Quick Look
Grade Level: 4 (3-5)
Time Required: 30 minutes
Expendable Cost/Group: US $0.90
Group Size: 2
Activity Dependency: None
Subject Areas: Physical Science
NGSS Performance Expectations:

| 3-PS2-3 |
Summary
In this hands-on activity, students explore the phenomenon of static electricity by engaging in the science and engineering practices of asking questions and planning and carrying out investigations. Students enact a “human diagram” to model the Bohr model of an atom. Then, in pairs, they charge a comb (on hair) and use it to attract an O-shaped piece of cereal and then watch the cereal jump away when it touches the comb. They do the same with Styrofoam pellets, observing them pulling towards a charged comb, then leaping back to the table. Through these investigations, students explore the disciplinary core ideas of electric and magnetic forces, the crosscutting concept of cause and effect, and they make sense of static electricity and what’s going on at the atomic level.
Engineering Connection
Engineers consider static electricity when designing, manufacturing and packaging electronic circuit boards. If too much static electricity is present, the electrical components on the circuit board may malfunction and the circuit board may become inoperable. When fabricating circuit boards for computers and electronics, engineers wear special white suits called "bunny suits" and work in "clean rooms" to help protect the circuit boards from contaminants and static electricity.
Learning Objectives
After this activity, students should be able to:
- Describe static electricity and how it affects different objects.
- Describe different manifestations of the electrostatic force that occur as a result of the transfer of electric charge.
- Explain that engineers must consider static electricity when designing, manufacturing and packaging electronic equipment like circuit boards.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
3-PS2-3. Ask questions to determine cause and effect relationships of electric or magnetic interactions between two objects not in contact with each other. (Grade 3) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Ask questions that can be investigated based on patterns such as cause and effect relationships. Alignment agreement: | Electric, and magnetic forces between a pair of objects do not require that the objects be in contact. The sizes of the forces in each situation depend on the properties of the objects and their distances apart and, for forces between two magnets, on their orientation relative to each other. Alignment agreement: | Cause and effect relationships are routinely identified, tested, and used to explain change. Alignment agreement: |
International Technology and Engineering Educators Association - Technology
-
Students will develop an understanding of the relationships among technologies and the connections between technology and other fields of study.
(Grades
K -
12)
More Details
Do you agree with this alignment?
-
The process of experimentation, which is common in science, can also be used to solve technological problems.
(Grades
3 -
5)
More Details
Do you agree with this alignment?
State Standards
Colorado - Science
-
Show that electricity in circuits requires a complete loop through which current can pass
(Grade
4)
More Details
Do you agree with this alignment?
Materials List
Each group needs:
- comb, rubber or plastic; alternatively, a balloon that is blown-up and tied-off
- thread, 12-inch piece
- O-shaped cereal, 1 piece
- long dry hair, or dry wool cloth
- a few Styrofoam packing pellets
- scotch tape
- counter top or table
- Fun Things to Do with a Rubber Comb Worksheet, one per student
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/cub_electricity_lesson01_activity1] to print or download.Introduction/Motivation
Have you ever removed a hat during the winter and noticed your hair standing up—or even crackle? (Listen to students; some may say yes.) What causes this to happen? (Answer: Static electricity.) Have you ever removed clothes from a dryer and noticed them sticking to each other? (Expect some to say yes.) And did you ever hear the crackle sound when you pulled the clothes apart? What causes that? (Answer: This is also static electricity.) How is it possible that what causes your hair to stand up after taking off a winter hat is the same as what makes your clothes stick together in the clothes dryer?
(On the classroom board, draw a diagram of a Bohr model, as shown in Figure 1.) All ordinary matter—everything around us—is made of atoms. Atoms have positively charged nuclei and negatively charged electrons surrounding the nucleus. In the nucleus are protons, which have a positive charge, and neutrons, which have a neutral charge (neither positive nor negative).
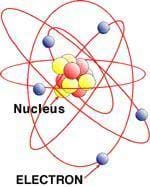
Let's demonstrate the Bohr model of an atom—at human scale. (Have students create a human diagram of the Bohr model. Set up two chairs in an open area. Ask for 12 volunteers. Have two students walk in one direction around the first chair and two students around the second chair. They represent the first shell of the atom. Next, have one student walk in the other direction around the first chair, and seven students walk around the second chair to represent the second shell of electrons.) Static electricity happens when the two atoms rub together and the single electron from the first atom is transferred to the seven-electron shell of the second atom.
Static electricity depends on the balance of charges in an atom. We know that atoms can move from one atom to another, creating electricity! Also, electrons (or charge) can move from one object to another by rubbing two objects together. Have you ever heard the phrase "opposites attract" when talking about magnets? Well, the same is true when it comes to charges in an atom. A positive and a negative charge (opposites) pull toward each other. However, negative charges push away other negative charges and positive charges repel other positive charges. The attraction and repulsion (pushing away) of charges is the basis of static electricity. We will learn more about static electricity during the upcoming activity.
Engineers consider static electricity when designing, manufacturing and packaging electronic circuit boards. If too much static electricity is present, the electrical components on the circuit board may malfunction and the circuit board may become inoperable. When fabricating circuit boards for computers and electronics, workers wear special white suits called "bunny suits" and work in "clean rooms" to help protect the circuit boards from things like static electricity. These bunny suits prevent human skin and hair particles, as well as lint particles from entering the clean room, which can damage the circuit boards by conducting electricity between two spots on the board that should not be connected. Also, a bunny suit helps prevent static electricity in your body from sending unwanted electric charges to the circuit boards and damaging them.
Procedure
Background: The Atom
Everything we see around us—all ordinary matter—is made of atoms. Every atom consists of negatively charged electrons and a positively charged center called a nucleus. The nucleus is made of positively charged protons and neutral-charged (neither positively nor negatively charged) neutrons. In a simple model of an atom, known as a Bohr model (see Figure 1), it is assumed that the electrons are spinning around the nucleus of the atom on paths called orbitals. One can visualize this by thinking of satellites orbiting the Earth, or the moon orbiting around the Earth. The positive charges of the protons in the nucleus attract the negative charges of the electrons orbiting around the nucleus (opposites attract), maintaining the electrons' orbit. Charge is a fundamental quantity in electricity. The smallest amount of charge that is known to exist is carried by an electron and has a charge of -1.602 x 10-19 coulomb [C]. The other charge-carrying portion of the atom is the proton, which has a charge of +1.602 x 10-19 coulomb [C]. The unit used to measure charge is known as the Coulomb, named after French engineer and physicist Charles Coulomb.
Background: Static Electricity
In an atom, the protons and neutrons that make up the nucleus are held together very tightly and rarely does the nucleus experience a change. However, some of the electrons that are associated with the atom are loosely held to their orbital. These electrons, which typically reside in the outer orbits, can move from one atom to another. When an atom loses electrons, it has more positive particles than negative particles, which results in a positive net charge for the atom. An atom that acquires electrons has more negative particles than positive particles and, thus, has a negative net charge.
If the atoms in a material hold the electrons in the outer orbits tightly, the electrons are less likely to move to another atom. Such materials are known as insulators. Alternatively, materials whose atoms willingly give up and accept electrons are known as conductors. Conductors permit electrons to move through the material easily.
It is possible to transfer (or move) electrons from one material to another. One way to do this is by rubbing two objects together. The longer that two objects are rubbed together, the larger the quantity of electron movement from one object to the other, which results in a charge build up on each object. Static electricity occurs when imbalance of positive charges and negative charges exists.
Positive and negative charges behave similarly to the north and south poles of magnets: Opposite poles attract and like poles repel. In the case of charges, a positive and negative charge pull towards each other. Positive charges repel other positive charges, and negative charges push away other negative charges. Therefore, an object that has a positive or negative charge build up (net charge) attracts an object that is neutral. For instance, when you rub a balloon on your hair or a piece of wool cloth, the balloon acquires additional electrons. If you hold the balloon against a wall, the balloon sticks. This is because the negatively charged electrons on the balloon push away the negatively charged electrons in the wall (like charges repel) and attract the positive charges in the wall (opposites attract), causing the balloon to "stick" to the wall. Additionally, when an object with charge build-up attracts a neutral object, the electrons tend to move to areas where the electrical charge is positive until the atoms in both objects are neutral or balanced. When a large number of electrons move in an effort to balance the atoms, sometimes it causes a spark. This spark is a result of static electricity.
Before the Activity
- Gather materials and make copies of the Fun Things to Do with a Rubber Comb Worksheet.
- You may want to cut the thread in advance.
With the Students
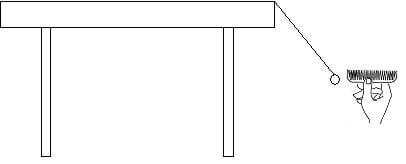
- Direct students to suspend an O-shaped piece of cereal from a tabletop, using tape and a piece of thread (see Figure 2).
- Next, quickly rub a comb (or balloon) through your hair or on a piece of wool cloth. Note: For this entire activity, a blown-up and tied-off balloon may substitute for a rubber comb.
- Hold the comb near the cereal. Expect the cereal to swing toward the comb (see Figure 1). Hold the comb still until the cereal touches the comb. Continue to watch and observe the cereal quickly move away from the comb
- Ask students: Why was the cereal attracted to the comb? And then why did it move away once it touched the comb? (Answer: The rubber comb attracted the loosely bound electrons that were on your hair or wool. Once the comb attracted the electrons, it gained a negative charge. The cereal initially has a neutral charge; therefore, it is attracted to the comb because of its different charge distribution. However, when the cereal touches the comb, it gains some of the comb's negative charge, and the cereal's charge distribution becomes the same as that of the comb. Next, the two objects repel each other since they are now "like objects," and as a result, the cereal moves away.)
- Direct each group to spread some Styrofoam pellets on a table.
- Rub the comb through your hair or against the wool cloth again. Next, move the comb close to the Styrofoam pellets. The pellets stick to the comb. After several seconds, the Styrofoam jumps back to the table. Discuss what was observed (similar to step 3).
- If time permits, extend the activity to include the math applications described in the Activity Extensions section.
Assessment
Pre-Activity Assessment
Prediction: Have students predict the outcome of the activity before it is conducted. First, give an overview of the activity, then ask students the following question and have them write their answer on a piece of paper. Then, perform the activity.
- What do you think will happen if you rub the comb on your sweater and hold it by a piece of cereal?
Activity Embedded Assessment
Worksheet/Pairs Check: At the beginning of the activity, hand out the Fun Things to Do with a Rubber Comb Worksheet. Have students work individually or in pairs to complete the worksheet. Have students who work in pairs check each others' answers. Review students' answers to gauge their depth of comprehension.
Post-Activity Assessment
Student-Generated Questions: Have students each come up with a question of their own to ask the rest of the class. Be prepared to help some students form questions. Example questions:
- What type of electricity attracted the cereal to the comb at first?
- Why did it repel quickly after it touched the comb?
- Do items with negative charges (more electrons) attract or repel other items with negative charges?
- What are the causes and effects of static electricity?
Question Races: Have each student group number off by threes or fours so that at least three groups have the same number. Ask a question and call a number. Have each group with that number send one student to run up to the board and write the answer for the question. Each student who gets it right earns their team a point.
- What is an example of static electricity? (Example answer: Walking across carpet and getting a shock when touching a door knob.)
- Who invented the light bulb? (Answer: Thomas Edison)
- What part of an atom can move to another atom? (Answer: Electron)
- How many electrons reside in the first orbital around the atom? (Answer: 2)
- How many electrons reside in the second orbital around the atom? (Answer: 2)
- How many electrons reside in the third orbital around the atom? (Answer: 6)
Troubleshooting Tips
When rubbing a comb in hair, it works best if the hair is dry and clean. Otherwise, a piece of wool material works best.
Humidity dissipates charge, so this activity does not work as well on humid or rainy days.
Activity Extensions
Have students bring in different types of cereal from home. Then ask them to predict how long the cereal will stay attracted to the comb. Have them graph the data: the length of attraction (time) vs. cereal type.
Extend the activity to include the following experimental variations and math applications:
- Does the size of the comb or cereal make a difference? Try measuring diameters of different cereals or measuring the comb length. Test a hypothesis on whether size of the objects matters.
- How far can the cereal be made to move? Measure the distances at which you can place the comb and still get the cereal to move.
- Try timing the rubbing time of the comb and see if doubling the rubbing time makes the cereal move farther or faster.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

This lesson introduces the concept of electricity by asking students to imagine what their life would be like without electricity. Students learn that electrons can move between atoms, leaving atoms in a charged state.

Students come to understand static electricity by learning about the nature of electric charge, and different methods for charging objects. In a hands-on activity, students induce an electrical charge on various objects, and experiment with electrical repulsion and attraction.

Students are introduced to the concept of electricity by identifying it as an unseen, but pervasive and important presence in their lives. They compare conductors and insulators based on their capabilities for electron flow. Then water and electrical systems are compared as an analogy to electrical ...

Students are introduced to the fundamental concepts of electricity. They address questions such as "How is electricity generated?" and "How is it used in every-day life?" Illustrative examples of circuit diagrams are used to help explain how electricity flows.
References
Reuben, Gabriel. Electricity Experiments for Children. New York, NY: Dover, 1968.
Static Electricty. Electricity and Magnetism, ThinkQuest. Accessed November 7, 2005. Formerly available at: http://library.thinkquest.org/CR0211620/
VanCleave, Janice. Physics for Every Kid. New York, NY: John Wiley & Sons, 1991.
Copyright
© 2004 by Regents of the University of ColoradoContributors
Monica Maxwell; Daria Kotys-Schwartz; Malinda Schaefer Zarske; Denise W. CarlsonSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under grants from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education, and National Science Foundation (GK-12 grant no. 0338326). However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: September 21, 2022






User Comments & Tips