
Summary
Contamination in drinking water sources or watersheds can negatively affect the organisms that come in contact with it. The affects can be severe — causing illness or, in some cases, even death. It is important for people to understand how they can contribute to the contaminants in drinking water and what treatment can be done to counter these harmful effects. Students learn about the various methods developed by environmental engineers for treating drinking water in the United States.Engineering Connection
Engineers design many chemical and filter systems to make water safe for drinking and bathing. Some of the most important services provided in a community are the water and sewage treatment plants designed by engineers. Engineers also design treatment processes for the unclean water from paper processing plants or other industrial factories. Water must be clean when it leaves the factory, or else it will contaminate soil, water and air even further.
Learning Objectives
After this lesson, students should be able to:
- Identify several processes used to treat water to make it safe to drink.
- Use vocabulary to help explain drinking water treatment.
- Describe how engineers use technology to help solve an individual or community drinking water problem.
- Describe contributions to the advancement of drinking water treatment made by people in different times in history.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ESS3-3. Apply scientific principles to design a method for monitoring and minimizing a human impact on the environment. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This lesson focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Apply scientific principles to design an object, tool, process or system. Alignment agreement: | Human activities have significantly altered the biosphere, sometimes damaging or destroying natural habitats and causing the extinction of other species. But changes to Earth's environments can have different impacts (negative and positive) for different living things. Alignment agreement: | Relationships can be classified as causal or correlational, and correlation does not necessarily imply causation. Alignment agreement: The uses of technologies and any limitations on their use are driven by individual or societal needs, desires, and values; by the findings of scientific research; and by differences in such factors as climate, natural resources, and economic conditions. Thus technology use varies from region to region and over time.Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ESS3-4. Construct an argument supported by evidence for how increases in human population and per-capita consumption of natural resources impact Earth's systems. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This lesson focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Construct an oral and written argument supported by empirical evidence and scientific reasoning to support or refute an explanation or a model for a phenomenon or a solution to a problem. Alignment agreement: | Typically as human populations and per-capita consumption of natural resources increase, so do the negative impacts on Earth unless the activities and technologies involved are engineered otherwise. Alignment agreement: | Cause and effect relationships may be used to predict phenomena in natural or designed systems. Alignment agreement: All human activity draws on natural resources and has both short and long-term consequences, positive as well as negative, for the health of people and the natural environment.Alignment agreement: Scientific knowledge can describe the consequences of actions but does not necessarily prescribe the decisions that society takes.Alignment agreement: |
International Technology and Engineering Educators Association - Technology
-
The management of waste produced by technological systems is an important societal issue.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
-
Analyze how the creation and use of technologies consumes renewable and non-renewable resources and creates waste.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
State Standards
Colorado - Science
-
Interpret and analyze data about changes in environmental conditions – such as climate change – and populations that support a claim describing why a specific population might be increasing or decreasing
(Grade
6)
More Details
Do you agree with this alignment?
-
Identify problems, and propose solutions related to water quality, circulation, and distribution – both locally and worldwide
(Grade
6)
More Details
Do you agree with this alignment?
Worksheets and Attachments
Visit [www.teachengineering.org/lessons/view/cub_enveng_lesson06] to print or download.Introduction/Motivation
Show the students a glass of muddy water. Can you hypothesize where it came from? Does anyone know what is in the water? Would you want to drink this if it came from your kitchen tap? Probably not. People have been trying to get water clean enough to drink for centuries. During the 1700s and 1800s, people developed ways to filter water to make it clean. They also discovered that there were microscopic bacteria and diseases that can travel in water that can make us sick. Today, environmental engineers have the job of cleaning up water to make it drinkable for communities. Refer to the associated actvity From Lake to Tap to give students a perspective of where their water comes!
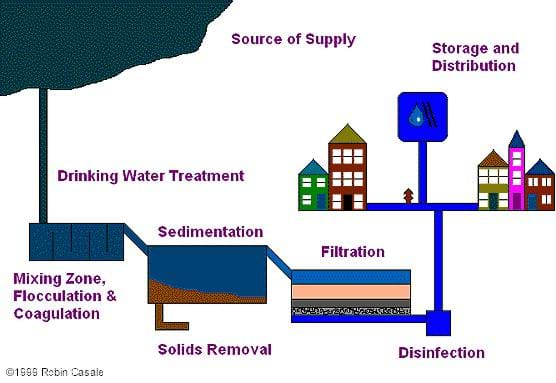
Environmental engineers are concerned about making drinking water safe for people to drink. They design the treatment facilities for drinking water processes, and they are responsible for getting rid of the contaminants and bacteria in untreated water that may make people sick. There are several processes that they use, including: coagulation, in which a powdery chemical, called a coagulant, is mixed in to the water to neutralize charges between particles in the water, enabling the particles to combine during flocculation, a slow mixing process that makes particles stick together into clumps called flocs. After the flocs begin to form, the water flows into a large basin so that the big clumps can settle to the bottom, where they are removed. Sedimentation is the settling process that removes most of the big particles from the water, but there may still be some small particles left in the water. To remove these remaining particles, the water flows through a filter. Lastly, engineers in the United States also disinfect the water. Disinfection is often performed by adding chlorine to the water to kill germs. After all these steps, it is now clean enough to drink! Refer to the associated activity Straining out the Dirt for students to create their own filter using simple materials.
In addition to designing ways to treat water, environmental engineers are called on when water quality in a drinking water facility changes or the design/machinery is not cleaning the water properly. These engineers figure out how to fix the problem and clean up the contaminated water. Today, we are going to work to clean up this glass of muddy water enough to drink it.
Lesson Background and Concepts for Teachers
Water engineering is a large and diverse branch of civil and environmental engineering. In one way or another, civil engineers are involved in almost every part of the water cycle – from the time water falls as rain until it is returned to the sea. Water engineers design and help build dams to create water storage reservoirs, use hydropower to generate electricity, divert water through tunnels, canals and pipelines to provide irrigation, transport water to cities, and supply safe drinking water to homes and industries http://www.ice.org.uk/ ().
People have been treating water to make it safe to drink for a long time. Originally, people thought water was safe to drink if it was clear, so they worked to remove cloudiness ( turbidity). Early Egyptian paintings show that ancient Egyptians filtered their water, and it is believed that they used alum, a chemical that is still used for water treatment today. During the 1700s and 1800s, filtration was developed to remove the suspended particles and clear up the water. In 1855, John Snow proved cholera is a waterborne disease caused by sewage; and in the 1880s, Louis Pasteur further described the ability of microscopic bacteria to travel through water. Federal regulation of drinking water in the United States started in the early 1900s, but regulation did not become strict until the Safe Water Drinking Act of 1974. This law underwent changes in 1986 and 1996 and is now governed mostly by the United States Environmental Protection Agency (US EPA). (For more on the history of drinking water, see http://www.epa.gov/safewater/dwh/treat.html under the "History of Drinking Water Treatment".)
Today, the EPA is responsible for creating standards in the United States for water that is supplied to people to drink. Some people that use their own water wells are using water not regulated by the EPA. Most states are responsible for making sure that water meets the drinking water standards set by the EPA, but some states have chosen to let the EPA keep that responsibility. The Federal and state agencies can take local water distributors to court for not meeting the EPA standards.
How is Drinking Water Treated?
The largest cities in the United States get their water from surface water – lakes, rivers and reservoirs. Water taken from any surface source usually has some amount of debris (i.e., sticks, leaves, dirt) in it. It can also contain small amounts of pathogens (substances that can cause disease). Untreated water, called "raw water" is sent to a drinking water treatment facility to make it safe to drink, or potable. At the drinking water treatment facility, the water flows through many different processes to remove any contaminants.
The first step in treating surface water is a little bit strange: we add a powdery chemical, called a coagulant, to the water. Although it may seem strange to add powder to water to make it clean, coagulants actually work very well. Coagulants make the particles in the water stick together in clumps. The coagulant is mixed into the water very quickly so that it gets evenly spread around. Next, the water is gently stirred so that the particles bump into each other and stick together. When particles stick together, they grow into clumps called flocs, so this gentle mixing process is called flocculation. After the flocs begin to form, the water flows into a large basin so that the big clumps can settle to the bottom, where they are removed. This settling process, called sedimentation, removes most of the big particles from the water, but there may still be some small particles left in the water. To remove these remaining particles, the water flows through a filter. In most water treatment facilities, filters are made of a special kind of sand, and the filters actually look like big sandboxes. After flocculation, coagulation, sedimentation and filtration, most of the contaminants are removed from the water. However, since everyone needs to drink water, it is wise to be extra safe, so engineers in the United States disinfect the water. Disinfection is often performed by adding chlorine to the water to kill germs. Some water treatment facilities also use other processes to treat water, such as using carbon to remove chemicals in the water, or ultraviolet light for disinfection. Many communities get water from a well in the ground. Ground water-supplied systems bypass some of the steps listed above because the passage of groundwater through soil in pores of the soil acts as a natural filtration system. Different treatment systems use slightly different processes because water from different sources might contain different kinds or amounts of contamination.
Everyone has to drink water, so it makes sense to think about where drinking water comes from and how we can keep it safe. Citizens can get involved in protecting their water resources by voting on land usage proposed by the local government and agencies as well as inform their local water suppliers of changes in the water quality or suspected contamination in the community.
Associated Activities
- Straining out the Dirt - In this activity, students build their very own water filter from simple materials.
- From Lake to Tap - This is a computer activity where you follow a drop of water from the source through the water treatment process.
Lesson Closure
Discuss with the students issues related to conservation of water in our country and in developing countries. Can they think of why water conservation is an issue? Ask students if they can think of anything they could do to help keep our water resources clean. Encourage the students to conserve water and discuss why it is important. (Expected answer: Everyone has to drink water, so it makes sense for everyone to think about where drinking water comes from, and how we can keep it safe.)
Vocabulary/Definitions
chlorinate: To treat with chlorine for disinfection.
coagulant: A chemical that makes particles in water stick together.
contaminant: Something that soils, stains, corrupts or infects by contact or association.
disinfection: To free from infection especially by destroying harmful microorganisms.
floc: A clump of particles in water created by flocculation.
flocculation: A gentle stirring process by which particles stick together and grow into flocs.
potable water: Water that is safe for people to drink.
raw water: Water that has not been treated.
sedimentation: The process of allowing water to sit so that particles drop to the bottom of the container and are removed.
turbidity: Cloudiness of water.
Assessment
Pre-Lesson Assessment
Discussion Questions: Ask the students and discuss as a class.
- Have you ever thought about the source for drinking water? Would you drink water out of the gutter next to the road? Why not?
- Would you want to drink muddy water? Can you think of any ways to make muddy water clean? Would your method work if you wanted to make a lot of muddy water clean (like a million gallons or more)?
- Do you think that large cities have challenges in treating water? (Answer: Yes, with a higher population, they have more volume of water to treat. Also, if there are industries which produce bad chemicals, they may be very difficult to treat (i.e. heavy metals, pharmaceuticals).
- True or False: People in Las Vegas, NV consume more water than people in any other city in the US? (Answer: Yes! Because it is naturally a desert, and there is much landscaping, it is a difficult place to maintain. This means lots of clean water!)
Post-Introduction Assessment
Voting: Ask a true/false question and have students vote by holding thumbs up for true and thumbs down for false. Count the votes and write the totals on the board. Give the right answer.
- True or False: Ancient Egyptians treated their water to make it safe to drink. (Answer: true)
- True or False: Bacteria in water often make people sick in the United States. (Answer: False; engineers have designed systems to treat the water that most people drink.)
- True or False: One way to remove particles from water is to let it sit for a long time. (Answer: True; the particles will settle at the bottom — which is how sedimentation works.)
- True or False: The first step in water treatment is to add powder to the water. (Answer: True; a powdery chemical, a coagulant, is added to help with flocculation.)
- True or False: Filtering and disinfection are not important steps in drinking water treatment. (Answer: False; filtering and disinfection get rid of the smallest particles and bacteria from our drinking water.)
- True or False: Environmental engineers have the job of cleaning up water to make it drinkable for communities. (Answer: true)
- True or False: Environmental engineers only work with new drinking water treatment facilities. (Answer: False; environmental engineers are called on when water quality in an existing drinking water facility changes or the design/machinery is not cleaning the water properly.)
- True or False: Texas and California consume the most water out of any other states. (Answer: True. Due to high populations and dry climates, these places use incredible amounts of water.)
Lesson Summary Assessment
Vocabulary Mix-Up: Write either the term or the definition of the ten vocabulary words on slips of paper. Mix them up, and then divide the slips among the students. Ask each student to match his or her term with its definition (or vice versa). After students have found their partner, ask each pair to read their term and definition out loud. At the end, give a brief explanation of the concepts.
Concept Reflections / Journal Writing: Have the students reflect on the drinking water pollution and write a journal entry on their thoughts.
Roundtable: Have the class form into teams of 3–5 students each. Ask the class a question with several possible answers. Have the students on each team make a list of answers by taking turns writing down ideas on a piece of paper. Students pass the list around the group until all ideas are exhausted. Have teams read aloud the answers and write them on the board. Ask the students:
- How would you remove or minimize pollutants in drinking water? (Answer: The first step in treating surface water is to add a powdery chemical, called a coagulant, to the water. Coagulants make the particles in the water stick together in clumps. When particles stick together, they grow into clumps called flocs, so this gentle mixing process is called flocculation. After the flocs begin to form, the water flows into a large basin so that the big clumps can settle to the bottom, where they are removed. This settling process, called sedimentation, removes most of the big particles from the water. To remove any of the remaining smaller particles, the water flows through a filter, made of a special kind of sand. After flocculation, coagulation, sedimentation and filtration, most of the contaminants are removed from the water. However, since everyone needs to drink water, disinfection is often performed by adding chlorine to the water to kill any remaining germs.)
Friction Boggle!: Repeat the same activity as above, except when the teams read aloud their answers and write them on the board, ask if any other teams came up with the same idea. If any other teams have the same answer on their sheet, they have to cross that answer out on their list. The team that ends up with the most "unique" ideas, wins.
Worksheet: Have the students complete the attached You Are What You Drink Worksheet, which are focused on connecting human population to water use.
Lesson Extension Activities
Take a tour of your local drinking water treatment facility, or ask someone who works there to visit your class to explain how it works.
Internet search: How do people in other countries treat their water? What are some inexpensive ways to make water safe to drink (examples: http://www.sodis.ch/index , http://www3.potpaz.org/)
Cleaning water with dirt? Have students investigate an experiment using soil to learn how dirt can work as a filter: http://soil.gsfc.nasa.gov/ .
There are great water posters that can be ordered (free of charge) from the U. S. Geological Survey at: http://water.usgs.gov/outreach/OutReach.html . Have students order a variety of posters for their classroom or the school.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn about water quality testing and basic water treatment processes and technology options. Biological, physical and chemical treatment processes are addressed, as well as physical and biological water quality testing, including testing for bacteria such as E. coli.

In this activity, students use a tutorial on the U.S. Environmental Protection Agency's website to learn about how surface water is treated to make it safe to drink.

Students learn about the differences between water source types (surface and ground) as well as the differences between streams, rivers and lakes. Then, they learn about dissolved organic matter (DOM) and the role it plays in identifying drinking water sources.

Students groups build water filters using activated carbon, cotton and other materials to clean a "dirty" water sample made from chocolate powder added to tap water. They test and make observations of filter effectiveness, suggesting material and layering design improvements.
References
The Institute of Civil Engineers, Education Zone, "What Do Civil Engineers Do?" www.ice.org.uk/
New Jersey American Water Works Association, Kids' Water Zone, "Water Treatment and Distribution to Your Home," www.njawwa.org/kidsweb/treatment/treatment_distrib_to_home.htm Accessed November 7, 2005
U.S. Environmental Protection Agency, Ground Water & Drinking Water, "How is drinking water treated?" water.epa.gov/drink/info/index.cfm Accessed November 7, 2005
U.S. Environmental Protection Agency, Ground Water & Drinking Water, "Drinking Water and Health: What you need to know," water.epa.gov/drink/index.cfm Accessed November 7, 2005
U.S. Environmental Protection Agency, Ground Water & Drinking Water, "Drinking Water & Ground Water Kids' Stuff," http://www.epa.gov/
Copyright
© 2005 by Regents of the University of Colorado.Contributors
Ben Heavner; Sharon D. Perez-Suarez; Malinda Schaefer Zarske; Janet YowellSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under a grant from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and National Science Foundation GK-12 grant no. 0338326. However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: July 3, 2019






User Comments & Tips