Summary
In this hands-on activity, students make sense of the phenomenon of the electrical force that takes place between two objects. Each student builds an electroscope and, utilizing the science and engineering practices of asking questions and making observations, uses the device to draw conclusions about objects’ charge intensity. Students also determine what factors influence electric force. Throughout this activity, students explore the disciplinary core ideas of electric and magnet force and measurement and the crosscutting concepts of cause and effect and standard units.
Engineering Connection
In the case of electricity, what you cannot see could hurt you, so engineers use their understanding of electricity to develop devices that measure how much electrical charge is present. On a spacecraft orbiting Earth or traveling in space, the presence of excess electrical charge can cause the malfunction of delicate electronics and put astronauts' lives at risk. Electrical engineers design devices that enable astronauts to monitor the amount of charge on a spacecraft and safely scatter excess electrical charge to keep the spacecraft operational.
Learning Objectives
After this activity, students should be able to:
- Understand why electrical force occurs between two objects.
- Determine what factors influence electric force
- Identify metal as a material that transmits electricity
- Understand that engineers use their understanding of electricity to develop devices that measure how much electrical charge is present.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
3-PS2-3. Ask questions to determine cause and effect relationships of electric or magnetic interactions between two objects not in contact with each other. (Grade 3) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Ask questions that can be investigated based on patterns such as cause and effect relationships. Alignment agreement: | Electric, and magnetic forces between a pair of objects do not require that the objects be in contact. The sizes of the forces in each situation depend on the properties of the objects and their distances apart and, for forces between two magnets, on their orientation relative to each other. Alignment agreement: | Cause and effect relationships are routinely identified, tested, and used to explain change. Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
5-PS1-3. Make observations and measurements to identify materials based on their properties. (Grade 5) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This activity focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Make observations and measurements to produce data to serve as the basis for evidence for an explanation of a phenomenon. Alignment agreement: | Measurements of a variety of properties can be used to identify materials. (Boundary: At this grade level, mass and weight are not distinguished, and no attempt is made to define the unseen particles or explain the atomic-scale mechanism of evaporation and condensation.) Alignment agreement: | Standard units are used to measure and describe physical quantities such as weight, time, temperature, and volume. Alignment agreement: |
Common Core State Standards - Math
-
Use a pair of perpendicular number lines, called axes, to define a coordinate system, with the intersection of the lines (the origin) arranged to coincide with the 0 on each line and a given point in the plane located by using an ordered pair of numbers, called its coordinates. Understand that the first number indicates how far to travel from the origin in the direction of one axis, and the second number indicates how far to travel in the direction of the second axis, with the convention that the names of the two axes and the coordinates correspond (e.g., x-axis and x-coordinate, y-axis and y-coordinate).
(Grade
5)
More Details
Do you agree with this alignment?
International Technology and Engineering Educators Association - Technology
-
Explain how various relationships can exist between technology and engineering and other content areas.
(Grades
3 -
5)
More Details
Do you agree with this alignment?
State Standards
Colorado - Math
-
Represent real world and mathematical problems by graphing points in the first quadrant of the coordinate plane, and interpret coordinate values of points in the context of the situation.
(Grade
5)
More Details
Do you agree with this alignment?
Colorado - Science
-
Show that electricity in circuits requires a complete loop through which current can pass
(Grade
4)
More Details
Do you agree with this alignment?
-
Describe the energy transformation that takes place in electrical circuits where light, heat, sound, and magnetic effects are produced
(Grade
4)
More Details
Do you agree with this alignment?
Materials List
Each student needs:
- a square of aluminum foil (about 10 cm by 10 cm)
- a wide-mouth glass or plastic jar (ask students to bring from home)
- thin cardboard, larger than the jar opening (ask students to bring cereal boxes from home)
- silver foil from a candy bar (ask students to bring from home)
- a pair of scissors
- a balloon
- rigid, bare wire (10-15 cm) or unbend a paper clip
- scotch tape
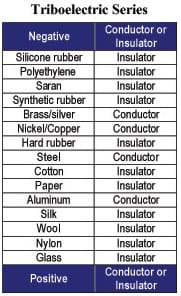
- pairs of items, such as wool and nylon, from the Triboelectric Series Reference Sheet (see Figure 1)
- Charge Detector Graphing Worksheet (optional math component)
For the entire class to use:
- few cups or bowls of warm water for soaking candy bar wrappers that have a paper backing
- a few pieces of wool (available at fabric stores)
- a few large sewing needles (to poke holes into the cardboard)
- small pliers (may be necessary to bend the wire)
- a few bar magnets (optional, for the activity introduction)
Worksheets and Attachments
Visit [www.teachengineering.org/activities/view/cub_electricity_lesson02_activity2] to print or download.Introduction/Motivation
Pass out magnets to each of the students, if available. Have them figure out which are the north/south ends.
Tell students that we know that every atom has negatively-charged electrons, positively-charged protons and neutral-charged neutrons. We also know that opposite charges (positive and negative) attract and like charges (negative and negative, or positive and positive) repel or push away from each other. Ask student to raise their hands if they think there is a lot of force pulling opposite charges together? Then ask the students to raise their hands if they think there is a lot of force pushing like charges away from each other. (Answer: The amount of force depends on the amount of charge and the distance between the charges.)
Explain to students that the force pushing or pulling on the charges is called electrical force. Any two objects with different charges from each other have an electric force between them. The amount of force pushing or pulling two objects together depends on the distance between the objects and the amount of charge on each object.
Electric charge can build up on a spacecraft when it is orbiting Earth or traveling in space. Excess charge buildup is dangerous to a spacecraft's delicate electronics, and can be harmful to astronauts on space walks. Scientists and engineers develop measuring devices that enable NASA engineers and astronauts to determine the amount of charge on a spacecraft when it is in orbit. Electrical and aerospace engineers design devices that can safely scatter this excess electric charge when the spacecraft is in operation in space.
Procedure
Background - Electric Force Concepts
The force that occurs between two charged objects is an electrical force. This electrical force is dependent on the amount of charge on the objects and the distance between the objects. The electric force increases when an object has a greater amount of charge. The electrical force decreases when the distance between two objects is increased.
Like electrical charges repel each other, while opposite electrical charges attract. The electric force between two charged objects is given by the equation:
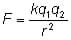
where q1 and q2 are the charges on the two objects, r is the distance between the objects and k is a constant equal to 9 x 109 Nm2/C2.
At the end of the activity, have students complete the Charge Detector Graphing Workshop to graph the electric force between two objects as a function of the objects' charges and as a function of the distance between them.
Background - Conductors and Insulators
In an atom, the protons and neutrons that make up the nucleus are held together very tightly, and rarely does the nucleus experience a change. However, some of the electrons that are associated with the atom are loosely held to their orbital (path around the nucleus). These electrons, which typically reside in the outer orbits, can move from one atom to another. When an atom loses electrons, it is left with more positive particles than negative particles, which results in a positive net charge for the atom. An atom that acquires electrons ends up with more negative particles than positive particles and, thus, has a negative net charge.
If the atoms of a material hold the electrons in the outer orbits tightly, the electrons are less likely to move to another atom. Such materials are known as insulators; for example: glass, plastic and paper. Alternatively, materials whose atoms willingly give up and accept electrons are known as conductors. Conductors allow electrons to move through the material easily. Some common conductors are metals such as steel, copper and aluminum.
Before the Activity
- Ask students to bring from home some of the items on the materials list (a wide-mouth glass or plastic jar, a piece of thin cardboard [such as from a cereal box], and silver foil candy bar wrapper). The cardboard should be larger than the mouth of the jar.
- Assemble an assortment of items from the list in Figure 1 (or see the Triboelectric Series Reference Sheet).
With the Students
- Before starting the activity, ask students the following true/false questions: A conductor is a material through which charge moves easily and an insulator is a material through which charge does not move easily. (Answer: True.) Like-charged objects attract, while unlike-charged objects repel. (Answer: False. The reverse is true.) When two items are rubbed together, electrons are transferred between them, leaving one negatively charged and the other positively charged. (Answer: True.)
- If the silver foil has a paper backing, have the students soak it in warm water, peel off the paper liner, and carefully dry it without tearing it.
- Cut the silver foil into rectangles about 2 cm by 4 cm (smaller rectangles also work). Very carefully smooth the wrinkles out of the foil.
- Cut a cardboard disk slightly larger than the jar opening. Use the needle to make a hole in the center of the disk.
- Pass the wire through the hole. Make a short bend at the top of the wire. Make a tight ball of the 10 cm by 10 cm aluminum foil. Push it firmly onto the bend at the top of the wire.
- Bend the other end of the wire into a hook shape. Fold the 2 cm by 4 cm silver foil rectangle in half and hang it over the wire hook (see Figure 2). If the ends do not hang side by side, press them together gently.

- Carefully lower the disk with its wire and foil into the jar. Tape the cardboard disk in place to cover the mouth of the jar. The finished electroscope should look similar to Figure 3.
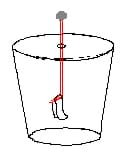
- Charge a balloon by rubbing it vigorously on your hair or on a piece of wool. Bring the charged balloon near the foil ball on top of the jar, being careful not to touch it. While you do this, WATCH THE SILVER FOIL INSIDE THE JAR. What happens? Write down your observations on a piece of paper or in your science journal.

- Recharge the balloon, touch it to the foil ball on the electroscope, and quickly pull the balloon away. BE SURE TO WATCH THE SILVER FOIL INSIDE THE JAR AS YOU MOVE THE BALLOON TOWARD THE JAR, TOUCH THE JAR, AND PULL THE BALLOON AWAY. Now, bring the charged balloon back to touch the foil ball and hold it there. What happened? Write down your observations.
- Next, use the electroscope to detect the charge on different pairs of objects. Between each test, discharge the electroscope by gently touching the foil ball with your finger to make it neutral. First, rub the two objects together. Then charge a neutral electroscope with one of the objects. Observe the behavior of the foil electroscope leaves. Record what you see happening to the silver foil leaves inside the jar. Then, charge the electroscope with the other object. You may need to rub the objects together again between these steps. Remember to discharge the electroscope before charging it again.
- Use the electroscope to detect the strength of charges in different types of materials. Compare the amount of charge in an object after rubbing it with a balloon. Does the amount of rubbing affect the amount of charge?
- Have the students complete the Charge Detector Graphing Worksheet. Refer to the equation described earlier in the Procedure section. What happens as the charge increases in the first graph? (Answer: The force between the two objects increases.) What happens as you move the objects farther apart in the second graph? (Answer: The force between them decreases.)
Activity Observations and Explanations
If you rub a balloon with wool or your hair, the balloon becomes negatively charged. As you bring the charged balloon near the foil ball on the top of the electroscope, the foil leaves on the electroscope gently swing toward the balloon. At some point, as you are moving the balloon near the electroscope, the foil leaves swing and move apart. Note that the electroscope is still neutral throughout this step of the activity. However, some of the electrons in the electroscope have moved to the end farthest from the balloon (the foil leaves). Therefore, the foil leaves repel each other. Some students may notice that if they move the balloon away from the electroscope, then towards the electroscope, the leaves repel when the balloon is farther from the electroscope.
Again, as you move the charged balloon toward the electroscope, the foil leaves repel even though the electroscope is neutral. When you touch the electroscope with the balloon, the foil leaves of the electroscope become negatively charged and repel because electrons move from the balloon to the electroscope. The leaves stay apart for a little while and then come back together as the electrons leak off the electroscope. If you bring a charged balloon back to touch the foil ball on the top of the electroscope, the leaves stay apart while you are touching the balloon to the electroscope.
To test whether an object is electrically charged, hold it near (or touch it) to the electroscope. If the object is charged, the leaves on the electroscope repel. This test does not indicate whether the object is positively or negatively charged.
You can give an object a greater charge by rubbing it longer.
Vocabulary/Definitions
electroscope: An instrument used to detect the presence of an electric charge by the mutual attraction or repulsion of metal foils.
Assessment
Pre-Activity Assessment
Voting: Ask a true/false question and have students vote by holding thumbs up for true and thumbs down for false. Count the votes, and write the totals on the board. Give the correct answers. Ask the students:
- True or False: A conductor is a material through which charge moves easily and an insulator is a material through which charge does not move easily. (Answer: True.)
- True or False: Like-charged objects attract, while unlike-charged objects repel. (Answer: False. The reverse is true.)
- True or False: When two items are rubbed together, electrons are transferred between them, leaving one negatively charged and the other positively charged. (Answer: True.)
Activity Embedded Assessment
Question/Answer: Ask the students and discuss as a class:
- What happened to the charge on the balloon when the balloon touched the electroscope? (Answer: The charge that built up on the balloon was transferred to the metal of the electroscope by contact.)
- Can the charge move on the balloon? (Answer: The charge cannot move on the balloon since it is an insulator. The charged part of the balloon must be touched to the electroscope.)
Worksheet / Pairs Check: Have students work individually or in pairs on the Charge Detector Graphing Worksheet. After students finish the worksheet, have them compare answers with a peer or another pair, giving all students time to finish the worksheet.
Post-Activity Assessment
Discussion Questions: Solicit, integrate and summarize student responses.
- In which parts of the electroscope do we use metal? Why do we use metal for these parts? (Answer: Metal is used to make the leaves, foil ball and wire connecting them because metal allows charges to move. Metal parts are used to show whether an object is charged.)
- In which parts of the electroscope do we use insulators? Why? (Answer: Insulators are used for the jar and the lid that holds the metal parts because insulators do not allow charges to move and we want to prevent the charge from leaking off the metal.)
- Why do we use such thin pieces of foil for the electroscope leaves? (Answer: Thick pieces of foil are heavier than thin pieces of foil. When they are charged to the same degree, thin pieces of foil move farther apart than thick pieces of metal. This makes it easier to see how much electrical repulsion exists between the foil leaves.)
- What purposes does the jar serve in building the electroscope? (Answer: The jar is the structure that holds the electroscope up, and it protects the delicate leaves from air currents that might knock them off.)
- After students have completed the Charge Detector Graphing Worksheet: What happens as the product of the charge increases in the first graph? (Answer: The force between the two objects increases.) What happens as you move the objects farther apart in the second graph? (Answer: The force between them decreases.)
Safety Issues
- To avoid burns, make sure students do not use hot water to remove the paper backing from candy wrappers.
- Ask students not to hold expanded (blown up) balloons next to their face or another student's face, in case the balloon pops.
Troubleshooting Tips
It is important to use the very thin and delicate silver metal foil from a candy bar for the electroscope leaves so you can notice any observable change when a charged balloon is brought near the detector. Using regular aluminum foil would create electroscope leaves that are too rigid and stiff to produce any observable movement.
The silver foil leaves of the electroscope are very delicate; students should be careful not to tear them.
For voting assessment, have students place their thumbs up or thumbs down under their chin to prevent their classmates from seeing their response. Another way to encourage safe answers is to have the students display one finger for "true" or no fingers for "false," also under their chin.
If students bump the table during this activity it will be difficult to see the movement in the electroscope leaves.
If the aluminum foil does not stay on the wire very well, use tape to secure it.
The electroscope will not be charged if students forget to touch the electroscope with the part of the balloon they rubbed. Since balloons are made of an insulating material, the charge is only on the location on the balloon that was rubbed.
Activity Extensions
Have students look on the Internet at animations that describe the movement of electrons when a charged object is brought near an electroscope.
As a variation, use different materials to build electroscopes. For example, a very simple electroscope can be made from tape. Balloons and pith balls (small balls made from plant matter) can also be used.
Activity Scaling
- For lower grades, have students work in groups on the graphing worksheet or complete the graphs together as a class.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

The purpose of this lesson is to teach students how a spacecraft gets from the surface of the Earth to Mars. Students first investigate rockets and how they are able to get us into space. Finally, the nature of an orbit is discussed as well as how orbits enable us to get from planet to planet — spec...

Students come to understand static electricity by learning about the nature of electric charge, and different methods for charging objects. In a hands-on activity, students induce an electrical charge on various objects, and experiment with electrical repulsion and attraction.

This lesson introduces the concept of electricity by asking students to imagine what their life would be like without electricity. Students learn that electrons can move between atoms, leaving atoms in a charged state.

Students are introduced to several key concepts of electronic circuits. They learn about some of the physics behind circuits, the key components in a circuit and their pervasiveness in our homes and everyday lives.
References
Pinelli, Thomas E. Making an Electroscope. April 15, 2004. The NASA SciFiles, Educators' Area.
Satellites Get a Charge out of Space, Marshall Space Flight Center, NASA.
Space Charge, Science @ NASA, accessed February 2004. https://science.nasa.gov/science-news/science-at-nasa/2001/ast13nov_1sidebar
Copyright
© 2004 by Regents of the University of Colorado.Contributors
Xochitl Zamora Thompson; Sabre Duren; Joe Friedrichsen; Daria Kotys-Schwartz; Malinda Schaefer Zarske; Denise CarlsonSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under a grant from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and National Science Foundation GK-12 grant no. 0338326. However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: October 2, 2020







User Comments & Tips