
Summary
Students groups build water filters using activated carbon, cotton and other materials to clean a "dirty" water sample made from chocolate powder added to tap water. They test and make observations of filter effectiveness, suggesting material and layering design improvements. They see how their filtering process compares to the drinking water treatment processes designed by engineers to provide communities with clean water supplies after natural disasters.Engineering Connection
Environmental and civil engineers design systems to process communities' water supplies to make them clean enough for people to drink. Depending on the starting water quality, most municipal water treatment systems involve many steps to eliminate the contaminants that could make people sick. Often, the treatment process includes the use of filters of sand and activated carbon to remove particulate matter and certain bacteria just before the water is disinfected with chlorine. Engineers also design water filtration devices to help communities affected by natural disasters.
Learning Objectives
After this activity, students should be able to:
- Design and build a model filter and compare it to processes used by engineers in drinking water treatment.
- Test and make observations on filter effectiveness.
- Use data from observations to construct a reasonable explanation of drinking water treatment through a filter.
- Describe how drinking water filters can help improve the drinking water quality of a community.
- Explain how drinking water filters can be used in the aftermath of a natural disaster.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-1. Define the criteria and constraints of a design problem with sufficient precision to ensure a successful solution, taking into account relevant scientific principles and potential impacts on people and the natural environment that may limit possible solutions. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Define a design problem that can be solved through the development of an object, tool, process or system and includes multiple criteria and constraints, including scientific knowledge that may limit possible solutions. Alignment agreement: | The more precisely a design task's criteria and constraints can be defined, the more likely it is that the designed solution will be successful. Specification of constraints includes consideration of scientific principles and other relevant knowledge that is likely to limit possible solutions. Alignment agreement: | All human activity draws on natural resources and has both short and long-term consequences, positive as well as negative, for the health of people and the natural environment. Alignment agreement: The uses of technologies and any limitations on their use are driven by individual or societal needs, desires, and values; by the findings of scientific research; and by differences in such factors as climate, natural resources, and economic conditions.Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-2. Evaluate competing design solutions using a systematic process to determine how well they meet the criteria and constraints of the problem. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Evaluate competing design solutions based on jointly developed and agreed-upon design criteria. Alignment agreement: | There are systematic processes for evaluating solutions with respect to how well they meet the criteria and constraints of a problem. Alignment agreement: | |
| NGSS Performance Expectation | ||
|---|---|---|
|
MS-ETS1-4. Develop a model to generate data for iterative testing and modification of a proposed object, tool, or process such that an optimal design can be achieved. (Grades 6 - 8) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Develop a model to generate data to test ideas about designed systems, including those representing inputs and outputs. Alignment agreement: | Models of all kinds are important for testing solutions. Alignment agreement: The iterative process of testing the most promising solutions and modifying what is proposed on the basis of the test results leads to greater refinement and ultimately to an optimal solution.Alignment agreement: | |
International Technology and Engineering Educators Association - Technology
-
Students will develop an understanding of the attributes of design.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Students will develop an understanding of engineering design.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
Create representations of the tools people made, how they cultivated food, made clothing, and built shelters to protect themselves.
(Grades
3 -
5)
More Details
Do you agree with this alignment?
-
Create solutions to problems by identifying and applying human factors in design.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- 0.5 liter (500 ml) plastic water or soda bottle
- ½ cup granulated activated carbon (GAC); available from chemical supply stores and pet supply stores in the aquarium section
- 2-3 cotton balls
- jar, smaller in diameter than the 500 ml bottle so that the water bottle can be supported in the jar without falling through
For entire class to share (and the teacher to use):
- drill with small diameter bit
- kitchen knife or scissors
- 3 teaspoons of chocolate powder mix, such as Hershey's® or Nestle Quik®
- water, enough water for each bottle to be filled up 3-4 oz.
- pitcher or bucket, for mixing powdered chocolate and water
- 4-5 cups of sand
- 50+ marbles
Introduction/Motivation
How many of you know someone who filters their tap water? How many of you know someone who buys bottled water? Have you ever been in the woods or mountains and drank water from a river after being filtered by a small pump? Why do we do this? Does the water look contaminated? (Answer: Sometimes, yes.) Naturally pure water does not exist. All water sources carry small particles that have been absorbed or dissolved in the water as it passes over river beds, rocks and soil.
In the early 1900s, the U.S. government began requiring specific levels of cleanliness for drinking water. These regulations did not become strict until the Safe Water Drinking Act of 1974, which then underwent changes in 1986 and 1996. This law tells us exactly which drinking water standards our states must meet to provide safe and healthy drinking water to our homes and communities. The U.S. Environmental Protection Agency (EPA) has set standards for more than 80 contaminants that may get into our drinking water and make us sick.
What happens when a natural disaster happens and a community loses access to clean, drinkable water? (Wait for student answers). Natural disasters (earthquakes, tsunamis, hurricanes, etc.) happen around the world and entire communities can lose access to food, water, and medical care.
Today, we are going to investigate drinking water filters and use the engineering design process to design your own drinking water filter for a community that lost access to clean water after a hurricane. We are going to take a sample of muddy water and use the available materials to clean up the water as much as possible. (If available, show students a Pur® filter and ask them if they know what it is composed of; tell them that they will prepare their own Pur® filter for "muddy" water.)
Procedure
Before the Activity
- With a knife or scissors, cut off and discard the bottoms of each empty 0.5 liter bottle.
- Using a drill, make two holes in each bottle screw top. Although this step is not required, it slows the water flow, resulting in less mess in the classroom.
- Prepare a supply of "dirty" water by mixing two or three teaspoons of chocolate powder and the cold water in the pitcher. Don't worry if the powder makes lumps at the top of the mixture.
- Construct one or two filters as examples and a control. For the control, turn one plastic bottle upside down, with the cut opening to the top, and place a cotton ball in the lid. Screw the lid back onto the bottle. Use this filter as the control filter for students by pouring the mixture into the bottle and watching the chocolate/water mixture pour out. Create another filter or two the same way, but add whatever mixtures of cotton, sand and marbles that you would like to serve as example filters.
With the Students
- Ask the class if they know what the engineering design process is. (Elicit student answers). Explain that the engineering design process (EDP) is series of steps that guides engineering teams as we solve problems. The design process is iterative, meaning that we repeat the steps as many times as needed, making improvements along the way as we learn from failure and uncover new design possibilities to arrive at great solutions. Either show them the image below or write the 7 steps on the board in a circle.
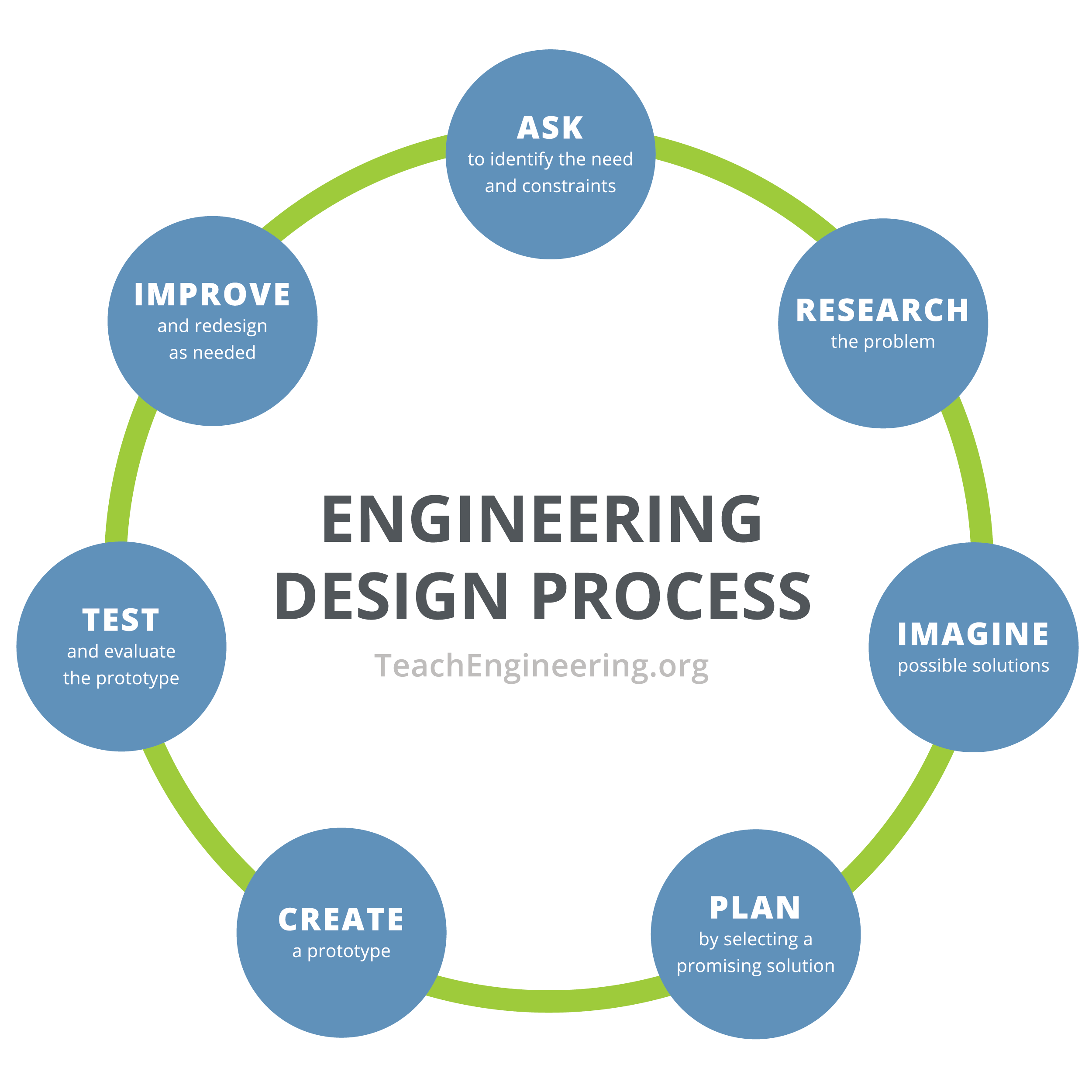
- Divide the class into groups of four to five students each.
- EDP Step 1: Identify Needs and Constraints: Explain to the class that they are going work as if they are environmental engineers designing water filters for a community that lost access to clean water after a hurricane. Their engineering challenge is to make the cleanest water possible out of the provided dirty water sample using any combination of the available materials. One requirement is that each filter must include a cotton ball in the bottle lid.
- Have students make predictions on which filter layer will be the most effective at cleaning the water and why.
- EDP Step 3: Imagine Possible Solutions. Direct students to brainstorm in their teams and then design their devices by making drawings along with short paragraphs that describe what they want to do and why. This is what engineers do. Doing this also encourages students to communicate their ideas to others, which is important when they work in groups, and helps them to analyze their ideas for merit.
- EDP Step 4: Plan by Selecting a Promising Solution. Ask students to revisit the needs, constraints and research from the earlier steps, compare their best ideas, select one solution and make a plan to move forward with it.
- EDP Step 5: Create a Prototype. Have students work together to create the design they decided on with the materials they have available.
- When each group has finished creating their filters, pour about 2" thick (approximately ½ cup) of granulated activated carbon (GAC) on the top of their layers. Now the filters are ready to be used.
- Have students place their filters with the caps downwards on the tops of their glass jars.
- EDP Step 6: Test and Evaluate the Prototype. Next, have the teacher pours the chocolate mixture into the prototype filters for each group. Direct students to observe the "clean" water move from the bottom of the filter into the jar. When the filtration is done, direct students to take their jars and filters to a location where light can illuminate the new mixtures. What happened to the water when it was filtered? (Answer: It turned clearer, less water was remaining, etc.)
- After each group has tested their prototype, ask them to discuss how well their prototype worked, and how well it met the criteria and constraints. Have each team discuss how water filters can be used in the aftermath of a natural disaster.
- Have the class vote on which team's filter cleaned the water the best. As time permits, lead a discussion on why the winning filter was the most effective and ask for suggestions and ideas for how to make the filter even more effective.
Assessment
Pre-Activity Assessment
Know / Want to Know / Learn (KWL) Chart: Before the lesson, ask students to write down in the top left corner of a piece of paper (or as a group on the classroom board), under the title Know, all the things they know about drinking water filters. Next, in the top right corner, under the title Want to Know, ask students to write down anything they want to know about drinking water filters. After the lesson, ask students to list in the bottom half of the page, under the title Learned, all of the things that they have learned about drinking water filters. Ask students to name a few items and write them on the board.
Activity Embedded Assessment
Drawing: Have students draw pictures of their drinking water filters. Require them to sketch and identify the different material layers, indicating as best they can what particles each layer will remove. Point out that this sort of visual documentation and planning is what engineers do when designing new products.
Post-Activity Assessment
Know / Want to Know / Learn (KWL) Chart: Finish the remaining section of the KWL Chart as described in the pre-lesson assessment section. After the lesson, ask students to list in the bottom half of the page, under the title Learned, all of the things that they have learned about drinking water filters. Ask students to name a few items and write them on the board.
Show and Tell: Have teams "show and tell" the filters they created, explaining their work to the rest of the class. Ask students to compare each others' filters and brainstorm other materials and fabrication order that might improve their filters.
Re-Design Practice: If time permits (or as a homework assignment), have students list any design and fabrication changes they would make to improve their filters. If it helps the explanation, have them draw pictures of the new designs. Emphasize that engineers typically re-design products many times before getting a satisfactory solution.
Student-Generated Questions: Have each student come up with one question of their own about filters and/or drinking water to ask the rest of the class. Be prepared to help some students form questions. If the answers are not known by anyone, encourage students to search the U.S. EPA website and report those answers to the class at a later time.
Safety Issues
For safety reasons, pre-cut and pre-drill the bottles so students do not handle the knife and drill.
Troubleshooting Tips
If you cannot find granulated activated carbon (GAC) in chemical stores, use carbon for aquarium filters, which is available at pet shops.
Feel free to make available other filter materials for students to use, such as paper towels, coffee filters, fabric, moss, felt and gravel.
Activity Extensions
Slice open a Pur® (or other type of) filter for students so they can examine its components. Discuss the concept of adsorption—the capability of a solid to attract to its surface molecules of other solid or gaseous materials.
Activity Scaling
For eighth-grade or older students, have each group first filter water using a different individual layer (investigate with sand OR marbles, etc.). Have each team record its observations of that filter type on a group chart on the classroom board. Have teams use these shared research results to inform their own filter designs, referring to them when they explain the logic of the material layers they chose for their final filters. For example, if marbles produce darker filtered water because they let fine particles pass through, while sand does not permit fine particles through, the combination may result in a successful final design approach.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students learn about the various methods developed by environmental engineers for treating drinking water in the United States.

Students learn about water quality testing and basic water treatment processes and technology options. Biological, physical and chemical treatment processes are addressed, as well as physical and biological water quality testing, including testing for bacteria such as E. coli.

Students learn about several possible scenarios of contamination to drinking water, which comes from many different sources, including surface water and groundwater. They analyze the movement of sample contaminants through groundwater, in a similar way to how environmental engineers analyze the phys...

Students experience the steps of the engineering design process as they design solutions for a real-world problem that negatively affects the environment. They use plastic tubing and assorted materials such as activated carbon, cotton balls, felt and cloth to create filters with the capability to re...
Copyright
© 2005 by Regents of the University of ColoradoContributors
Malinda Schaefer; Sharon D. Pérez-Suárez; Matt Lundberg; Ben Heavner; Janet YowellSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under grants from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and National Science Foundation (GK-12 grant no. 0338326). However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: March 14, 2023







User Comments & Tips