Quick Look
Grade Level: 12 (10-12)
Time Required: 30 minutes
Lesson Dependency: None
Subject Areas: Chemistry, Physical Science, Physics
NGSS Performance Expectations:

| HS-PS2-6 |

Summary
Students are presented with the question: "Why does a liquid jet break up into droplets?" and introduced to its importance in inkjet printers. A discussion of cohesive forces and surface tension is included, as well as surface acting agents (surfactants) and their ability to weaken the surface tension of water. Students observe the effects of surface tension using common household materials. Finally, students return to the original question through a homework assignment that helps them relate surface tension and surface area to the creation of water droplets from a liquid jet.Engineering Connection
The inkjet printer has become one of the most widely used printer types. The fundamental principle that enables the operation of inkjet printers is the tendency of a continuous stream of water to break apart and form droplets. Droplets form due to the surface tension of the liquid—a larger surface area requires more energy to maintain due to the molecular forces associated with surface tension. When water transitions from a column into droplets, the same volume of water has a smaller surface area and therefore requires less energy. Chemical engineers must finely adjust the ink's surface tension so it forms droplets of the desired size and so it adheres to the paper surface without smearing or bleeding. Aside from desktop printing on paper, inkjet printers are today being used for many industrial applications such as automotive coatings, decoration of curved and irregularly-shaped surfaces, printing conductive patterns with metallic particles, replacing screen printing on everything from ceramics to textiles, and creating rapid 3D prototypes.
Learning Objectives
After this lesson, students should be able to:
- Explain certain properties of water using the concepts of cohesive forces and surface tension.
- Describe how the properties of water change when surfactants are added.
- Describe how surface tension encourages liquid droplets and soap films to minimize their surface areas.
- Discuss why columns of liquids form droplets, as happens in inkjet printers.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
HS-PS2-6. Communicate scientific and technical information about why the molecular-level structure is important in the functioning of designed materials. (Grades 9 - 12) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This lesson focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Communicate scientific and technical information (e.g. about the process of development and the design and performance of a proposed process or system) in multiple formats (including orally, graphically, textually, and mathematically). Alignment agreement: | Attraction and repulsion between electric charges at the atomic scale explain the structure, properties, and transformations of matter, as well as the contact forces between material objects. Alignment agreement: | Investigating or designing new systems or structures requires a detailed examination of the properties of different materials, the structures of different components, and connections of components to reveal its function and/or solve a problem. Alignment agreement: |
International Technology and Engineering Educators Association - Technology
-
Explain how knowledge gained from other content areas affects the development of technological products and systems.
(Grades
6 -
8)
More Details
Do you agree with this alignment?
State Standards
North Carolina - Science
-
Compare physical and chemical properties of various types of matter.
(Grades
9 -
12)
More Details
Do you agree with this alignment?
Introduction/Motivation
(In advance, have printouts of the pictures in Figures 1 and 2 [or similar], the ability to show youtube videos, the capability to blow bubbles [bubble wand and soap solution], and a round-shaped balloon to blow up, as described below.)
(Show the class a photograph of falling water, such as Figure 1, and one or more of the high-speed camera videos of inkjet printers posted on youtube by user imagexpertinc, at: https://www.youtube.com/user/imagexpertinc.)
When you turn on a water faucet so that only a small stream comes out, the water starts out as cylindrical column, and ends up as droplets. Have you noticed this? This also happens to ink ejected from the nozzles of inkjet printers. Why do the water and ink do that? Why doesn't the liquid stay in a stream? And why does it form round drops—why doesn't it break into little cubes instead?
(The answer is: Creating the air-water boundary requires additional energy. The more surface area at the air-water boundary, the more energy is required. The water drops have a smaller surface area compared to the same volume of water in a column, so the water droplets are the lower energy state. But, you do not need to introduce this to the students yet. Some students may suggest that gravity pulls the water apart, but the drops will form even if a thin jet of water is shot upwards or to the side as in some inkjet printers. If a student says that it is "easier" for the water to form spherical drops instead of cubes, encourage them to think about why it is easier. Also, if a student points out that water does not form droplets if you turn the water on high, explain that it would if the water had further to fall.)
(Show the class a picture of the water strider in Figure 2.)
This insect is called a water strider. How do you think it got its name? What is it doing? What would happen if you tried to stand on the water's surface like this bug is doing? Why can he stand on top of the water, but you cannot? (The answer is not that the insect is less dense than water. You can float because you are less dense than water but you cannot stand on the surface of the water like the water strider.)
When was the last time you blew soap bubbles? (Optional; use a bubble wand and soap solution to blow bubbles at the students.) Why can't we make bubbles with just plain water? What shape are all of the bubbles I blow? Unless the bubble touches something else, it always makes a sphere. Why? Following the lesson refer to the associated activity Surface Tension Lab for students to conduct a hands-on experiment to further investigate this question!
What happens if I touch a soap bubble? Why does it pop? (Burst some of the blown bubbles.) Can you think of another toy that pops like a bubble? (Bring out a balloon or inflate one in class after a student guesses correctly.) How is the balloon like the bubble? How is it different? Do you think the soap film that makes up the bubble is anything like rubber that makes up the balloon? How could we find out? What happens when I pop the balloon? What happens to the rest of the balloon? Now, what do you think happens to the rest of the soap bubble when I touch it? (Blow bubbles at students and let them pop the bubbles to see what happens.)
Water molecules really like to stick together and they really do not like being on the outside. Those simple facts help to explain all of the examples we have just talked about. We are going to talk about why water molecules really like to stick to each other and why that causes water to act the way it does.
Lesson Background and Concepts for Teachers
Surface Tension

One striking and interesting property of all liquids is surface tension. In any liquid, intermolecular forces cause the liquid molecules to be attracted to each other. These forces that pull liquid molecules towards each other are known as "cohesive" forces. In the body of a liquid, a molecule is surrounded by molecules in all directions, so the attractive forces cancel and the molecule feels no overall force (see Figure 3).
However, on the surface of the interface between the liquid and air, a molecule in the liquid feels the attractive forces of the other molecules within the liquid, but none from outside. This causes the outer layer of the liquid to act like a stretched membrane and minimize the surface area. Just as in a filled balloon, "tension" exists in this elastic surface, and energy is stored in this surface, just like elastic energy is stored in the rubber of the balloon. To minimize the stretching of the skin, and lower the amount of energy in tension on the surface, the liquid molecules move to create the least surface area possible. It is for this reason that falling water drops and ink in inkjet printers form spheres—it allows for the least amount of surface area for a given volume of liquid. This is why we call the elastic pull on the surface that minimizes the area on the liquid-air boundary surface tension.
In the ink delivery system in a continuous inkjet printer (see Figure 5), ink is removed from a reservoir and pumped through a nozzle. Soon after leaving the nozzle, the column of ink separates into spherical droplets. These ink droplets are first charged and then directed into position by charged deflection plates. The surface tension of the ink is vital for this process to work: if the surface tension is too high the ink may clog the nozzle or not adhere properly to the paper, and if the surface tension is too low it can cause ink to leak from the nozzle or cause the ink to bleed on the paper. 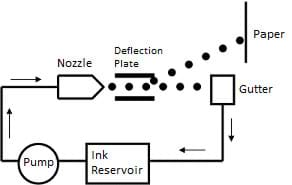
Originally used only for desktop printing, inkjets are now used for many different applications. By using an ink with metallic particles, circuits can printed onto a variety of substrates. Specially designed inkjet devices paint automobiles and apply decorations to irregular or rounded objects. Ink jet printing has already begun to replace screen printing in textiles and ceramics because of advantages in speed, choice of design, and ease of use. Inkjet printing is also used to create 3D prototypes of computer-generated objects.
Changing Surface Tension

Under certain conditions, it is desirable to lower the surface tension. A high surface tension encourages a liquid to bead rather than spread evenly across a surface. For this reason surface-active agents, or surfactants, are used for various applications to lower the surface tensions of liquids. Inks and paints (see Figure 6) are everyday examples in which lowering surface tension is useful in making liquids spread.
Using surfactants to lower surface tension is also used in the oil industry. Surface tension causes oil to become trapped in the pores of the containing rock due to a phenomenon called capillary action. By adding surfactants to the oil deposit, it helps the oil release from the pores and become available for extraction (see Figure 6).
Finally, certain semi-aquatic insects release surfactants for locomotion. The released surfactant lowers the surface tension behind them, and the insects are pulled forward by the stronger surface tension in the untreated water ahead of them.
Associated Activities
- Surface Tension Lab - Students design and test water and soap solutions for making bubbles. They experiment with additives to their best "recipes" to enhance the bubbles. In a math homework, students perform calculations that explain why soap bubbles form spheres.
Vocabulary/Definitions
surface tension: The property of the surface of a liquid that allows it to resist an external force. This property is caused by cohesion of like molecules and explains many of the behaviors of liquids. Source: Wikipedia, May 2011.
Assessment
Pre-Lesson Assessment
Discussion Questions: Ask the students and discuss as a class:
- How do water bugs walk on the surface of the water? (Answer: The water molecules are pulled inwards and create a slightly thicker film on the surface that acts like a springy membrane. The water bug can walk on top of this surface. We call this phenomenon surface tension.)
- Why do soap bubbles form spheres instead of cubes? (Students will find out during this lesson and associated activity. Answer: This is the result of a relationship between the volume of air inside the bubble and the surface area of the soap film of the bubble. The surface tension tries to minimize the surface area. For the same volume of air, a sphere has a smaller surface area than a cube.)
- What happens when a soap bubble pops? (Answer: The trapped air is released and the surface tension pulls the water into water droplets.)
- Why does a stream of water form droplets as it falls? (Answer: Again, the water droplets have a smaller surface area than the column of water, so the surface tension causes the water to form droplets.)
Post-Introduction Assessment
Creative Writing and Illustration: Ask students to imagine that they are water molecules and describe what is happening to them in the situations below. Have them draw illustrations to go along with their descriptions.
- You are in the middle of a water drop.
- You are on the surface of a water drop.
- You are in a soap bubble. (Note: This is a bit tricky and a good challenge to see if students really understand. A soap bubble is actually made of two surfaces, inside and outside of the bubble. Both surfaces are trying to make the bubble as small as possible, hence the spherical shape, but the air trapped inside the bubble means the bubble cannot squeeze down into a simple water droplet.)
Lesson Summary Assessment
Two Circles Game: To reinforce ideas about surface tension, have students form two circles, one inside the other. Arrange for equal (or close to equal) numbers of students in both circles. Have students in each circle face their "partners" in the other circle. Have the students of one circle to explain one of the concepts listed below to their partners in the other circle, and then the partners explain the concepts back to them. Between questions, have the two circles move in opposite directions so that partners are changed. If students have trouble explaining a concept, change partners and repeat the same question. Example questions:
- What is surface tension?
- Why does surface tension try to minimize surface area?
- What are surfactants? What do they do?
Additional Multimedia Support
See short, high-speed camera videos of inkjet printers posted to youtube by user imagexpertinc, at: https://www.youtube.com/user/imagexpertinc.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students extend their understanding of surface tension by exploring the real-world engineering problem of deciding what makes a "good" soap bubble. Student teams first measure this property, and then use this measurement to determine the best soap solution for making bubbles. They experiment with ad...

Students are presented with a short lesson on the difference between cohesive forces (the forces that hold water molecules together and create surface tension) and adhesive forces (the forces that causes water to "stick" to solid surfaces. Students are also introduced to examples of capillary action...

Students learn about the basics of molecules and how they interact with each other. They learn about the idea of polar and non-polar molecules and how they act with other fluids and surfaces. Students acquire a conceptual understanding of surfactant molecules and how they work on a molecular level. ...

Student teams are challenged to evaluate the design of several liquid soaps to answer the question, “Which soap is the best?” Through two simple teacher class demonstrations and the activity investigation, students learn about surface tension and how it is measured, the properties of surfactants (so...
References
Bush, John W.M. and David L. Hu. "Walking on Water: Biolocomotion at the Interface." Annual Review of Fluid Mechanics. 38 (2006): p. 339-369. Accessed August 2010. (Includes a discussion on how insects move on the surface of the water including water-walking, meniscus climbing and Marangoni [surfactant] propulsion.) http://www.me.gatech.edu/hu/Publications/Hu06_Bush.pdf
Chemical Functional Definitions: Surfactants. Posted 2005. Science in the Box, Proctor and Gamble. Accessed August 17, 2010. (Explanation of surfactants) http://www.scienceinthebox.com/en_UK/glossary/surfactants_en.html
de Gennes, Pierre-Gilles, et al. Capillarity and Wetting Phenomena: Drops, Bubbles, Pearls, Waves. New York, NY: Springer, 2004. (Engineering connections)
Jones, Andrew Zimmerman. Surface Tension. About.com. Accessed August 17, 2010. http://physics.about.com/od/physicsexperiments/a/surfacetension.htm
Magdassi, Shlomo, et al. The Chemistry of Inkjet Inks. Worldscibooks.com: World Scientific Books, July 2009. http://www.worldscibooks.com/etextbook/6869/6869_chap01.pdf.
Surface Tension. Wikipedia.com.. Last updated August 16, 2010. Wikimedia Foundation, Inc. Accessed August 17, 2010. http://en.wikipedia.org/wiki/Surface_tension
Tabeling, Patrick. Introduction to Microfluidics. New York, NY: Oxford University Press, 2005, p. 118-119.
Copyright
© 2013 by Regents of the University of Colorado; original © 2011 Duke UniversityContributors
Jean Stave, Durham Public Schools, NC; Chuan-Hua Chen, Mechanical Engineering and Material Science, Pratt School of Engineering, Duke UniversitySupporting Program
NSF CAREER Award and RET Program, Mechanical Engineering and Material Science, Pratt School of Engineering, Duke UniversityAcknowledgements
This digital library content was developed under an NSF CAREER Award (CBET- 08-46705) and an RET supplement (CBET-10-09869). However, these contents do not necessarily represent the policies of the National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: July 2, 2019






User Comments & Tips