Quick Look
Grade Level: 6 (6-7)
Time Required: 5 hours
(five 60-minute sessions)
Expendable Cost/Group: US $0.00
Group Size: 3
Activity Dependency: None
Subject Areas: Biology, Chemistry, Earth and Space, Measurement, Problem Solving
NGSS Performance Expectations:

| MS-ETS1-1 |
| MS-ETS1-2 |
| MS-PS1-1 |
| MS-PS1-2 |
| MS-PS1-3 |

Summary
Students learn about polymers, plastics, and bioplastics by exploring both natural polymers (e.g., hair, DNA, and cotton) and synthetic polymers found in everyday items (e.g., clothing, toothbrushes, and carpets). They begin by modeling polymer chains from paper clips, starting with simple chains and then modifying their structures to observe how changes affect flexibility, rigidity, and strength. Students also consider the environmental impacts of synthetic plastics, including their persistence in the environment for hundreds of years, and the challenges of recycling, such as high energy costs. They explore how bioplastics (i.e., materials with plastic-like properties that are biodegradable) could offer a more sustainable alternative. Using a guided recipe, students design their first bioplastic piece and then modify the recipe to achieve a different outcome, applying the engineering design process and analyzing how changes in ingredients influence material properties.Engineering Connection
This activity connects engineers who work with materials, chemicals, and the environment. Materials engineers design and test materials to make them stronger, more flexible, or more durable. Chemical engineers figure out how to turn natural or synthetic substances into useful products such as plastics or bioplastics. Environmental engineers focus on making materials safer for the planet, reducing pollution, and finding sustainable alternatives.
Learning Objectives
After this activity, students should be able to:
- Model polymer structures using everyday materials to understand how polymers form plastics.
- Design and create a bioplastic using specific materials and constraints.
- Test and evaluate bioplastic performance by measuring shrinkage and observing flexibility and rigidity.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-1. Define the criteria and constraints of a design problem with sufficient precision to ensure a successful solution, taking into account relevant scientific principles and potential impacts on people and the natural environment that may limit possible solutions. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Define a design problem that can be solved through the development of an object, tool, process or system and includes multiple criteria and constraints, including scientific knowledge that may limit possible solutions. Alignment agreement: | The more precisely a design task's criteria and constraints can be defined, the more likely it is that the designed solution will be successful. Specification of constraints includes consideration of scientific principles and other relevant knowledge that is likely to limit possible solutions. Alignment agreement: | All human activity draws on natural resources and has both short and long-term consequences, positive as well as negative, for the health of people and the natural environment. Alignment agreement: The uses of technologies and any limitations on their use are driven by individual or societal needs, desires, and values; by the findings of scientific research; and by differences in such factors as climate, natural resources, and economic conditions.Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-2. Evaluate competing design solutions using a systematic process to determine how well they meet the criteria and constraints of the problem. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Evaluate competing design solutions based on jointly developed and agreed-upon design criteria. Alignment agreement: | There are systematic processes for evaluating solutions with respect to how well they meet the criteria and constraints of a problem. Alignment agreement: | |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-PS1-1. Develop models to describe the atomic composition of simple molecules and extended structures. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Develop a model to predict and/or describe phenomena. Alignment agreement: | Substances are made from different types of atoms, which combine with one another in various ways. Atoms form molecules that range in size from two to thousands of atoms. Alignment agreement: Solids may be formed from molecules, or they may be extended structures with repeating subunitsAlignment agreement: | Time, space, and energy phenomena can be observed at various scales using models to study systems that are too large or too small. Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact to determine if a chemical reaction has occurred. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Analyze and interpret data to determine similarities and differences in findings. Alignment agreement: Science knowledge is based upon logical and conceptual connections between evidence and explanations.Alignment agreement: | Each pure substance has characteristic physical and chemical properties (for any bulk quantity under given conditions) that can be used to identify it. Alignment agreement: Substances react chemically in characteristic ways. In a chemical process, the atoms that make up the original substances are regrouped into different molecules, and these new substances have different properties from those of the reactants.Alignment agreement: | |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-PS1-3. Gather and make sense of information to describe that synthetic materials come from natural resources and impact society. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Gather, read, and synthesize information from multiple appropriate sources and assess the credibility, accuracy, and possible bias of each publication and methods used, and describe how they are supported or not supported by evidence. Alignment agreement: | Each pure substance has characteristic physical and chemical properties (for any bulk quantity under given conditions) that can be used to identify it. Alignment agreement: Substances react chemically in characteristic ways. In a chemical process, the atoms that make up the original substances are regrouped into different molecules, and these new substances have different properties from those of the reactants.Alignment agreement: | Structures can be designed to serve particular functions by taking into account properties of different materials, and how materials can be shaped and used. Alignment agreement: Engineering advances have led to important discoveries in virtually every field of science, and scientific discoveries have led to the development of entire industries and engineered systems.Alignment agreement: The uses of technologies and any limitations on their use are driven by individual or societal needs, desires, and values; by the findings of scientific research; and by differences in such factors as climate, natural resources, and economic conditions. Thus technology use varies from region to region and over time.Alignment agreement: |
Common Core State Standards - Math
-
Use ratio and rate reasoning to solve real-world and mathematical problems, e.g., by reasoning about tables of equivalent ratios, tape diagrams, double number line diagrams, or equations.
(Grade
6)
More Details
Do you agree with this alignment?
-
Summarize numerical data sets in relation to their context, such as by:
(Grade
6)
More Details
Do you agree with this alignment?
-
Use variables to represent two quantities in a real-world problem that change in relationship to one another; write an equation to express one quantity, thought of as the dependent variable, in terms of the other quantity, thought of as the independent variable. Analyze the relationship between the dependent and independent variables using graphs and tables, and relate these to the equation.
(Grade
6)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Assess design quality based upon established principles and elements of design.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Refine design solutions to address criteria and constraints.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Explain how knowledge gained from other content areas affects the development of technological products and systems.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
Materials List
Each student needs:
- 1 Polymers, Plastics, and Bioplastics Booklet
- 1 writing utensil
- 1 pair of safety goggles (mandatory for each student)
- 1 pair of rubber gloves (optional, for handling hot mixtures)
Each group needs:
- 30 paperclips (Day 1)
- 1 small saucepan or heat-resistant pot (Days 2 and 4; will be reused for Day 4)
- 1 spatula (Days 2 and 4; for stirring)
- 1 hot plate (Days 2 and 4)
- 2 pieces of aluminum foil or parchment paper (30 cm x 30 cm) (Days 2 and 4)
- 1 ruler or measuring tape (for measuring length, width, area, and shrinkage)
- 1 calculator (for shrinkage calculations)
- For plant-based bioplastics (split between Days 2 and 4):
- 20 mL distilled water
- 1-3 g glycerol
- 3 g cornstarch
- 2 mL of white vinegar
- 2-4 drops food coloring
- For the animal-based bioplastic: (split between Days 2 and 4):
- 480 mL cold water
- 96 g gelatin powder
- 24 g glycerol
- 2-4 drops food coloring
- (optional) 2 drops of essential oil (scent)
- (optional) 1 spoon for removing white foam
For the class to share:
- 1 laptop/tablet with projector and with internet access for YouTube videos and to display the Polymers, Plastics, and Bioplastics Presentation
- extra paper clips in case of loss or breakage
- drying rack or safe area to leave plastic to cool and dry
- 1-2 digital scales
- weigh boats (for ingredients)
- graduated cylinders or beakers (for measuring liquids)
- extra glycerol, cornstarch, white vinegar, gelatin powder, and glycerol (for students to use in their improved bioplastic recipes)
- paper towels
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/uok-3025-polymers-plastic-bioplastics-activity] to print or download.Pre-Req Knowledge
Students should:
- Know the states of matter (solid, liquid, gas).
- Understand basic material properties such as strength and flexibility.
- Know the difference between renewable and nonrenewable resources.
- Be aware of environmental problems caused by plastic waste.
Introduction/Motivation
(Begin by showing the first slide of the Polymers, Plastics, and Bioplastics Presentation and distribute a Polymers, Plastics, and Bioplastics Booklet to each student.)
Polymers, plastics, and bioplastics: What do you think of when you see these words? (Let students discuss for a few minutes.)
We are going to take 5 minutes to fill out Parts 1, 2, and 3 in your booklet. (Start timer on Slide 2 and give students 5 minutes to complete these sections.)
So, what are some things you know about plastics? (Let students offer answers.)
What are some things you wonder about plastics? (Let students offer answers.)
Why do you think plastic is used in so many things? (Answers: It’s light, it’s cheap to make, it’s durable, easy to clean, etc.)
Plastic is very helpful in many ways. However, we also know that plastic is a major environmental problem. We have all seen pictures of plastic floating in the ocean, and we have learned that plastic does not break down quickly like food scraps or cardboard.
You’ve probably heard the phrase “reduce, reuse, recycle.” The challenge with plastic is that very little of it is actually recycled. According to the Environmental Protection Agency (EPA), as of 2018, only about 8.7% of plastic waste in the United States was recycled.
Today we are beginning an activity that will explore the following questions: (Display Slide 3)
- What are polymers, and why are they important?
- What is the difference between natural and synthetic polymers?
- Could bioplastic be a possible solution for some of our plastic usage?
(Display Slide 4.)
Our challenge is this: We are going to think like engineers and design a product that could help reduce plastic waste and improve sustainability.
Let’s get started!
Procedure
Background
Plastics are materials made from synthetic polymers, which are long chains of repeating units called monomers. Most synthetic plastics are derived from natural resources such as petroleum, but the monomers are chemically altered and linked into polymers that would not naturally form. Plastics are engineered to have specific properties—such as being lightweight, flexible, durable, or heat-resistant—depending on how the polymer chains are arranged. This engineered structure makes plastics incredibly versatile, with uses ranging from packaging, bottles, and clothing to electronics and medical equipment.
The characteristics of a polymer are determined by the arrangement of its monomers and the structure of the chain. Short, tightly packed polymer chains create strong, rigid materials, while long, flexible chains produce bendable substances. Understanding this relationship helps students see how the molecular structure of a polymer influences its physical properties.
Synthetic polymers are unique because scientists and engineers deliberately assemble the monomers into chains using heat, catalysts, or other chemical processes. This controlled assembly allows the creation of plastics with very specific characteristics, enabling the wide variety of products we rely on every day.
A major challenge with synthetic plastics is that most cannot naturally break down. Few organisms can digest these polymers, which leads to long-lasting environmental waste. This is why bioplastics—made from renewable plant- or animal-based materials—are becoming increasingly important. Bioplastics can retain many useful properties of traditional plastics while offering the potential for biodegradability, reducing the environmental impact of synthetic materials.
Before the Activity
- Make copies of the Polymers, Plastics, and Bioplastics Booklet and the Polymers, Plastics, and Bioplastics Quiz (1 per student).
- Prepare and review the Polymers, Plastics, and Bioplastics Presentation.
- Gather paper clips and bioplastic materials together.
- Make sure there is a space for the bioplastics to cool where they won’t be disturbed. They will need to dry for 2 days.
During the Activity
Day 1: Introduction and Model Polymers (60 minutes)
- Display the Polymers, Plastics, and Bioplastics Presentation.
- Distribute a Polymers, Plastics, and Bioplastics Booklet to each student.
- Introduce the activity using the Introduction and Motivation section.
- (Slide 5) Introduce polymers by explaining: Plastics and bioplastics are made of polymers! You are going to learn about polymers today, and once you understand them, you’ll start to see polymers everywhere.
- Show the video clip “Polymers: NSF Chalk Talk (Slide 5): https://www.youtube.com/watch?v=z7ict1VLyF4 (1:53 minutes)
- Explain that the reason we are looking at polymers is to better understand plastics.
- Plastics are made of synthetic polymers.
- They are made from a naturally occurring resource, petroleum, but the petroleum has been chemically altered—it has been broken down into monomers and re-formed into polymers to make different types of plastics.
- Instruct students to list or draw some examples of natural polymers and synthetic polymers in their booklet.
- Display Slides 6 and 7 showing examples of natural and synthetic polymers.
- Lead a brief class discussion using the following prompts:
- In what ways have synthetic polymers been helpful? (Potential answer: They make products lighter. Potential examples: We have plastics that allow us to have lighter, stronger seats in airplanes; we have durable parts that can easily be disinfected for hospital use; we have packaging that allows products to travel thousands of miles safely; carpets, clothing etc.)
- How do you think using synthetic polymers has caused issues in our environment? (Potential answers: There are many plastics that cannot be recycled. There are many areas in the world that don’t have recycling programs in place. Recycling plastic takes a great deal of energy, so it is often not a viable option.)
- Group students into pairs.
- (Slide 8) Instruct students to turn to the “Constructing polymer models” section of their booklet.
- Pass out 30 paper clips to each pair.
- Read through the paper clip activity instructions with students to ensure they understand their objective.
- Give students time to complete the paper clip activity.
- They need to design a short chain (10 clips/monomers).
- Once they build their chain, they should pull the chain on both ends and describe and record the result.
- Next, challenge them to change their design by adding more monomers to their chain. (Note: This is where they can try different ways of adding arms or branches to their chain.)
- Have them draw a sketch of their design.
- As partners, have them discuss how the new design affects its flexibility and strength.
- Have the pairs pull on the ends of their chain once again and see how their new design has changed the strength of the polymer chain.
- As pairs work, circulate the room and ask clarifying questions.

- (Slide 9) After the activity is complete, lead a class discussion using the following prompts:
- Let’s share some of your designs with the class and talk about what you did to change the shapes of your polymers. In what ways did the changes affect your polymer?
- How does your polymer chain model real polymer behavior/formation? (Potential answers: Some polymers have branches, ladder shapes, jumbled chains etc.)
- Why is it important for engineers to understand the shapes of polymers? (Potential answers: Engineers can look at how the shape of polymers affects the characteristics of the substance. They can put monomers together to recreate or mimic a desired polymer shape.)
Day 2: Create a Plant-Based or Animal-Based Bioplastic (60 minutes)
- Tie the polymers (both natural and synthetic) to the importance of bioplastics: We’ve learned about polymers, both natural and synthetic. We’ve talked about the issues surrounding the disposal of plastics. Now we are going to look at a possible solution for some of our plastic use. What if you could engineer a type of bioplastic that could be used to replace a synthetic plastic?
- (Slide 10) Introduce bioplastics: Bioplastics are plastics that are made from renewable, plant-based materials and/or are designed to biodegrade more easily than traditional plastics.
- Show the YouTube video on Slide 10: Healthy Oceans: Preventing Plastic Pollution | California Academy of Sciences: https://www.youtube.com/watch?v=RgzEcR_Xc-s (5:27 minutes).
- Give students 5 minutes to complete the Bioplastics Pre-Activity Questions in their booklet.
- (Slide 11) instruct students to take notes in their booklets over the next few slides.
- Go through Slides 12-15. (Note: These slides show the different ingredients in the activity and what they “do” within the substance. Students will fill in the information for the ingredients on their sheet so they can reference it later.)
- Give students 2 minutes to predict which products in the recipe will affect the final product. (Note: They should record their prediction in their booklet.)
- Divide the class into groups of 3-4 students.
- Assign half of the groups to the cornstarch recipe (Recipe #1) and half of the groups to the gelatin recipe (Recipe #2). (Note: This will allow for some extra comparison of results between plant-based and animal-based bioplastics at the very end of the unit.)
- (Slide 16) Instruct students to carefully read through their recipe and instructions. (Optionally, you can play the relaxing classroom music on Slide 16.)
- Review activity safety procedure before beginning activity. Go over hot plate safety and discuss careful handling of the bioplastic when it is poured onto the foil or parchment.
- Give students the rest of the session to follow their booklet instructions to create their plant-based or animal-based bioplastic.

Day 3: Drying Day and Measurements (25-60 minutes)
- Instruct students that this class period will be spent observing their bioplastic piece (which may not be fully dry), filling out information, doing the math to figure out shrinkage, and discussing any questions.
- Have students re-measure their dried bioplastic and record their data in their booklets.
- Instruct students to calculate percent shrinkage using their original area (A) and final dried area (B) using the equation (A-B)/A x 100.
Example: (80 cm - 76 cm)/80 cm x 100 = 4 cm/80 cm x 100 = 0.05 x 100 = 5% shrinkage
- (Slide 17) Have students test for flexibility.
- (Slide 18) Have students test for rigidity.
- Facilitate class discussion about drying, shrinkage, and material behavior.
- Explain that in the next class period, the challenge will be to change the outcome of your bioplastic. For example:
- If a group’s plastic is rigid, they will need to change ONE variable in their recipe to make the new batch flexible (e.g., add more cornstarch.)
- If a group’s plastic is flexible, they will need to change ONE variable in their recipe to make the new batch more rigid.
- Give students time to discuss what variable they will change to get the result they desire.
- Have each group fill out the “Remaking Your Bioplastic!” section with their plan for improvement.
- Optional: If more time needs to be filled, the video about plastics on Slide 10 could be a starting point for further discussion.
Day 4: Bioplastic Improvement Activity (60 minutes)
- (Slide 19) Explain the engineering redesign challenge: Each group will modify one variable to design a more flexible or more rigid bioplastic.
- Keep students in the same groups with the same recipes.
- Instruct groups to choose one ingredient to change and justify their choice.
- Have students follow the same procedure as before but incorporating their variable change.

- Remind students to record ingredient changes and measurements in their booklet in the “Remaking Your Bioplastic!” section.
- When students finish their modified bioplastic, instruct them to move their finished samples to the drying area.
Day 5: Activity Wrap Up (60 minutes)
- Have students retrieve their improved bioplastic samples from the drying area and return to their groups.
- Have students re-measure their dried bioplastic and record their data in their booklets.
- Instruct students to carefully observe their bioplastic pieces, noting changes in flexibility and rigidity, recording their qualitative observations in their Polymers, Plastics, and Bioplastics Booklet.
- Instruct students to calculate the new area (B) of their bioplastic pieces using their measurements.
- Optional: Review the shrinkage formula with the class:
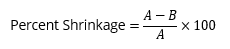
where A is the original area and B is the new area.
- Circulate around the room, checking calculations and providing support as needed.
- Have students complete the “Bioplastic Activity Wrap-Up” questions in their booklet.
- Distribute a Polymers, Plastics, and Bioplastics Quiz to each student.
- Give students time to complete the quiz. Optionally, they can use their booklet to complete the quiz.
- Lead a brief class discussion, prompting students to compare:
- Shrinkage values
- Differences between gelatin and cornstarch plastics
- Observed material changes
- Facilitate a short reflection discussion, using selected wrap-up questions to highlight:
- What ingredients did you use to make your bioplastic?
- What surprised you about your bioplastic?
- Think about your design challenge for the second batch of bioplastic (to make it more rigid or more flexible): If your changes did work, explain what you think you did right. If your changes didn’t work, what do you think you would try differently?
- Would it be possible to replace some of the plastic we use regularly with bioplastic? Why or why not?
- What product(s) could your bioplastic be used for?
Vocabulary/Definitions
biodegradable: Capable of being broken down naturally by microorganisms.
bioplastic: A plastic-like material made from renewable or biodegradable materials.
branching (in polymers): A polymer chain structure in which side chains attach to the main chain.
cross-linking: Chemical bonds that link one polymer chain to another, creating a network.
flexibility: The ability of a material to bend without breaking.
monomer: A single repeating unit that can join with others to form a polymer.
natural polymer: A polymer that occurs in nature.
plastic: A material made from synthetic polymers that can be molded or shaped.
polymer: A large molecule made of repeating units called monomers.
recycling: The process of converting waste materials into new materials or products.
rigidity: The resistance of a material to bending or deformation.
shrinkage: Reduction in size of a material as it dries or cools.
strength: The ability of a material to withstand force without breaking.
sustainability: Using resources in a way that meets present needs without harming the environment.
synthetic polymer: A polymer created by humans through chemical processes.
Assessment
Pre-Activity Assessment
Pre-Activity Questions: Students have 5 minutes to complete Parts 1, 2 and 3 in their Polymers, Plastics, and Bioplastics Booklet. Students fill out these parts on their own before the activity starts to demonstrate their pre-activity knowledge.
Formative Assessment
Observation & Guidance: Circulate during the activity as groups measure, mix, and pour bioplastics. Ask questions such as:
- “What do you predict will happen if you change the amount of glycerin?”
- “How will this ingredient affect flexibility or rigidity?”
Data Recording Checks: After students record their measurements for shrinkage, flexibility, and rigidity, evaluate them for correct calculations and thoughtful analysis of results.
Post-Activity (Summative) Assessment
Bioplastic Activity Wrap-Up Questions: Students reflect on which changes improved or reduced bioplastic performance. They also evaluate whether bioplastics could replace everyday plastics and justify their reasoning.
Quiz: Students take the Polymers, Plastics, and Bioplastics Quiz that measures knowledge, understanding, application, and reflection directly tied to the learning goals of the polymers and bioplastics activity.
Safety Issues
Hot Plate Safety
- Students will use hot plates, which can become extremely hot. The surface of the hot plate and the pan on it will remain very hot even after turning it off.
- Students must handle all hot materials carefully. Have them use oven mitts or tongs when moving the pan.
- When pouring the hot bioplastic onto foil or parchment, remind students that the mixture is very hot, will take several minutes to cool, and can cause burns if it touches skin.
- Emphasize slow, careful pouring and no touching of the hot mixture or pan with bare hands.
Ingredient Safety
- All ingredients are food safe (cornstarch, gelatin, glycerol, vinegar, and food coloring), except for optional scent oils in the gelatin recipe.
- Avoid contact with eyes, wash hands after handling ingredients, and prevent spills.
- Because food coloring can stain skin and surfaces, use aprons if available.
General Activity Safety
- Ensure that safety goggles are always worn.
- Keep hair tied back and sleeves rolled up.
- Work on a protected surface (tray, foil, or parchment).
- Clean up spills immediately to prevent slips or burns.
- Store partially dried bioplastics in a labeled container if continuing the next day.
Troubleshooting Tips
Polymer Chain Modeling (Paper Clips)
- Chains too short or break easily: Ensure that students use the recommended number of clips (10–15 for initial chains) and connect the clips tightly.
- Difficulty visualizing flexibility/rigidity: Compare different chains side by side; discuss how branching or chain length affects strength and flexibility.
Bioplastic Preparation
- Mixture too runny or not setting: Double-check ingredient measurements. Too much water or too little cornstarch/gelatin affects firmness. Stir continuously while heating.
- Mixture too thick or lumpy: Gradually add water while stirring; make sure powders are fully dissolved before pouring.
- Uneven drying: Spread bioplastic in thin, even layers; rotate or flip pieces to dry evenly.
- Unexpected flexibility/rigidity: Encourage careful recording of ingredient amounts; small variations can change properties. Discuss results and compare predictions.
Activity Extensions
Extensions (For Students Ready for More Challenge)
- Ingredient Experiments:
- Have students create bioplastic samples with different ratios of glycerol, cornstarch, gelatin, or vinegar to see how these changes affect flexibility, rigidity, and shrinkage.
- Advanced Material Testing:
- Measure tensile strength, elasticity, transparency, or water resistance of bioplastics.
- Environmental Impact Research:
- Compare synthetic plastics versus bioplastics in recycling, biodegradability, and life-cycle analyses.
Enrichment (Advanced or Multi-Day Activities)
- Chemistry of Plastics:
- Dive deeper into the chemical processes behind polymer formation and plastic production.
- Explore why synthetic plastics are resistant to degradation and why recycling can be difficult.
- Math and Technology Integration:
- Graph shrinkage, flexibility, or degradation data over time.
- Model relationships between polymer structure, ingredient ratios, and material properties.
- Careers in Engineering:
- Investigate roles of materials engineers, chemical engineers, and environmental engineers, connecting activity work to real-world careers in sustainable materials.
Activity Scaling
- Lower Grades / Introductory Version:
- Focus only on the paper clip polymer activity, which provides a hands-on way to visualize molecular structures on a large scale.
- Challenge students to design polymers within specific parameters (e.g., length, strength, flexibility) without moving into the full bioplastic activity.
- Simplified Version for Students with Motor or Learning Challenges:
- Provide pre-measured ingredients or pre-cut polymer chains to reduce fine motor demands.
- Allow students to observe and record results rather than manipulate materials directly.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

The Great Pacific Garbage Patch (GPGP) is an intriguing and publicized environmental problem. Through exploring this complex issue, students gain insight into aspects of chemistry, oceanography, fluids, environmental science, life science and even international policy.

Students explore the chemical identities of polymeric materials frequently used in their everyday lives. They learn how chemical composition affects the physical properties of the materials that they encounter and use frequently, as well as how cross-linking affects the properties of polymeric mater...

Students explore the basic characteristics of polymers through the introduction of two polymer categories: thermoplastics and thermosets. During teacher demos, students observe the unique behaviors of thermoplastics.

Students learn how plastics in the human trash stream end up as microplastic particles entering the food chains via polluted water, harming animals and people. They think of ways to reuse or replace the common plastic items they discard daily. They learn how microplastics persist in the environment ...
References
Baheti, Payal. “How is Plastic Made? A Simple Step-by-Step Explanation.” British Plastics Federation, https://www.bpf.co.uk/plastipedia/how-is-plastic-made.aspx#. Accessed 20 June 2025.
Davis, Clara. BIOPLASTIC recipe. Fab Lab Barcelona. https://class.textile-academy.org/tutorials/BIOPLASTIC-RECIPE.pdf. PDF file. Accessed 20 June 2025.
“Plastic Polymers: The Chemistry Behind Plastics.” Arizona State University Learning Sparks, uploaded by ASU, https://sparks.learning.asu.edu/videos/plastic-polymers-the-chemistry-behind-plastics. Accessed 10 July 2025.
“Science of Plastics” Science History Institute Museum & Library, https://www.sciencehistory.org/education/classroom-activities/role-playing-games/case-of-plastics/science-of-plastics. Accessed 8 July 2025.
Sheridan, Heather. “Hands on Engineering Activity-Making Bioplastic.” All Together, 25 Aug, 2021, https://alltogether.swe.org/2021/08/hands-on-engineering-activity-making-bioplastic.
Copyright
© 2026 by Regents of the University of Colorado; original © 2025 University of KansasContributors
Christena Graves; Emma Sage; Nicholas Gorschak; Dr. Prajna Dhar; Dr. Meagan Patterson; Dr. Massa Mafi; Dr. Douglas Huffman; Esraa Abdallah Abdelsalam; Tarannom SalehiSupporting Program
Inquiry-Driven Engineering Activities using Bioengineering (IDEA-BioE), University of KansasAcknowledgements
This material is based upon work supported by the National Science Foundation under grant no. ECC-2055716 - a Research Experience for Teachers program titled Inquiry-Driven Engineering Activities using Bioengineering (IDEA-BioE) at the University of Kansas. Any opinions, findings and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation.
Last modified: March 7, 2026







User Comments & Tips