Quick Look
Grade Level: 5 (3-5)
Time Required: 1 hours 30 minutes
(Add 15-minutes at the beginning if the class makes the "polluted" water and sets up the aeration; can be split into two 45-minute sessions)
Expendable Cost/Group: US $3.00
Group Size: 3
Activity Dependency: None
NGSS Performance Expectations:

| 3-5-ETS1-2 |
| 3-5-ETS1-3 |
Summary
In this hands-on activity, students investigate different methods—aeration and filtering—for removing pollutants from water. Working in teams, they design, build and test their own water filters—essentially conducting their own "dirty water projects." A guiding data collection worksheet is provided.
Engineering Connection
Civil, chemical and environmental engineers work together to make existing water treatment systems better, and to develop new water treatment systems. Some engineers design state-of-the-art seawater treatment system technologies that process ocean water cost-effectively for safe domestic use.
Learning Objectives
After this activity, students should be able to:
- Use sight and smell to identify pollutants in a water sample.
- Explore what types of pollutants are removed from water by aeration and filtration.
- Design, build and test a water filtration system.
- Explain the role of engineers in water treatment systems.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
3-5-ETS1-2. Generate and compare multiple possible solutions to a problem based on how well each is likely to meet the criteria and constraints of the problem. (Grades 3 - 5) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Generate and compare multiple solutions to a problem based on how well they meet the criteria and constraints of the design problem. Alignment agreement: | Research on a problem should be carried out before beginning to design a solution. Testing a solution involves investigating how well it performs under a range of likely conditions. Alignment agreement: At whatever stage, communicating with peers about proposed solutions is an important part of the design process, and shared ideas can lead to improved designs.Alignment agreement: | Engineers improve existing technologies or develop new ones to increase their benefits, to decrease known risks, and to meet societal demands. Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
3-5-ETS1-3. Plan and carry out fair tests in which variables are controlled and failure points are considered to identify aspects of a model or prototype that can be improved. (Grades 3 - 5) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Plan and conduct an investigation collaboratively to produce data to serve as the basis for evidence, using fair tests in which variables are controlled and the number of trials considered. Alignment agreement: | Tests are often designed to identify failure points or difficulties, which suggest the elements of the design that need to be improved. Alignment agreement: Different solutions need to be tested in order to determine which of them best solves the problem, given the criteria and the constraints.Alignment agreement: | |
International Technology and Engineering Educators Association - Technology
-
Students will develop abilities to assess the impact of products and systems.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop an understanding of the effects of technology on the environment.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Waste must be appropriately recycled or disposed of to prevent unnecessary harm to the environment.
(Grades
3 -
5)
More Details
Do you agree with this alignment?
-
Models are used to communicate and test design ideas and processes.
(Grades
3 -
5)
More Details
Do you agree with this alignment?
-
Explain how various relationships can exist between technology and engineering and other content areas.
(Grades
3 -
5)
More Details
Do you agree with this alignment?
-
Develop innovative products and systems that solve problems and extend capabilities based on individual or collective needs and wants.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- Data Collection Worksheet, one per student
- 2-liter plastic bottle cut in half horizontally, as shown in Figure 1; ask students to bring empty bottles from home or get from local recycling center near you; wash before use; consider using the same ones used in Lesson 4's This Landfill is a Gas activity.)

- 3-inch square of mesh, such as fine nylon screen or fine cheese-cloth
- 1 rubber band
- 1 spoon or other stirring utensil; a chopstick works well
To share among all groups:
- filter materials, such as filter paper or large coffee filter (at least 6" in diameter), 6 cotton balls, ~6 cups soil, ~6 cups sand, 1 dozen large and small pebbles (total), ~6 cup activated charcoal (such as used for potting plants and in aquariums)
- aquarium aerator or a mechanical stirrer/mixer; aeration pumps for fish tanks work well
- measuring cups
- 2 large jugs/jars, ~1 gallon size, such as plastic gallon milk jugs with lids; for mixing/storing "polluted water" (recipe follows)
- "polluted water" made by mixing the following items in amounts at the teacher's discretion: water (enough to fill the jugs/jars ~¾ full), green liquid food coloring, soil, organic matter such as grass clippings and orange rinds, dishwashing detergent, vinegar, baking soda, salt, pepper, pieces of polystyrene foam (foam peanuts), small pieces of newspaper, and your own ideas for other items
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/cub_environ_lesson06_activity2] to print or download.Introduction/Motivation
Due to its incredible chemical properties, water is often considered the "universal solvent." It can mix with organic (natural) or synthetic (human-made) substances. Some of these products easily break down in water, while others break down very slowly, or perhaps even never. Water naturally cleans itself via filtration through the ground and evaporation via the water cycle.
At one time, communities disposed of their waste and garbage directly into lakes, streams and oceans. Now, most countries require that unclean (contaminated, polluted) water be treated before it is permitted to be released into natural bodies of water like lakes, rivers and oceans.
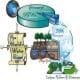
Generally, three different ways are used to treat raw sewage (waste) water before it is released. First, the liquid is given time to settle and then is exposed to oxygen by stirring or bubbling air through it (aeration). This helps many harmful organic pollutants react with oxygen and change into carbon dioxide and water. Second, the liquid is filtered to remove the particulate matter. Third, it is treated chemically with chlorine or ozone to kill any remaining harmful components such as bacteria.
Environmental, chemical and civil engineers work together to improve existing water treatment systems and design new ones to ensure that we have clean water both now and in the future. Today, let's imagine that we are engineers working for the Clean Water Environmental Engineering Company. The company has been asked to design a new water filtration system for a small community with a polluted water supply using limited materials. We are going to focus on the second step in the water treatment process, filtering. First, we are going to look at different types of filter material to determine which ones work well. Then each group in the company will design a filtering system to clean the polluted water. The best filtering system will be used in the small community.
Procedure
Before the Activity
- Prepare the "polluted water" supply and let it ripen in a sunny spot for a day or two. Alternatively, do this as a class demonstration so that students know exactly what is in the water. If you have student create the "polluted water" supply, have them write down the ingredients and their sight and smell (not taste) observations about the solution as it changes.
- Place the aerator/mixer in one sample of "polluted water" and let it sit overnight before Part 1. You will probably need to aerate a large sample of water for a day or so before Part 2, depending on how many groups choose to use aerated water for their best filter. Note: Aeration, the process of adding air to water, is often part of the water purification process in order to help many harmful organic pollutants react with oxygen and change into non-threatening carbon dioxide and water.
- Be sure to mix the solution thoroughly before preparing the student samples.
- Prepare the 2-liter bottles: cut them in half horizontally. Place a square of mesh over the bottle opening and secure it with the rubber band. If you use cheese cloth, you will need to replace it before Part 2.
- Make copies of the Data Collection Worksheet, one per person.
- Make a transparency or large chart of the class data section for use in Part 1.
- Review the water cycle with the class. Pay special attention to where the water can be purified. See the following book for a great description: The Magic School Bus – Wet All Over: A Book about the Water Cycle by Joanna Cole and Pat Relf (New York, NY: Scholastic Books, Inc., 1996).
With the Students
Part 1
- Divide the class into groups of three students each.
- Distribute a worksheet to each group.
- Remind the students that they are now working for the Clean Water Environmental Engineering Company and have been asked to design a new water filtration system for a small community with a polluted water supply. First, the company is going to look at different types of filter material to determine which ones work well. Then each group in the company will design a filtering system to clean up the polluted water.
- Give the following supplies to each group: a pre-cut 2-liter bottle, a ½-¾ cup (100-200 ml) sample of the "polluted water" in a beaker or cup, one type of "filter" (one group will not get a filter in order to test the mesh only), and a spoon.
- Ask each group to draw a picture of the "polluted water." Ask them to describe in words what it looks and smells like. Remind them to gently stir the solution and record their sight and smell observations on the worksheet. Remind students to never taste the solution.
- Ask students to come up with questions they think are important to answer throughout the activity about water filters (i.e., what makes a good filter?). Have them also write down on their worksheets their predictions for what they think their particular filter material will do.
- Ask students to set up their filters by placing the filter material into the inverted 2-liter bottle top, as shown in Figure 2. Note: Place the filter in the end of the bottle with the neck, so it functions like a funnel. Use the other half of the bottle as a stand. Prompt students to draw sketches of their setups on the worksheet.

- Ask students to gently stir the "polluted water" and then slowly pour it into the filter. Make sure the group with the filter paper is careful to not pour liquid above the top of the filter.
- Direct students to observe what happens during the filtration. Expect some filtrations to take longer than others. Remind students to record on their worksheets their observations and draw pictures of the filtered water.
- After all groups have collected data, share the results as a class by filling in the information on the transparency or chart made earlier. Have students record all team results in the class data section on the worksheets.
- As a class, look at the aerated sample. Discuss what aeration is and how it works (refer to the aeration explanation in the Before the Activity section).
- Ask students to work in their engineering design groups to design the best water filtration system given the filter material options and their choice of aerated or non-aerated water. Have them fill in the worksheet to record and explain their design choices. Permit them to use as many of the filtering materials as they want.
- Collect all supplies and dispose of used items properly. Rinse and save the 2-liter bottles Part 2.
Part 2
- Have students sort into their Part 1 groups.
- Give each team a prepared 2-liter bottle, ½-¾ cup (100-200 ml) of the "polluted water" in a beaker or cup (aerated or non-aerated, whichever they chose) and a spoon.
- Distribute the filter materials as needed. Note: It helps if teams each send a designated "materials" person from to collect their supplies from a central classroom location.
- Ask students to fabricate their groups' water filter systems and draw pictures of them on their worksheets.
- For testing, direct students to gently stir the polluted water supply and then slowly pour an amount into the filter. For teams that used filter paper, remind them to be careful not to pour the liquid above the top of the filter.

- Alert students to carefully observe and record on their worksheets what happens during the filtration process. Note: Some filtration systems take longer than others to process the "polluted water," so students should not worry if their filtration systems takes longer than other systems. Also have teams draw pictures on their worksheets of the filtered water.
- Direct students to record their results and answer the worksheet discussion questions, comparing answers with team members.
- After all the groups are finished, label and line up the filtered samples. Ask each team to present its filter system to the class (aka Clean Water Environmental Engineering Company). Have students discuss similarities and differences in the filters.
- Conclude with a class vote and discussion about which water is the cleanest and why.
Assessment
Pre-Activity Assessment: Part 1
Questions: Have students come up with questions they think are important to answer throughout the activity about water filters (i.e., what makes a good filter?). Tell them to keep the questions in mind and answer them at the end of the activity.
Picture Drawing: Ask each student to draw a picture of their group's "polluted water" in the space provided on the Data Collection Worksheet.
Prediction: Ask students to write down a prediction for what they think their particular filter materials will do in the space provided on the worksheet.
Recorded Observations: Ask students to stir the solution and record their observations on their worksheets.
Pre-Activity Assessment: Part 2
Picture Drawing: Ask each student to draw a picture of their best water filter in the space provided on worksheet.
Activity Embedded Assessment: Parts 1 and 2
Recorded Observations: Students observe and record what happens during the filtration process.
Picture Drawing: Have each student draw a picture of the filtered water in the space provided on the worksheet.
Post Activity Assessment: Part 1
Data Recording: After all groups have collected data, share the results as a class by filling in the information on the transparency or chart made earlier. Have students record all team results in the Class Data Section on the worksheet.
Clean Water Environmental Engineering Company Design Project: Ask students to work in their engineering design groups to design the best water filter system given the filter material options and their choice of aerated or non-aerated water. Have them record and explain their choices on the worksheet.
Post Activity Assessment: Part 2
Worksheet Questions: Have students answer the worksheet discussion questions, comparing answers with a team member. Collect and review student worksheets to assess their engagement, comprehension and mastery of the subject matter.
Engineering Presentations: Ask each team to present its filter system design to the class, explaining their logic. Examine the filtered solutions. Conclude with a class vote and discussion about which water is the cleanest and why.
Safety Issues
Remind students to only make sign and smell observations of the "polluted water" solution and never taste a solution, even if it looks "clean."
Troubleshooting Tips
Have some paper towels, rags or sponges on hand in-case of spills.
Consider any student allergies before creating the dirty water sample.
Advise students to fold the filter paper so it fits into the bottle top and suggest they pre-wet the paper so that it sticks to the sides of the "funnel." An eyedropper and tap water are useful for pre-wetting the filter paper.
Remember to dispose of the waste from this experiment properly! Typically, the "polluted water" solution can be poured down the drain. But if any contaminating chemicals were used, dispose of it using responsible disposal methods.
Activity Extensions
Provide students with pH paper and a pH guide so they can determine the pH of the solution during different stages of the process: plain water, "polluted water" before treatment, after aeration, after filtering with one filter, and after using their final filter. Discuss how the different components in the solution affect the pH. How would the pH of the solution affect the rest of the environment? (Refer to pH table.)
Ask students to measure the volume before and after filtration. Younger students can describe it as more or less or use measuring spoons/cups. Older students can use labeled beakers or graduated cylinders.
Experiment with some simple chemical treatments. For example, add chlorine to a water sample as a class demo or with older students. Remember to wear protective equipment when handling chemicals!
Ask students: Does the order of the filter layers matter? Why or why not?
Direct students to filter their samples more than once, keeping a small sample after each filtration for comparison purposes. Does the water get (visually) cleaner on subsequent filtrations? Why or why not?
Activity Scaling
For younger students, conduct the activity as a demo with fewer filter choices. Demonstrate each filter type individually and then ask students to predict what will happen when both filter types are used together. Ask students to draw pictures of the results.
For older students, let the teams work more independently so more time is spent on the design portion of the project. Ask students to make their own suggestions for filter materials and other ways to treat the "polluted water." Have students bring in some materials from home to test as filters and have each team test its own items after you have modeled the filtration procedure.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn about water quality testing and basic water treatment processes and technology options. Biological, physical and chemical treatment processes are addressed, as well as physical and biological water quality testing, including testing for bacteria such as E. coli.

Through the use of models and scientific investigation, students explore the causes of water pollution and its effects on the environment. Through the two associated activities, they investigate filtration and aeration processes that are used for removing pollutants from water.
References
Cole, Joanna and Relf, Pat. The Magic School Bus – Wet All Over: A Book About the Water Cycle. New York, NY: Scholastic Inc., 1996 (ISBN 0-590-50833-4).
Glencoe Science: An Introduction to the Life, Earth and Physical Sciences. Student Edition. Blacklick, OH: Glencoe/McGraw-Hill, 2002.
Hassard, Jack. Science as Inquiry – Active Learning, Project-Based, Web-Assisted and Active Assessment Strategies to Enhance Student Learning. Tucson, AZ: Good Year Books, 1999. (ISBN 0-673-57731-7)
Lucas, Eileen. Water: A Resource in Crisis. Chicago, IL: Children's Press, Inc., 1991.
Prentice Hall Science. Ecology Earth's Natural Resources Activity Book. NJ: Prentice Hall, Inc., 1993.
Spurling Jennett, Pamela. Investigations in Science – Ecology. Westminster, CA: Creative Teaching Press, Inc., 1995.
Stille, Darlene. The New True Book – Water Pollution. Chicago, IL: Childrens Press, Inc., 1991.
Copyright
© 2005 by Regents of the University of ColoradoContributors
Amy Kolenbrander; Jessica Todd; Malinda Schaefer Zarske; Janet YowellSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under grants from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and National Science Foundation (GK-12 grant no. 0338326). However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: November 11, 2020



User Comments & Tips