you pour the cabbage extract into each cup. Ask for volunteers to share their predictions with the class.
- Pour some cabbage extract into the first cup (water).
- Ask students to record their observations and explanations for what happens.
- Pour some of the extract into the second cup (vinegar).
- Ask students to record their observations and explanations for what happens.
- Ask students if they would like to change their original predication about what will happen when the extract is poured into the third cup.
- Pour some of the extract into the third cup (ammonia).
- Have students record their observations and explanations for what happens.
- As a class, have students share their explanations for what happened. What is the difference between the liquid in the three cups? (Ask for volunteers to come up and smell [wafting!] the three liquids to help figure out why the color changes occurred.)
Demo 2: Goldenrod Paper
- Position the goldenrod paper so that the entire class can see it.
- Have students record on their worksheets their original observations and predications about what will happen when the paper is sprayed.
- Spray some of the window cleaner on the paper.
- Ask students to record on their worksheets their final observations and explanation about what happens.
- Engage the class in a discussion about the demonstration they just saw.
Demo 3: Luminol
- Have students record on their worksheets their initial observations about the beaker of liquid.
- Take the 400 ml beaker containing the luminol solution.
- Darken the room and add 100 ml of the bleach or peroxide solution.
- Ask students to record their observations and explanation for what happens.
- Engage the class in a discussion about the demonstration they just saw.
Conclude by asking students which demos they thought represented additive and subtractive color phenomenon. Ask other questions, as provided in the Assessment section and Lesson Closure.
Lesson Closure
Who can tell me the difference between opaque, translucent and transparent objects? And, what are the differences between additive and subtractive color systems?
Now that you're all experts in understanding how objects appear to be certain colors, tell me, what are some ways this might be applied in your everyday lives? (Listen to student ideas.) Those are great examples. What about computer monitors, TV screens, fiber optic cables and medical tests? These are common examples in which engineers apply their understanding of light and color to design all types of useful products and tools for us to use.
Fiber optic cables have given us the ability to communicate and transmit data over long distances at high rates. These cables work by guiding light through refraction, and were first demonstrated in the 1840s. Without a complete understanding of lights' properties, engineers and scientists would not have been able to adapt fiber optics to so many applications, including communication and medical imaging.
Biomedical engineers use color change as a method to detect a variety of chemicals found in the body. Portable glucose monitors are one example of such a medical device. These devices work by placing a drop of blood on a small test strip. The glucose in this drop of blood acts in a chemical reaction that yields a color. The strength of the color is connected to the amount of glucose present in the blood. These devices enable diabetes patients to monitor their glucose levels themselves.
Before designing and creating a solution to a challenge, engineers conduct research and gather information, just like you did today. This step is a crucial part of the engineering design process.
Vocabulary/Definitions
additive color system: Involves light emitted directly from a source, before an object reflects the light. Mixes various amounts of red, green and blue light to produce other colors. Examples include computer monitors and TVs.
concave or negative lens: A lens that diverges or spreads out light rays.
convex or positive lens: A lens that converges or focuses light, and can form images.
cyan: A highly saturated green-blue that is the complementary color of red and forms, with magenta and yellow, a set of primary colors.
laser: Acronym for light amplification by stimulated emission of radiation. Lasers only produce one wavelength of light, resulting in a beam of light that is very distinct and does not spread out.
opaque: A characteristic of an object that does not allow light to pass through; it absorbs or reflects all light.
prism : A transparent optical object that refracts light.
subtractive color system: Creates color by subtracting or absorbing certain wavelengths of color while reflecting other wavelengths back to the viewer. Examples include photographs and printed magazines.
translucent: A characteristic of an object that can be seen through, but not clearly; it absorbs, reflects and transmits light, such as wax paper or frosted glass.
transparent: A characteristic of an object that allows almost all the light to pass through, so it can be seen through clearly, such as glass or clear plastic.
Assessment
Note Taking: During the lecture, have students complete the All About Light—Notes Outline and refer to it for visuals that supplement the lecture material. Then, with the notes turned over on their desks, ask students various questions that were covered in the lecture material. Evaluate students' answers to gauge their mastery of the subject.
Worksheet: After the lecture, conduct the three demos. Have students use and complete the Color Magic Demos Worksheet, including all questions. Evaluate students' answers to see how well they apply what they learned.
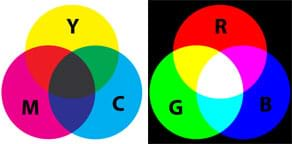
Closing Discussion: After the demos, lead a class discussion to see how well students predicted the results and asking them which demos they thought represented additive and subtractive color phenomenon (see Figure 5). Demos 1 and 2 were subtractive color systems and Demo 3 was an additive color system. (Tip: One of the easiest ways to tell the difference between additive and subtractive color systems are whether the colors can be seen in the dark. Additive colors can be seen in the dark because the light is emitted directly from a source. Subtractive color requires external light to be seen.) Then close with some commentary on the real-life application of what students learned today (see the Lesson Closure section).
Lesson Extension Activities
Hot and Cold Colors: Have students discover what color t-shirt it is best to wear on a hot, sunny day to stay cool! Working in pairs, have them conduct the experiment outlined on the Hot and Cold Colors Worksheet. Materials each group needs: thermometer, construction paper (at least four different colors), tape, lamp with light bulb, measuring ruler, protractor, timer, cup of room temperature water, paper towel. Have students complete the worksheet by graphing their data and answering the questions.
Laser Fun! Present students with the following challenge that requires them to understand wave properties, specifically reflection. Materials: laser pointer and 4-6 hand-size mirrors. Tell the students: Without lasers it would be impossible to listen to your favorite CDs. Lasers also make it possible to view our favorite DVDs. New uses for lasers are being invented all the time. Let's have some fun with lasers.
- Divide the class into groups of 4-6 students each (trying to keep the group sizes the same, if possible).
- Give each team a starting place from which to point a laser, and pick a target in the room to eventually hit.
- Have students arrange themselves wherever they want between the starting point and target and use the mirrors to bounce the laser beam to hit the target faster than the other teams. Since students will learn from the teams before them, change the starting (or target) position each time to keep it fair. Darken the room so that the laser is more easily detected.
- Keep time with a stopwatch.
- As time permits, give teams multiple opportunities to accomplish the exercise.
Research Project: Have students conduct web searches to discover the latest advances in laser technology and have them report their findings to the class. Encourage creative and interactive presentations.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn about the types of waves and how they change direction, as well as basic wave properties such as wavelength, frequency, amplitude and speed. During the presentation of lecture information on wave characteristics and properties, students take notes using a handout.
References
Color Mixing. 1997-2010. Robert Truscio. Accessed February 14, 2012. (Includes information on human vision and color, the primary colors of light, additive color mixing, and subtractive color mixing) http://home.comcast.net/~RTRUSCIO/COLORMX.htm
Electro-Optical Sciences. Published January 2006. Medical Technology, Business Week. Accessed February 14, 2012. (Example medical imaging device designed to aid in the detection of melanoma in skin tissue, an often deadly type of cancer; the gadget shines 10 different wavelengths of light into suspicious looking lesions, with each wavelength producing an image of the tissue. The system compares the images to a database of thousands of examples of melanoma to determine whether a biopsy is necessary.) http://images.businessweek.com/ss/06/01/medtech/source/7.htm
Xillix. Published January 2006. Medical Technology, Business Week. Accessed February 14, 2012. (Example medical product that uses light to diagnosis lung cancer; a long flexible tube shines white light and "blue" fluorescent light designed to measure the decreased response to fluorescence exhibited by precancerous and cancerous tissue, which are broadcast onto a monitor, the images display problem areas in easy-to-spot red) http://images.businessweek.com/ss/06/01/medtech/source/8.htm
Copyright
© 2013 by Regents of the University of Colorado; original © 2010 Clemson UniversityContributors
Ellen Zielinski; Courtney Faber; Marissa H. ForbesSupporting Program
Research Experience for Teachers (RET) Program, Center of Advancement of Engineering Fibers and Films, Clemson UniversityAcknowledgements
This lesson was developed through Clemson University's "Engineering Fibers and Films Experience – EFF-X" Research Experience for Teachers program, funded by National Science Foundation grant no. EEC-0602040. However, these contents do not necessarily represent the policies of the National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: January 28, 2021



User Comments & Tips