Quick Look
Grade Level: 11 (10-12)
Time Required: 1 hours 45 minutes
(can be split into two different sessions)
Lesson Dependency: None
Subject Areas: Biology
Summary
For students interested in studying biomechanical engineering, especially in the field of surgery, this lesson serves as an anatomy and physiology primer of the abdominopelvic cavity. Students are introduced to the abdominopelvic cavity—a region of the body that is the focus of laparoscopic surgery—as well as the benefits and drawbacks of laparoscopic surgery. Understanding the abdominopelvic environment and laparoscopic surgery is critical for biomechanical engineers who design laparoscopic surgical tools.Engineering Connection
Biomechanical engineers work closely with laparoscopic surgeons to develop procedures and tools for surgery. Biomechanical engineers must have an understanding of anatomy, physiology and surgery in order to communicate well with surgeons and other medical professionals. Most surgeons are not trained in mechanical design or product development, nor do they have time to focus on the engineering aspects of new surgical tools; therefore, engineers partner with surgeons to create, design, test and manufacture new tools. This partnership helps advance life-saving and life-enhancing technology such as Laparosopic Surgery tools practiced in the hands on activity Challenges of Laparoscopic Surgery.
Learning Objectives
After this lesson, students should be able to:
- Identify the major cavities in the human body.
- Identify the major organs of the abdominopelvic cavity.
- Describe the anatomy and physiology of the major organs of the abdominopelvic cavity.
- Explain why surgeons try to reduce trauma when performing surgical procedures on organs in the abdominal cavity.
- Explain benefits and drawbacks of laparoscopic surgery (a form of minimally invasive surgery).
- Outline the development of minimally invasive surgery through history.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
International Technology and Engineering Educators Association - Technology
-
Advances and innovations in medical technologies are used to improve healthcare.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
State Standards
Colorado - Science
-
Discuss how two or more body systems interact to promote health for the whole organism
(Grades
9 -
12)
More Details
Do you agree with this alignment?
Worksheets and Attachments
Visit [www.teachengineering.org/lessons/view/cub_surg_lesson01] to print or download.Introduction/Motivation
(Have available the capability to use the attached Human Body Cavities Animated Reference Sheet, a PowerPoint file, to help students identify the locations of human body cavities. If possible show students a three-minute animation of a laparoscopic surgery [see the Additional Multimedia Support section.] Note: In the text below, words in italics may be found in the Vocabulary/Definitions section.)
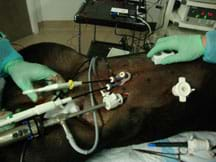
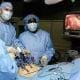
Laparoscopic surgery is a surgical technique in which operations in the abdomen are performed through small incisions in the abdominal or pelvic cavities (see Figure 1). It is a form of "minimally invasive surgery." A trocar creates incisions in the abdomen and provides a sealed conduit for various instruments used to perform the surgery. Typically, three incisions are made during each surgery: one for a laparoscope and dedicated ports for the surgical instruments. The laparoscope consists of a telescopic lens system that is connected to a video camera. The camera is ex vivo. Light is usually provided by a powerful bulb that is piped into the abdominal cavity via a fiber optic or gel cable. The abdomen is insufflated with carbon dioxide gas to create a region large enough to work in and view.
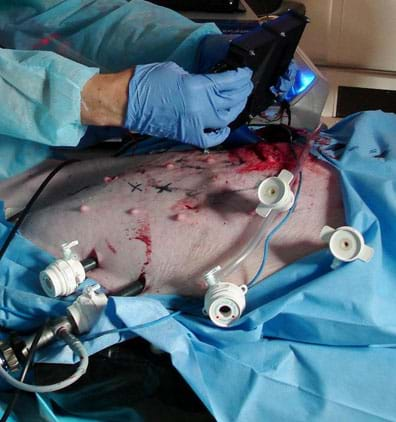
During the past 20 years, this type of surgery has been adopted by many surgical subspecialties and has eliminated the need for open surgery in many cases [1]. Its wide acceptance is due to several advantages. For example, smaller incisions are made with laparoscopic surgery than with laparotomy (open abdominal surgery), which leads to quicker recovery time; in many cases, patients are able to leave the hospital on the day of the surgery. Another advantage compared to traditional surgery is that smaller incisions inflict less pain during recovery, and therefore less pain medication is needed. Finally, internal cavities are not as exposed to the external environment, which reduces the patient's risk of infection.
Despite these advantages, laparoscopy has several drawbacks such as a restricted view of the interior anatomy, difficult handling of the surgical instruments [2], a constrained camera port, lack of tactile feedback to the surgeon, and lack of depth perception.
In traditional laparotomy, members of the surgical team are focused on the patient; that is, they are visually engaged with the real-life, three-dimensional anatomy. In laparoscopy, the view of the interior anatomy is transmitted through a two-dimensional video image, which eliminates the surgeon's depth perception and makes it difficult for him or her to coordinate the surgical instruments [3]. The video image is not usually positioned immediately beside the patient, so the surgeon's attention is not devoted to the patient. The limited field of view from the video presents another challenge to the surgeon. Sometimes it is difficult or impossible to safely operate using the small field of view [4]. In many cases, feasibility cannot be determined until the surgery has commenced; therefore, the surgeon must convert to a conventional open laparotomy. Overall, conversion occurs in 3-5% of operations [5]. The field of view of the abdominal cavity during surgery is limited to the viewing angle of the laparoscope and the scope's range of motion. This limitation makes it difficult to view the internal incision through which the tool is inserted and the immediately surrounding peritoneum. One way to overcome this limitation is to create a second incision and entry port that is removed from the first port and then use a laparoscope inserted into the second port to inspect the first, and vice versa. However, sometimes the surgeon is reluctant to add more ports due to the increased trauma and risk to the patient.
Another drawback to laparoscopic surgery is that it requires the surgeon to manipulate surgical instruments that provide little or no tactile feedback and a limited range of motion, especially for more obese patients. Furthermore, laparoscopic incisions and trocar insertions have occasional complications. Each incision and insertion raises the risk of infection and injury to the delicate internal organs, and each entry site provides a location for ventral herniation after the entry wounds have healed. Significant additional surgical training is required to mitigate these disadvantages, and even then, some complications are unavoidable [2].
Equipment cost is also a drawback of laparoscopic surgery. Equipment and accessories have become more complex and expensive as technology has improved to increase patient comfort and safety, speed recovery time, and expand the surgical technique to more subspecialties. A typical laparoscopic imaging system, for example, costs about US$40,000 [6].
Because every entry port created during laparoscopic surgery increases the risk of complication, the state-of-the-art is continually striving to reduce the invasiveness of surgery [7,8,9]. Surgeons and engineers are presently working on devices and techniques to further limit invasiveness by reducing the number of entry ports. For example, a new laparoscopic technique called single incision laparoscopic surgery (SILS) allows all three trocars to enter the abdomen through the umbilicus. This technique is made possible by innovative, multi-axis instruments that can be triangulated after they have been inserted. The instrument manufacturers (such as Novare, Inc.) claim less post-operation pain, quicker recovery, and better cosmetic results.
We expect the laparoscopic surgical tools of tomorrow to be less-invasive, perhaps entering the abdominopelvic cavity through a natural orifice. They will be smaller and smarter, maybe remotely controlled, semiautonomous, or even fully autonomous. They will be created by engineers who understand the present-day state-of-the-art and have anatomical and physiological knowledge of the body. Through this lesson, you will learn about the abdominopelvic cavity, which is the anatomy associated with laparoscopic surgery.
Lesson Background and Concepts for Teachers
(Unit overview note: To begin the unit, as part of lesson 1, introduce the culminating engineering design challenge of the unit, which is the design project described in the Designing a Robotic Surgical Device activity. After lesson 1, have student teams conduct the Challenges of Laparoscopic Surgery activity as a hook to engage them in the subsequent background lessons on the mechanics of biomaterials [lessons 2, 3, 4 and 5, and their associated activities]. Then, after lesson 5 is presented, conduct the concluding design activity to complete the design challenge.)
Body Cavities
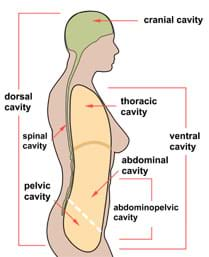
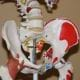
Most of the body's organs are housed in "cavities." A cavity is a protected compartment that insulates, cushions, and protects delicate organs from external shocks. Most cavities are lined by a serous membrane, which is a thin layer of tissue made of epithelial cells. The cells secrete (and absorb) fluid that lubricates the lining of organs to prevent chaffing as tissues slide past one another due to muscle movement. Figure 2 shows the major cavities of the human body. The organs associated with each cavity are listed below:
- cranial: brain
- spinal (or vertebral): spinal cord
- thoracic: heart, lungs, esophagus, trachea
- abdominal: stomach, liver, small and large intestines, spleen, pancreas, gallbladder, kidneys
- pelvic: reproductive organs, rectum, urinary bladder
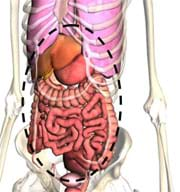
Cavities can contain other cavities, as well as organs. For example, the ventral cavity contains the thoracic and abdominal cavities; the abdominopelvic cavity (see Figure 3) contains the abdominal and pelvic cavities. The cavities themselves are surrounded by bone, muscle, fat, and fascia. The surgeon must cut through multiple layers of tissue to reach organs located in cavities. Surgical trauma caused to the patient is a function of an incision's size, so surgeons and biomechanical engineers who design surgical tools are continually developing new techniques and tools to minimize the size and number of the incision needed to access and operate on organs within cavities. Surgery that reduces trauma to the patient, perhaps at the expense of time and cost, is called "minimally invasive surgery." For more information on the history of minimally invasive surgery, see the additional background information provided in the Other section of this document.
Organs in the Abdominopelvic Cavity
Typically, surgeons specialize and become experts with one cavity and its respective organs. In this lesson, we focus on laparoscopy and the abdominopelvic cavity. See the Abdominal Cavity Anatomy & Physiology Worksheet Answers for diagrams and a list of the organs of the abdominopelvic cavity of a human female, as well as an anatomical description and brief physiology of each organ. The primary (human female) organs are: esophagus, kidney, liver, stomach, large intestine, small intestine, spleen, gallbladder, pancreas, ovary, uterus and bladder.
Associated Activities
- Challenges of Laparoscopic Surgery - Student teams use a surgical trainer device to perform simple laparoscopic surgery tasks (dissection, suturing) using laparoscopic tools. Just like in the operating room, where the purpose is to perform surgery carefully and quickly, students' surgery time and mistakes are observed and recorded to quantify their performances.
Vocabulary/Definitions
ex vivo: Signifying outside the living body.
fascia: Tough, fibrous connective tissue.
in vitro: Signifying in a laboratory or controlled environment.
in vivo: Signifying inside the living body.
insufflation: Pressurizing or filling with air. The abdominal cavity is insufflated prior to surgery to create a space for the surgeon to work in.
laparoscope: A camera attached to a long, slender light pipe that allows a laparoscopic surgeon to view inside the abdominal cavity without invasive surgery.
laparotomy: Open surgery on the abdominal cavity (surgery through a large incision).
peritoneum: Membrane that lines the inside wall of the abdominal cavity.
trocar: A specialized surgical knife used for creating small incisions for the introduction of a surgical tool through the abdominal wall.
umbilicus: Belly button.
ventral herniation: When an organ of the abdominal cavity pushes through the abdominal wall, typically at an incision site.
Assessment
Worksheet: Have students use the attached Abdominal Cavity Anatomy &Physiology Worksheet to search online or at the library for names and descriptions of the organs in the abdominopelvic cavity. Review their answers to gauge their mastery of the subject matter.
Additional Multimedia Support
Show students a 3:19-minute computer-generated video of a laparoscopic appendectomy surgery for appendicitis by Nucleus Medical Art on YouTube at: https://www.youtube.com/watch?v=MAppEy9Umcg
Watch a 5:18-minute video showing the daVinci® surgical robot on YouTube at: https://www.youtube.com/watch?v=Zb01Ep9KuLA
Watch a 1:07-minute video animation showing a single-port laparoscopy using Novare RealHand® surgical tools at YouTube at: https://www.youtube.com/watch?v=OU1F35b5f3c
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students teams use a laparoscopic surgical trainer to perform simple laparoscopic surgery tasks (dissections, sutures) using laparoscopic tools. Just like in the operating room, where the purpose is to perform surgery carefully and quickly to minimize patient trauma, students' surgery time and mista...

Student teams create laparoscopic surgical robots designed to reduce the invasiveness of diagnosing endometriosis and investigate how the disease forms and spreads. Using a synthetic abdominal cavity simulator, students test and iterate their remotely controlled, camera-toting prototype devices, whi...

Students learn about the sorts of devices designed by biomedical engineers and the many other engineering specialties that are required in their design of medical diagnostics, therapeutic aids, surgical devices and procedures, and replacement parts. They discuss the special considerations that must ...

Students act as surgical residents for the day. Working in teams, they use surgical instruments to complete tasks that are inside of a box, hidden from direct view. They are able to see inside of the box with the help of a "laparoscope" (webcam and flashlight).
References
[1] A. Gordon and A. Magos, "The development of laparoscopic surgery," Baillire's Clinical Obstetrics and Gynaecology, vol. 3, no. 3, pp. 429-449, September 1989.
[2] B.J. Feder, "Surgical Device Poses a Rare but Serious Peril," The New York Times, March 17, 2006.
[3] C. Muratore, B. Ryder, and F. Luks, "Image display in endoscopic surgery," Journal of the Society for Information Display, vol. 15, no. 6, pp. 349-356, June 2007.
[4] N.B. Halpern, "The Difficult Laparoscopy," Surgical Clinics of North America, vol. 76, no. 3, pp. 603-613, June 1996.
[5] A. Lorenzo, M. L. Samuelson, S.G. Docimo, L.A. Baker, and Y. Lotan, "Cost analysis of laparoscopic versus open orchiopexy in the management of unilateral nonplpable testicles," The Journal of Urology, vol. 172, no. 2, pp. 712-716, August 2004.
[6] Benjamin S. Terry. Based on a price quote from Stryker on October 13, 2009: laparoscope: $3,600, light source: $7,500, light cord: $295.46, camera box: $11,500, camera head: $12,000, display: $6,247.50, Total: $41,142.96.
[7] M. Rentschler, S. Farritor, and K. Lagnemma, "Mechanical design of robotic in vivo wheeled mobility," Journal of Mechanical Design, vol. 129, no. 10, pp. 1037-1045, October 2007.
[8] Tie Hu, P. Allen, and D. Fowler, "In-vivo pan/tilt endoscope with integrated light source," in Intelligent Robots and Systems, 2007. IROS 2007. IEEE/RSJ International Conference, pp. 1284-1289, 2007.
[9] A. Gumbs, F. Crovari, C. Vidal, P. Henri, and B. Gayet, "Modified robotic lightweight endoscope (ViKY) validation in vivo in a porcine model," Surgical Innovation, vol. 14, no. 4, pp. 261-264, December 2007.
[10] M. J. O'Dowd and E. E. Philipp, The History of Obstetrics and Gynaecology, 2000.
[11] A. K. Saxena, "History of Endoscopic Surgery," Essentials of Pediatric Endoscopic Surgery, 2008. Accessed November 18, 2008. http://dx.doi.org/10.1007/978-3-540-78387-9_1
[12] Fritz Spiegl, Fritz Spiegl's Sick Notes. New York, NY: Parthenon, 1996.
[13] T. Barnett and South-Western Obstetrical and Gynaecological Society, The obstetrician, anaesthetist and the paediatrician in the management of obstetric problems: transactions of the South-Western Obstetrical and Gynaecological Society's autumn meeting, New York, NY: Macmillan, 1963.
[14] D. M. Harland, Water and the search for life on Mars. Berlin; New York; Chichester, UK: Springer; In association with Praxis Publishing, 2005.
[15] W. H. Woglom, Discoverers for Medicine. 1949.
[16] W. C. Campbell and A. H. Crenshaw, Campbell's operative orthopaedics. St. Louis, MO: Mosby, 1987.
[17] A. K. Saxena and M. E. Höllwarth, Essentials of pediatric endoscopic surgery. Berlin; London: Springer, 2008.
[18] J. Gillenwater, Adult and pediatric urology, 3rd edition, St. Louis, MO: Mosby Year Book, 1996.
[19] N. F. Phillips, E. C. Berry, and M. L. Kohn, Berry & Kohn's operating room technique. St. Louis, MO: Mosby, 2004.
[20] C. J. G. Sutton and M. P. Diamond, Endoscopic surgery for gynaecologists. London; Philadelphia: W.B. Saunders, 1993.
[21] C. A. Dickman, D. J. Rosenthal, and N. I. Perin, Thoracoscopic spine surgery. New York, NY: Thieme, 1999.
[22] B. E. Jarrell and R. A. Carabasi, NMS Surgery. Philadelphia; London: Wolters Kluwer/Lippincott Williams & Wilkins, 2008.
[23] P. Clavien and J. Baillie, Diseases of the gallbladder and bile ducts : diagnosis and treatment. Malden, MA: Blackwell Publishing, 2006.
[24] D. Ruan, "Applied computational intelligence," Proceedings, 6th International FLINS Conference, Blankenberge, Belgium, September 1-3, 2004. Singapore; Hong Kong: World Scientific, 2004.
Other Related Information
Additional Background Information
For the interested reader, the following is a brief history of minimally invasive laparoscopic surgery with references to support material.
As long as physicians have practiced medicine, people have been curious about the anatomical nature of the human body and its physiology. The earliest recorded references of minimally invasive surgery (MIS) are from Hippocrates around 400 BC. His writings suggest that he was well aware of life-threatening ileus (bowel obstruction) and explains several minimally invasive techniques for examination and treatment. For example, Hippocrates explains the procedure for rectal examination with a speculum (a tool for examining body cavities) [10]. The first significant advancement in viewing the interior of the human body did not come until 1585 when Julius Caesar Aranzi, a leading anatomist, was the first to use a light source for an endoscopic procedure, focusing sunlight through a flask of water and projecting the light into the nasal cavity [11]. In 1706 the term "trocar" was first used to describe a three-faced instrument consisting of a knife-like instrument enclosed in a metal cannula [12]. Later, in 1762, William Smellie of London developed an endoscopic-like procedure for treating asphyxia in newborns by inserting catheters into the respiratory track [13]. A turning point in endoscopic surgery occurred in 1729 with the first documented use of lenses to redirect light [14]. In this year, British physician Archibald Cleland invented a biconvex lens illumination device that was placed in front of a candle in order to redirect light into the nasal cavity [15]. This was a great improvement from using flasks filled with water to redirect and magnify light.
Laparoscopic procedures were much slower to develop than other endoscopic specialties. It was treatment of the kidney, bladder, and gallbladder stones with the use of abdominally placed trocars that accelerated the development of laparoscopic surgery. One advantage to using a cannula and trocar was to keep the bladder empty, so some of the rationale for using laparoscopic devices was not even for providing surgical access. Another breakthrough was achieved in 1805 when Philip Bozzini made the first observations of a human body directly through a tube he called a "lichtleiter" (light guiding instrument). His first examinations were of the urinary tract, rectum and pharynx [16]. In 1853, Antoine Jean Desormeaux of France built upon Bozzini's inventions and developed an instrument to examine the urinary tract and the bladder. He called his instrument "endoscope" [17]. Finally, in 1901 the first laparoscopy was performed by a German surgeon named Georg Kelling, who insufflated the abdomen of a dog and then examined the interior with a scope [18]. Important accessories to trocars, cannulas, and endoscopes came along shortly thereafter. For example, in 1938, Janos Veress of Hungary developed a spring-loaded needle to create the pneumo-peritoneum. His basic design is still in use today [19].
By 1970, gynecologists had thoroughly incorporated laparoscopy into their practices [20,21]. General surgeons, however, were slow to give up the practice of traditional open surgery in favor of laparoscopic techniques. These walls rapidly came down after the first laparoscopic assisted appendectomy was performed by Dekok in 1977 [22]. In 1985, the first laparoscopic surgical removal of the gallbladder was performed by Erich Mühe in Germany [23]. With the tremendous growth of laparoscopy by the late 1980s, new specialties such as thoracoscopy in cardiothoracic surgery came into existence. In 1994, a robotic arm was designed to hold the laparoscope with the goal of improving safety and reducing the need for a skilled camera operator. And in 1996, the first live telecast of laparoscopic surgery performed remotely via the Internet gave birth to robotic telesurgery [24]. This brings us to the modern era of laparoscopic surgery.
Copyright
© 2011 by Regents of the University of Colorado.Contributors
Benjamin S. Terry, Brandi N. Briggs, Stephanie Rivale, Denise W. CarlsonSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
This digital library content was developed by the Integrated Teaching and Learning Program under National Science Foundation GK-12 grant no. DGE 0338326. However, these contents do not necessarily represent the policies of the National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: February 25, 2020


User Comments & Tips