Quick Look
Grade Level: 6 (5-7)
Time Required: 30 minutes
Expendable Cost/Group: US $0.00
Group Size: 1
Activity Dependency: None
Subject Areas: Earth and Space

Summary
In this activity, students use a tutorial on the U.S. Environmental Protection Agency's website to learn about how surface water is treated to make it safe to drink.Engineering Connection
Every community's source of water is unique since it may come from rivers, lakes, aquifers, snowmelt or some combination. Engineers design unique treatment plants, depending on the quality of the water that enters the plant, to remove the harmful visible and invisible particles from our drinking water.
Learning Objectives
After this activity, students should be able to:
- Describe technologies developed by engineers for drinking water treatment.
- Assess the importance of each process used to treat water.
- Identify which process takes place at each point in a water treatment facility.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
-
All human activity draws on natural resources and has both short and long-term consequences, positive as well as negative, for the health of people and the natural environment.
(Grades 6 - 8)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Students will develop an understanding of the effects of technology on the environment.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Differentiate between inputs, processes, outputs, and feedback in technological systems.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
State Standards
Colorado - Science
-
Identify problems, and propose solutions related to water quality, circulation, and distribution – both locally and worldwide
(Grade
6)
More Details
Do you agree with this alignment?
Materials List
For each student:
- Computer with Internet access to https://www3.epa.gov/safewater/kids/index.html
- One copy of the Water Treatment Path Worksheet per student.
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/cub_enveng_lesson06_activity2] to print or download.Introduction/Motivation
Where does water originate? Water may be treated differently in different communities depending on the quality of the original water that enters the plant. Groundwater is water located underground and typically requires less treatment than water from lakes, rivers and streams. Why do you think this is? Engineers design complex water treatment facilities to get all of the visible and invisible particles out of our drinking water. Some of these treatments include coagulation (adding a powdery chemical, called a coagulant to the water), and flocculation (a mixing process that makes particles stick together in clumps called flocs), sedimentation (the settling process that removes most of the big particles from the water), and disinfection (the removal of harmful microorganisms from the water). Disinfection is often performed by adding chlorine to the water to kill germs; this method is specifically called chlorination. After all this, it is finally clean enough to drink! To be sure that the water is clean enough, environmental engineers will test the water for different chemicals of concern before distributing the water to people for drinking! In the US, the Environmental Protection Agency (EPA) sets limits on the maximum allowable concentration of a particular chemical in drinking water, based on the toxicity of each chemical. You can learn more about the limits for different chemicals on this EPA website: https://www.epa.gov/laws-regulations.
In this activity, we will follow a drop of water from its source through the treatment process on the computer.
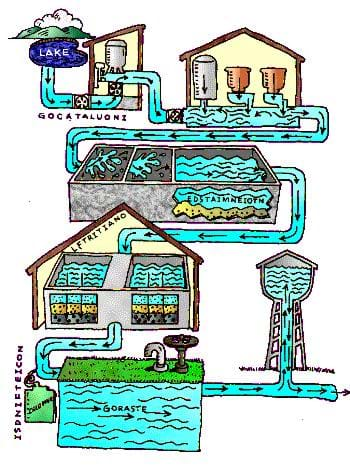
Procedure
- Figure 1 contains five scrambled words. Try to unscramble the words, which label each treatment point. You can check your answers and learn about the processes at https://www3.epa.gov/safewater/kids/index.html.
- Complete the Water Treatment Path Worksheet.
Vocabulary/Definitions
chlorination: To treat with chlorine for disinfection.
coagulation: A chemical process that makes particles in water stick together.
contamination: Something that soils, stains, corrupts or infects by contact or association.
disinfection: To free from infection especially by destroying harmful microorganisms.
flocculation: A gentle stirring process by which particles stick together into clumps, called flocs.
sedimentation: The process of allowing water to sit so that particles fall to the bottom of the container and then removed.
Assessment
Pre-Activity Assessment
Vocabulary: Ask the students to write down the following vocabulary words and definitions on a sheet of paper or in their science journals: coagulation, sedimentation, filtration, chlorination, and disinfection.
Ask students how environmental engineers can be sure that the water is safe enough to drink after the disinfection step? (Answer: Engineers will take a sample of effluent water (water coming out of the treatment plant) and test it for different chemicals of concern.)
Activity Embedded Assessment
Worksheet: Have the students complete the Water Treatment Path Worksheet; review their answers to gauge their mastery of the subject.
Post-Activity Assessment
Discussion: Ask the students where in the water treatment plants can an environmental engineer deal with a situation where water is contaminated with pathogens. Have students point out the different points in the plant where viruses and bacteria can be removed (i.e., coagulation, chlorination).
Discussion: If we think about the design water treatment plants as an engineering design problem, what is the goal or purpose of designing a water treatment plant? (Answer: To treat our water for drinking!) How do engineers know that their design is working well enough? In other words, what are some criteria or constraints for this design problem? (Answers: There are many possible answers here, but one key idea is that the water has to be treated until it is safe enough to drink, and the effluent water can be tested to make sure it is safe.) What are some consequences for people if the water treatment plant does not meet the design criteria? In other words, what happens if the water is not treated enough to be safe for drinking? (Answer: People could get sick!)
Troubleshooting Tips
If students are not used to working with a computer, they should partner with a student who does know how to use the Internet. Students should be supervised when they are working on computers to ensure they stay on task.
Activity Extensions
Students can participate in other water games and online activities at https://www3.epa.gov/safewater/kids/kids_4-8.html
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn about the various methods developed by environmental engineers for treating drinking water in the United States.

Students learn about water quality testing and basic water treatment processes and technology options. Biological, physical and chemical treatment processes are addressed, as well as physical and biological water quality testing, including testing for bacteria such as E. coli.
References
U.S. Environmental Protection Agency, Drinking Water and Groundwater Kids Stuff. https://www3.epa.gov/safewater/kids/kids_4-8.html
Copyright
© 2005 by Regents of the University of ColoradoContributors
Sharon D. Perez-Suarez; Ben Heavner; Malinda Schaefer Zarske; Janet YowellSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under a grant from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and National Science Foundation GK-12 grant no. 0338326. However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: October 1, 2021



User Comments & Tips