Quick Look
Grade Level: 6 (6-7)
Time Required: 1 hour
Lesson Dependency:
Subject Areas: Physical Science, Physics, Science and Technology
NGSS Performance Expectations:

| MS-PS3-5 |

Summary
Students learn more about the concept of energy conversion, and how energy transfers from one form, place or object to another. They learn that energy transfers can take the form of force, electricity, light, heat and sound and are never without some energy "loss" during the process. Two real-world examples of engineered systems—light bulbs and cars—are examined in light of the law of conservation of energy to gain an understanding of their energy conversions and inefficiencies/losses. Students' eyes are opened to the examples of energy transfer going on around them every day. Includes two simple teacher demos using a tennis ball and ball bearings. A PowerPoint® presentation and quizzes are provided.Engineering Connection
Energy conversions take place around us every day in many forms, including energy transfers in many engineered systems. Engineers routinely consider, examine and address issues of energy transfer/conversion and efficiency/loss with every invention, device and technology design they create. Engineers are always working on ways to make the energy transfer processes of power plants and distribution grids more efficient, too. In this activity, students consider the inefficiencies associated with the powering of light bulbs and cars, just as engineers involved in the design of these technologies do.
Learning Objectives
After this lesson, students should be able to:
- Explain the law of conservation of energy—that energy can neither be created nor destroyed.
- Identify energy conversion in some real-life examples.
- Identify general causes of energy loss.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-PS3-5. Construct, use, and present arguments to support the claim that when the kinetic energy of an object changes, energy is transferred to or from the object. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This lesson focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Science knowledge is based upon logical and conceptual connections between evidence and explanations. Alignment agreement: | When the motion energy of an object changes, there is inevitably some other change in energy at the same time. Alignment agreement: | Energy may take different forms (e.g. energy in fields, thermal energy, energy of motion). Alignment agreement: |
Common Core State Standards - Math
-
Reason abstractly and quantitatively.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Recognize and represent proportional relationships between quantities.
(Grade
7)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Energy is the capacity to do work.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
State Standards
California - Math
-
Reason abstractly and quantitatively.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Recognize and represent proportional relationships between quantities.
(Grade
7)
More Details
Do you agree with this alignment?
California - Science
-
Construct, use, and present arguments to support the claim that when the kinetic energy of an object changes, energy is transferred to or from the object.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
Worksheets and Attachments
Visit [www.teachengineering.org/lessons/view/ucd_energy_lesson03] to print or download.Introduction/Motivation
(Note: Be ready to show the class the Energy Conversion PowerPoint® Presentation, and have handy a few items for simple class demonstrations, as described in the Teacher Background section.)
(Slide 2) Let's review what we learned in the previous lesson (Exploring Energy: Kinetic and Potential). Motion energy is called kinetic energy and depends on the mass and speed of an object. We also discussed that stored energy is called potential energy, and that type of energy can be stored in many ways. Who can tell me a form of stored energy? (Answers: Gravitational, chemical, heat/thermal and elastic energy.)
(Slide 3) Today we're going to talk about energy transfer. Energy can be transferred from one form to another, from one object to another, or even from one place to another. For example, when an arrow is released, the stored potential/elastic energy of a stretched bow is converted into kinetic energy in the arrow. Energy transfer also takes many forms. Force, electricity, light, heat and sound are terms describing different kinds of energy transfers.

(Slide 4) What happens when you shoot a basketball? What are you exerting on the ball to give it energy? Listen to student responses. When you shoot a basketball, you exert a force (push) on the ball to give it more energy.
What energy conversion takes place in bungee jumping? Listen to student responses. In bungee jumping, gravitational potential energy is transferred to kinetic energy by the gravitational force on the person. Then that kinetic energy is transferred to elastic potential energy, then back to kinetic energy by the forces between the moving person and the elastic bungee cord.
(Slide 5) What about electricity? Electricity is a way for energy to be easily transferred. How? Listen to student responses. It can be transferred such as from a power plant to our homes and schools. Once the energy arrives in our buildings, it can be transferred into many useful forms by electrical appliances, like spinning and heating your clothes in a clothes dryer, providing light and heat, powering computers, playing movies and music, etc.

(Slide 6) What other ways can energy be transferred? Listen to student responses. Light is how energy from the sun is transferred through space to Earth. Sound is energy transfer through pressure waves in the air. Heat is the flow of thermal energy.
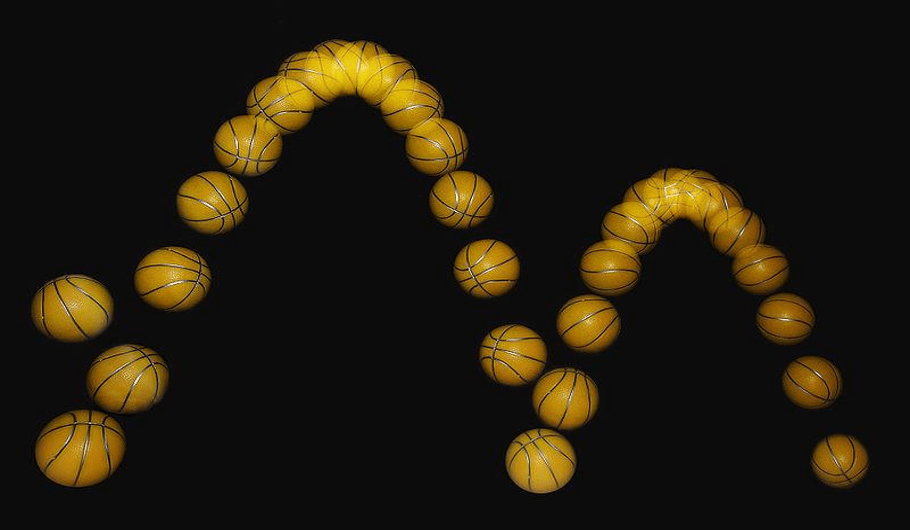
Bouncing tennis ball demonstration: Bounce a tennis ball on the floor. Ask students what ongoing energy changes are taking place? (Students should point out the ongoing changes in gravitational potential energy and kinetic energy.) Ask students what type of energy is it converted to? Mention that each time it hits the floor, some of the energy is converted to sound and heat (although the increase in heat in the tennis ball is not really noticeable). Ask students why each bounce is lower than the last? (Because of the energy "lost" to sound and heat, each bounce is lower than the last.)
(Slide 7-9) Energy transfer is very useful, but the transfer is never perfect. We usually cannot get a 100% energy conversion rate due to energy loss. When an energy conversion takes place, some energy gets converted to a form that is not useful to us. Keep in mind that energy is never created or destroyed; it just changes form. Thus, the term "energy loss" does not mean that the energy disappears; it just means that a portion of energy is converted into a form that is not desired or useful to us.
(optional) Ball bearing demonstration: With a piece of paper between them, hit together two large steel ball bearings. The heat of collision is enough to char the paper. Have students look at and smell that the paper has been burned where the bearings collided.
Who can fill in the blanks at the bottom of this slide (9)? (Listen to student answers; click for each answer to appear.) That's right, when energy is transferred, some energy is usually "lost" as heat, sound and light. Now let's look at some ways that we use energy conversion every day.
(Slide 10) Energy conversion makes light bulbs possible. How? A power plant transfers the chemical energy from natural gas or coal or another fuel into electricity. That electricity is carried by power lines to our homes and schools. Then the electricity is transferred to light by a light bulb. This is a convenient way to make light, but as we said before, energy transfer is never perfect.
(Slide 11) How much energy is "lost" in the use of light bulbs? This diagram shows how much energy it takes to power a 60-watt light bulb and how energy is "lost" along the way. (Walk students through the diagram; remind them that "lost" means transferred to a form that is not desired or useful.) Less than 1% of the energy we started with gets turned into light! (At this point in the lesson, discuss the broader implications of energy conversion and energy efficiency.) If we had a more efficient power plant, distribution grid or light bulb we could do more with less and we wouldn't need 188 joules of energy to create 900 lumens of light. New lighting technologies are already more efficient than the incandescent bulb used in this example (such as compact fluorescent and LED bulbs) and engineers are always working on ways to make power plants and distribution grids more efficient. (If desired, use this discussion to foreshadow future lessons on energy consumption, energy efficiency, pollution and global warming.)
(Slide 12) What about a car’s engine? What energy conversion takes place? (Listen to student responses.) A car's engine converts potential energy from gasoline or batteries into kinetic energy, or motion, so we can travel long distances.
(Slide 13) How much energy is "lost" in the powering of our cars? This chart shows that only 17-21% of the energy in gasoline goes to moving the car! The rest of the energy is "lost" due to various thermal effects, frictions and other factors. (Have students discuss the following question in small groups.) How can automotive engineers minimize these losses so cars can travel further on a gallon of gas? How can the energy transfer process be made more efficient?
(Slide 14) Write about something you saw today that wouldn't be possible without energy transfer. In your writing, make sure to answer the questions shown on this slide: What was the original source of energy? What was the final use of the energy? How was energy transferred? How was energy "lost"?
Lesson Background and Concepts for Teachers
Teach the lesson using the 14-slide Energy Conversion Presentation, a PowerPoint® file, along with the script and notes provided in the Introduction/Motivation section. The presentation is animated, so clicking brings up the next image, text or slide. The presentation contains review questions and activities that ask students to use their new energy knowledge. The associated activity, Maximum Mentos Fountain, also provides opportunities to discuss and review key concepts.
In addition, for two class demonstrations, have handy a tennis ball, two steel ball bearings (~2-inch diameter works well) and a piece of paper.
Associated Activities
- Maximum Mentos Fountain - Students observe the potential energy stored in Diet Coke® be converted into kinetic energy due to nucleation on the surface of Mentos® candies. Then the class is challenged to optimize the design of the basic soda/candy geyser made by the teacher. Three research teams investigate how different variables—nozzle shape, soda temperature, number of candies—affect fountain height. They combine their results to face-off in a final competition to see which fountain design blasts highest.
Vocabulary/Definitions
conservation of energy: A fundamental law in thermodynamics. It states that energy cannot be created or destroyed; it can only change from one form to another.
electricity: The flow of electric charge through a conductor. Energy can be transferred from one place to another and from one form to another by electricity.
energy: The ability to make things happen. More advanced definition: The ability to do work.
force: Energy can be transferred from one object to another by a force (by the objects pushing or pulling on each other). A force can also transfer energy from one form to another.
Assessment
Pre-Lesson Assessment
Concept Review: Use the four questions on slide 2 of the Energy Conversion Presentation to review as a class the concepts learned in the previous lesson: kinetic energy (motion) depends on the mass and speed of an object; stored energy is called potential energy and has many forms, such as gravitational, chemical, heat/thermal and elastic energy.
Post-Introduction Assessment
Discussion Questions: Use class discussions and student writing assignments to evaluate student knowledge. Throughout the presentation are many opportunities for quick assessments.
Informal Quizzes: Interject the Energy Quizzes & Answers throughout the lesson to gauge student comprehension.
Lesson Summary Assessment
Writing: To assess learning from this lesson, ask students to communicate using the energy concepts they have learned. Use the writing prompt on slide 14: Write about something you saw today that would not be possible without energy transfer. Include your observations about [the five questions provided on the slide]. As an alternative, assign students to explain the exploding water heater video (or other demonstration) from the beginning of the first lesson of this unit, explaining it in more detail than they did after the first lesson. See the four-minute video, MythBusters-Exploding Water Heater, at https://www.youtube.com/watch?v=jbreKn4PoAc.
Informal Quizzes: The Energy Quizzes & Answers may also be used to evaluate student learning from the previous lessons.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students makes sense of kinetic and potential energy, including various types of potential energy: chemical, gravitational, elastic and thermal energy. They identify everyday examples of these energy types, as well as the mechanism of corresponding energy transfers.

Students are introduced to the definition of energy and the concepts of kinetic energy, potential energy, and energy transfer. This lesson is a broad overview of concepts that are taught in more detail in subsequent lessons and activities in this curricular unit.

Students learn about the definition of heat as a form of energy and how it exists in everyday life. They learn about the three types of heat transfer—conduction, convection and radiation—as well as the connection between heat and insulation.
Copyright
© 2014 by Regents of the University of Colorado; original © 2013 University of California DavisContributors
Eric Anderson, Jeff Kessler, Irene ZhaoSupporting Program
RESOURCE GK-12 Program, College of Engineering, University of California DavisAcknowledgements
The contents of this digital library curriculum were developed by the Renewable Energy Systems Opportunity for Unified Research Collaboration and Education (RESOURCE) project in the College of Engineering under National Science Foundation GK-12 grant no. DGE 0948021. However, these contents do not necessarily represent the policies of the National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: May 5, 2021





User Comments & Tips