Quick Look
Summary
While many students understand that fruits and vegetables can give our bodies energy, they may not know that these same fruits and vegetables can power small electrical devices! In this activity, students experiment with various fruits and vegetables to determine the amount of electrical energy each produces. Students use an engineering design process to determine an optimal circuit design configured using a selection of fruits and vegetables. After evaluating the effectiveness of the fruits and vegetables individually, they design an electrical circuit that produce sufficient energy to power a small electronic device.
Engineering Connection
Like engineers, in this activity students use the process of data collection and analysis to design an electrical circuit that incorporates various fruits and vegetables that can deliver sufficient electrical energy to power a small electrical device. Students employ an engineering design process when using their data to redesign their circuits for optimal performance. The knowledge gained from recording the amount of electrical energy produced by each food type informs the decision on how to configure the fruits and vegetables to form an electrical circuit that produces sufficient electrical energy to power a small electronic device. Students get an introduction to circuit design, which is a fundamental skill of an electrical engineer. Students also learn about how the chemical make-up of the materials used in the battery influences the amount of power produced as a chemical or material science engineer develops novel materials to enhance the production of and flow of electrons through an electrical circuit for optimal power and energy production. Students also participate in the production of electrical energy from a non-fossil fuel source, which is a fundamental objective of environmental engineers concerned with man-made global climate change induced by greenhouse gases resulting from the use of fossil fuels.
Learning Objectives
After this activity, students should be able to:
- Create a simple circuit.
- Describe the primary parts of a battery.
- Produce and measure voltage and current from a fruit/vegetable battery system.
- Determine the circuit set-up and fruit/vegetable for optimal energy production by comparison of power (calculated) generated by fruits/vegetables using the formula used to calculate power.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-PS3-5. Develop and use a model of two objects interacting through electric or magnetic fields to illustrate the forces between objects and the changes in energy of the objects due to the interaction. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Develop and use a model based on evidence to illustrate the relationships between systems or between components of a system. Alignment agreement: | When two objects interacting through a field change relative position, the energy stored in the field is changed. Alignment agreement: | Cause and effect relationships can be suggested and predicted for complex natural and human designed systems by examining what is known about smaller scale mechanisms within the system. Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-4. Develop a model to generate data for iterative testing and modification of a proposed object, tool, or process such that an optimal design can be achieved. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Develop a model to generate data to test ideas about designed systems, including those representing inputs and outputs. Alignment agreement: | Models of all kinds are important for testing solutions. Alignment agreement: The iterative process of testing the most promising solutions and modifying what is proposed on the basis of the test results leads to greater refinement and ultimately to an optimal solution.Alignment agreement: | |
Common Core State Standards - Math
-
Use appropriate tools strategically.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Reason abstractly and quantitatively.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Construct and interpret scatter plots for bivariate measurement data to investigate patterns of association between two quantities. Describe patterns such as clustering, outliers, positive or negative association, linear association, and nonlinear association.
(Grade
8)
More Details
Do you agree with this alignment?
-
Reason quantitatively and use units to solve problems.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Solve equations and inequalities in one variable
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Solve linear equations and inequalities in one variable, including equations with coefficients represented by letters.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Summarize, represent, and interpret data on a single count or measurement variable
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Represent data on two quantitative variables on a scatter plot, and describe how the variables are related.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Students will develop an understanding of the characteristics and scope of technology.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop an understanding of the attributes of design.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop an understanding of engineering design.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop an understanding of the relationships among technologies and the connections between technology and other fields of study.
(Grades
K -
12)
More Details
Do you agree with this alignment?
State Standards
New Jersey - Math
-
Use appropriate tools strategically.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Reason abstractly and quantitatively.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Construct and interpret scatter plots for bivariate measurement data to investigate patterns of association between two quantities. Describe patterns such as clustering, outliers, positive or negative association, linear association, and nonlinear association.
(Grade
8)
More Details
Do you agree with this alignment?
-
Reason quantitatively and use units to solve problems.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Solve equations and inequalities in one variable
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Solve linear equations and inequalities in one variable, including equations with coefficients represented by letters.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Summarize, represent, and interpret data on a single count or measurement variable
(Grades
9 -
12)
More Details
Do you agree with this alignment?
-
Represent data on two quantitative variables on a scatter plot, and describe how the variables are related.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
New Jersey - Science
-
Develop a model to generate data for iterative testing and modification of a proposed object, tool, or process such that an optimal design can be achieved.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
Materials List
For the teacher, selected videos are provided to assist with the activity:
- How Batteries Work by TED-Ed: https://www.youtube.com/watch?v=9OVtk6G2TnQ
- How to Make a Lemon Battery by SciShow: https://www.youtube.com/watch?v=GhbuhT1GDpI (3:21-min; video about fruit batteries)
- Projector and computer to show the YouTube videos on battery technology
Each group needs:
- two or three different types of fruits, such as a lemon, orange, grapefruit or a vegetables, such as, a potato, cucumber (or a pickle), or onion
- penny or copper strip
- galvanized zinc nail or screw
- folded strip of aluminum foil
- 3 wires with alligator clips attached
- 2 coated electrical wires
- disposable gloves (1 pair per student)
- Student Lab Handout
In addition to the materials listed above, students should have access to the following items either at their group table or in a centralized location in the classroom:
- multimeter
- ruler
- LED bulbs
- calculators
- breadboard (optional)
- cleaning materials (such as cleaning cloths or paper towels)
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/rut-2497-food-electrical-circuit-device-design] to print or download.Pre-Req Knowledge
Students should:
- Have a basic understanding of circuits.
- Be able to identify independent and dependent variables.
- Know how to graph information provided in a table.
- Know how to compose a testable hypothesis.
Introduction/Motivation
Small electronic devices such as cell phones, tablets, and hand-held digital media players are ever-present in our lives, and are practically an indispensable tool for communication. These devices are always on (think about the last time you turned off your device and put it away) and we are engaged with them all the time. How do you feel when your phone has 10% or even 5% of battery life left, and you aren’t able to charge it?
While we understand the importance of power and energy in our lives, how it is produced and transferred to operate electrical devices is not always transparent. In this activity, we will learn about the primary components used in an electrochemical cell, like a battery, and will fabricate our own batteries! We will measure key battery performance characteristics such as voltage and current, and plot the power of produced by cells in series or current. We will also learn how to create our own circuit to power an LED light and design a system to produce enough power to turn on a cell phone.
How is this activity related to engineering? We will learn how to design electrochemical cells using household materials and analyze the cells performance via measurement of current and voltage as material science, electrical and chemical engineering professionals. In the activity, we will also calculate the theoretical voltage of cells and compare these values with experimental values that they measure from their experiments. In addition to examining the performance of the cells by graphically by plotting the current and voltage of battery systems as a function of cell configuration, we will perform a parametric analysis in the engineering design cycle to determine the best configuration and design of battery
Procedure
Background
Students learn about batteries and how mathematical models can be used to describe the behavior of battery cells. In this activity, students will produce batteries from household materials using fruits and vegetables as electrolyte material and metal materials as anode and cathodes, calculate the theoretical voltage of various types of cells, measure current and voltage from cells and use mathematical relationships to predict how different configurations of batteries can increase voltage or current for powering an LED light. Students will pretend to be shipped wrecked on a deserted island where they need to produce light for rescue teams to find them. The goal of the project is for students to design a system to turn on an LED light. This activity is broken up into three main parts.
Before the Activity
The teacher should present the following scenario to the class and introduce the Pre-Activity Discussion Questions.
Present the following question to the students: Have you ever been out with your friends or running errands with a parent and your cell phone ‘died’ and you thought “Oh no!? What can I do now?” Then to make the moment worse, you find that you’ve forgotten your charger somewhere. Explain to students that many of the electronics that we carry with us today like our cell phones and iPads require power. Ask them to discuss in a classroom format or in groups the Pre-Activity Discussion Questions.
After discussing the Pre-Activity questions, explain that they will be doing an activity allows students to learn about batteries and incorporate math in the analysis of the batteries. Students will produce different batteries using different fruits and vegetables as the electrolyte and various metals as anode/cathode materials with the goal of determining the optimal electrolyte/anode/cathode active materials. They will compare their experimental measurement of voltage with theoretical calculations of the voltage potential using the Equation 1, where Voxid is the oxidation reaction and Vreduc is the reduction reaction.
Vtheor = Voxid - Vreduc (1)
Students will determine the amount of electrical energy that various foods produce. Electrical measurements are taken so an electrical power value is calculated. That data is subsequently graphed and analyzed. The findings from the data analysis informs the students how to configure circuits made of the foods evaluated to achieve the desired which are tested for the amount of electrical energy produced from each. The results are used to determine an optimal circuit design which is capable of producing sufficient electrical energy to power a small electrical device.
After students have completed the discussion questions, begin to discuss the parts of a battery system and circuit.
As the following question: Does anyone know how a battery works? Show the video that describes the primary components in a battery, anode, cathode, and electrolyte, and how they to work together in producing power from the flow of electrons from in oxidation/reduction reactions.
- How Batteries Work by TED-Ed: https://www.youtube.com/watch?v=9OVtk6G2TnQ (describes how batteries work)
After watching the video describing how batteries work, review battery fundamentals and types (see Figure 1) with the class described below.

What is a Battery?
A battery is a standalone electrochemical unit (often called a cell) that has the potential to supply energy by converting chemical energy into electrical energy. The materials in the battery, such as the cathode, anode, electrolyte and the way in which they are configured within the battery (design) influence the amount of power that a battery can provide.
Electrical energy is a form of energy resulting from the flow of an electric charge (positive or negative) (electrons). Keep in mind that electrons flow because there is a force acting on the electrons. The energy carried by flowing electrons depends on the force inducing the flow and the distance traveled. Electrons are inherently attracted to positive charge and will travel towards positive charge if a pathway exists.
General concepts to review prior to having students do the activity.
- Batteries consist of chemical substances that react to either release or consume an electron. These types of reactions are called oxidation-reduction reactions as shown in Figure 2, that when added together using Equation (1) can be used to compute the theoretical voltage delivered by an electrochemical cell.
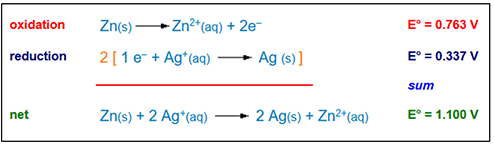
- Key components in a battery are described below and illustrated in Figure 3.
- They store electrical energy for on-demand use.
- Batteries have three key components all housed in a single container: a chemical substance that supplies electrons, a chemical substance that is a receiver of electrons, and a fluid composed of charged chemical constituents referred to as an electrolyte.
- The chemical substances supplying and receiving charge (+)/electrons are accessed through exposed conductive tips which are referred to as terminals; through these terminals electrical energy is extracted from the battery when charge (positive)/electrons flow along a conductive pathway between the two substances, that is an electrical circuit.
- The electrolyte fluid within the battery has 3 primary functions essential for the operation of a battery: i) keeps charge flowing through the battery system, ii) ensures no net charge builds up in the battery even though chemical reactions are occurring that result in the formation of charged species and iii) enables electrical connectivity between the 2 types of chemical substances.
- Typically the chemical substance that supplies electrons is a metal referred to generically as an electrode, specifically as the anode.
- The anode is where the oxidation reaction occurs; the chemical substance making up the anode itself is oxidized, and the released electron(s) travel(s) through the external circuit to arrive at the cathode due to the attractive pull of the (+) cathode.
- Typically the chemical substance that receives the electrons is a metal as well referred to generically also as an electrode, more specifically as the cathode.
- The cathode is where the reduction reaction occurs; the chemical substance making up the cathode can itself be reduced by the action of the arriving electron(s) which traveled through the external circuit due to the attractive pull of the (+) cathode, or the electrons can be passed to a charged species in the electrolyte solution.
- Remember that negative charges exit the anode; for a battery this terminal is always negative.
- Remember that the cathode terminal is positive which attracts negative charges; for a battery this terminal is always positive.
- The external circuit formed by making a conductive connection between the two terminals allows for charge (+)/electrons flow which compels the oxidation-reduction chemical reaction to occur enabled by the charge balancing action of the electrolyte.
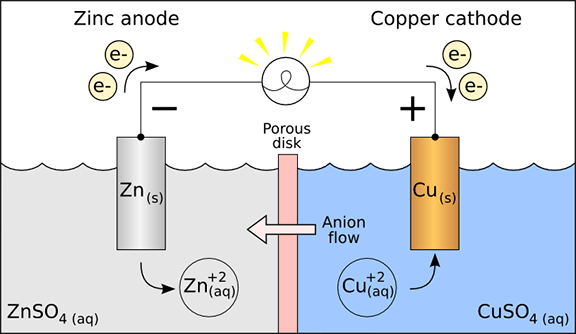
With the Students
After reviewing the background information, propose the scenario provided in the Student Lab Handout to establish the engineering problem the students are going to solve in the activity.
- Pose the following scenario to the class: Suppose you and your class have taken a school trip on a cruise and a hurricane pushes your ship onto the shore. Within minutes you and your class discover that the you have lost power to your ship and must leave the ship to see if there are any people on the island who may be able to assist you with getting back home. After walking around on the island you and your classmates realize that the island is uninhabited and that you need to be rescued before your fresh water supplies and food are depleted. You decide that you need to power LED lights to alert a local rescue team to come to save you.
- You have a few items at your disposal to assist you in powering the LED lights such as fruit and vegetable, and wires. You will be tasked with making your own batteries from household supplies and designing an optimal circuit that you hope will eventually be used to power a LED light. In this activity, we will see if we can provide enough power for our cell phone or an LED light using our engineering skills, which will require us to know about mathematical relationships between the variables: current and voltage and power production from battery cells.
- Explain to the students that they will be constructing batteries from fruits and vegetables. Play the video illustrating how to construct fruit batteries: How to Make a Lemon Battery by SciShow: https://www.youtube.com/watch?v=GhbuhT1GDpI.
- After watching the video pass out the Student Lab Handout to the students. In the handout, students will be asked to formulate hypotheses regarding the optimal electrolyte, distance between anode/cathode and battery configuration to power an LED light.
- As put forth in their hypotheses, instruct students to assemble the proposed electrical circuits using the evaluated fruits and/or vegetables. Students will test their hypothesis with measurements of the current and voltage delivered from each cell.
- Students will first begin by determining the optimal electrolyte by varying fruits, and then determine the best anode and cathode materials. They will plot the results in tables.
- The general set up for a battery cell is shown in Figure 4. In addition to testing different anode/cathode and electrolyte materials, students will be asked to configure their batteries in series, which adds the voltage of each cell as shown in Figure 5 and batteries in parallel to add current as shown in Figure 6.
- Once determining the best cell materials, they will calculate the relationship between cells in series and parallel to determine the optimal configuration of cells to power the LED light. Students will also use Equation (1) to calculate the theoretical voltage of each cell given materials used for anode and cathode. A table of common electrode reactions is provided in Figure 7.
- Remind students that when calculating the theoretical voltage, the reactions must be oriented in the correct direction, i.e. in oxidation reactions, electrons are released and the voltage is positive and in reduction reactions, electrons are added for the reaction to be positive. So, reactions presented as negative in the Figure 6 must be arranged properly for the correct half reaction to proceed and the theoretical calculation to be correct.
- Students will use their data to design the optimal battery configuration to power a LED light. The teacher can assist the students in reviewing the charging requirements of specific LED lights.



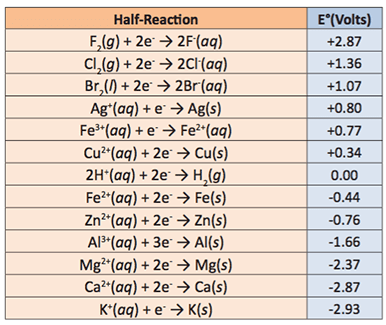
Vocabulary/Definitions
circuit: Path in which electrons from a voltage or current source flow.
current: A flow of electric charge is measured in units of Amperes (Amp). Symbol for current is usually, I.
dependent variable: Variable (often denoted by y ) whose value depends on that of another.
independent variable: Variable (often denoted by x ) whose variation does not depend on that of another.
power : Energy that is produced by mechanical, electrical, or other means and used to operate a device. The symbol for power is P. Power may be calculated using the equation, P = I V.
voltage: An electromotive force or potential difference expressed in Volts (V). The symbol for voltage is usually, V.
Assessment
Pre-Activity Assessment
Discussion Questions: Students learn about how they interact with electrochemical cells like batteries in their everyday lives by doing the Pre-Activity Discussion Questions and watching a video showing a woman have a tantrum when her cell phone loses its power when riding a train.
The teacher will introduce students to electrochemical batteries via a video, How Batteries Work by TED-Ed: https://www.youtube.com/watch?v=9OVtk6G2TnQ and in-class discussion of battery operation, battery vocabulary and mathematical functions that allow students to calculate power.
An overview of critical vocabulary and battery fundamental components is provided in this document. After the teacher reviews to key concepts pertaining to batteries, students will watch a video about fruit batteries, How to Make a Lemon Battery by SciShow: https://www.youtube.com/watch?v=GhbuhT1GDpI.
Activity-Embedded Assessment
Handout: A handout detailing the steps in the activity is provided in the Student Lab Handout.
Post-Activity Assessment
Post-Quiz: Following the activity, students will complete the Post-Activity Quiz.
Investigating Questions
Can sufficient electrical energy be achieved in an electrical circuit configured using fruit and/or vegetables to yield sufficient power to increase the charge level of a cell phone battery a bar or more? (Answer: Yes, if enough fruits are used, like using 30 lemons connected in series: https://www.youtube.com/watch?v=phSbh4Rt0PY)
Safety Issues
Be aware of student food allergies.
Have students wear gloves when executing the activity.
Have students wipe their work area clean using a wet cloth to cleanse any food residue from the surfaces.
Be mindful of how the alligator clips are positioned, including the negative/positive placements.
Troubleshooting Tips
Be sure that multimeters are functioning. Have a few resistors of known resistance (1 ohm, 10 ohm, etc.) available to be used to confirm that the current measurements obtained from the multimeter are valid.
If the LED bulb does not light up, make sure it is connected in the right direction (the longer wire should be connected to the positive side of the battery).
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn that charge movement through a circuit depends on the resistance and arrangement of the circuit components. In one associated hands-on activity, students build and investigate the characteristics of series circuits. In another activity, students design and build flashlights.

Students learn about current electricity and necessary conditions for the existence of an electric current. Students construct a simple electric circuit and a galvanic cell to help them understand voltage, current and resistance.

Students are introduced to several key concepts of electronic circuits. They learn about some of the physics behind circuits, the key components in a circuit and their pervasiveness in our homes and everyday lives.

Students create solar cells using dyes extracted from fruits and vegetables to test how color affects the conversion of solar energy to electrical energy. They apply the engineering design process by asking questions and hypothesizing which fruit or vegetable will produce the most energy. After asse...
Copyright
© 2020 by Regents of the University of Colorado; original © 2017 Rutgers UniversityContributors
Elizabeth Parker; Dr. Kimberly Cook-Chennault; Kimberly Bassolino; Dr. Valarie ThomasSupporting Program
Rutgers University Research Experience for Teachers in Engineering – Green Energy TechnologyAcknowledgements
This curriculum was based upon work supported by the National Science Foundation under Research Experience for Teachers in Engineering (RU RET-E) for Green Energy and Technology grant no. 1407266. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation.
Last modified: December 15, 2025








User Comments & Tips