
Summary
Students come to make sense of the phenomena of static electricity as they use the science and engineering practices of asking questions and making observations to learn about the nature of electric charge and different methods for charging objects. In a hands-on activity, students induce an electrical charge on various objects, and experiment with electrical repulsion and attraction. They discover the disciplinary core ideas of energy and electric and magnetic forces while applying the crosscutting concepts of energy transfer and cause and effect relationships.Engineering Connection
Engineers are so creative: Would you ever think that the air we breathe could be cleaned by using the behavior of electrical charges? Engineers who fully understand the scientific principles of static electricity have invented many modern products and industrial processes that help our environment. For example, different types of electrostatic precipitators clean both factory and home air, and electronic paper can be used for communication and re-used over and over again.
Learning Objectives
After this lesson, students should be able to:
- Understand the basic properties of electric charge.
- Describe technologies associated with static electricity.
- Explain the transfer of electrons between two objects.
- Compare different objects based on their electric charge.
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
3-PS2-3. Ask questions to determine cause and effect relationships of electric or magnetic interactions between two objects not in contact with each other. (Grade 3) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This lesson focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Ask questions that can be investigated based on patterns such as cause and effect relationships. Alignment agreement: | Electric, and magnetic forces between a pair of objects do not require that the objects be in contact. The sizes of the forces in each situation depend on the properties of the objects and their distances apart and, for forces between two magnets, on their orientation relative to each other. Alignment agreement: | Cause and effect relationships are routinely identified, tested, and used to explain change. Alignment agreement: |
| NGSS Performance Expectation | ||
|---|---|---|
|
4-PS3-2. Make observations to provide evidence that energy can be transferred from place to place by sound, light, heat, and electric currents. (Grade 4) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This lesson focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Make observations to produce data to serve as the basis for evidence for an explanation of a phenomenon or test a design solution. Alignment agreement: | Energy can be moved from place to place by moving objects or through sound, light, or electric currents. Alignment agreement: Energy is present whenever there are moving objects, sound, light, or heat. When objects collide, energy can be transferred from one object to another, thereby changing their motion. In such collisions, some energy is typically also transferred to the surrounding air; as a result, the air gets heated and sound is produced.Alignment agreement: Light also transfers energy from place to place.Alignment agreement: Energy can also be transferred from place to place by electric currents, which can then be used locally to produce motion, sound, heat, or light. The currents may have been produced to begin with by transforming the energy of motion into electrical energy.Alignment agreement: | Energy can be transferred in various ways and between objects. Alignment agreement: |
International Technology and Engineering Educators Association - Technology
-
Explain how various relationships can exist between technology and engineering and other content areas.
(Grades
3 -
5)
More Details
Do you agree with this alignment?
State Standards
Colorado - Science
-
Show that electricity in circuits requires a complete loop through which current can pass
(Grade
4)
More Details
Do you agree with this alignment?
Worksheets and Attachments
Visit [www.teachengineering.org/lessons/view/cub_electricity_lesson02] to print or download.Pre-Req Knowledge
atoms, electrons, electric charge
Introduction/Motivation
Have a student volunteer rub a balloon on a sweater or piece of wool and then hold the balloon close to their hair. Observe that the student's hair stands on end.
Challenge the rest of the students in the class to explain why this happens using the information from the Teach Engineering Lesson Lights Out! regarding the movement of electrons and charged objects. Ask the students: Why do you think that your hair stands up after rubbing it with a balloon? (Answer: Electrons move from your hair to the balloon, causing each of the hairs to have the same positive charge. Since objects with the same charge repel each other, the hairs try to get as far from each other as possible. The farthest they can get is by standing up and away from the others.)
Next, ask the students for examples from their own experiences of the buildup or discharge of static electricity. (Possible answers: Walking across the carpet [buildup] and touching a door knob [discharge].) Students can conduct their own experiment with the associated activity Charge It! All About Electrical Attraction and Repulsion.
Lesson Background and Concepts for Teachers
Electric Charge Basics
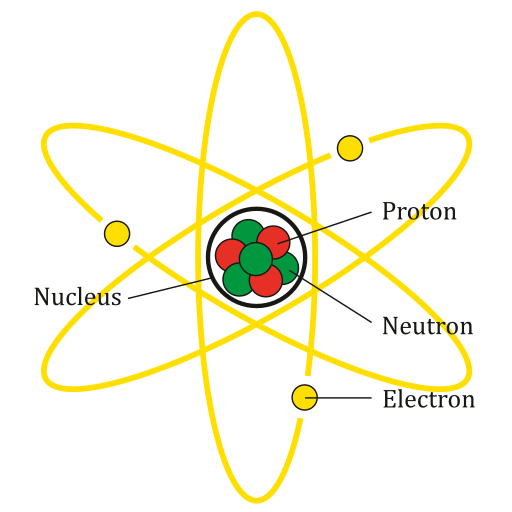
Every atom is made of negatively-charged electrons surrounding a positively-charged nucleus. The nucleus contains protons, which are positively charged, and neutrons, which are neutral (they have no net electric charge), as illustrated in Figure 3. Electrons can move from one atom, molecule or material, to another. Most objects do not have an electric charge because there is a balance of electrons and protons in the material that makes up the object. In certain circumstances, there can be an imbalance of protons and electrons. An object with a greater number of electrons than protons is negatively charged. An object with more protons than electrons is positively charged.
Conductors and Insulators
Materials can be classified by how well they conduct electricity. Insulators do not conduct very well, while conductors do. In insulators, every electron is attached to a particular atom. However, in good conductors, some electrons are more loosely bound to the atom and can move about in the material. Metals are good conductors, while materials such as wood, glass, plastics and rubber are good insulators. Electrical and mechanical engineers use their understanding of the differences in material properties when they design the safe appliances and machines we use every day, such as toasters, coffee makers, hair dryers, stoves and extension cords. Conduct the fun and hands-on associated activity Build a Charge Detector to develop students' understanding of different methods of charging, and the differences between conductors and insulators.
Charging by Friction
When two materials are rubbed together, some electrons may be transferred from one material to the other, leaving them both with a net electric charge. The material that lost electrons becomes positively charged, while the material that gained electrons becomes negatively charged. Both insulators and conductors can gain a net charge in this way. This is how clothing gets charged in the clothes dryer, or our bodies get charged when we walk across a carpeted floor.
Charging by Contact
We can also charge a neutral object (either a conductor or insulator) by just touching it to a charged object. Electrons move from a negatively-charged object to a neutral object, leaving the neutral object with a negative charge. Electrons also move from a neutral object to a positively-charged object, such that the originally neutral object gains a net positive charge. The final charge on the originally-charged object depends on a number of factors, and is not important for what we are studying today. Charging by close proximity is another way to give a neutral conductor a net charge. However, it requires two steps and will not be covered in this lesson.
Excess charge on an object may slowly leak off the object (especially if it is very humid), or the excess charge may produce an electric spark. If a spark is created, electrons move off one object and onto another. During the spark, energy is also released in the form of light, heat and sound. The buildup of excess charge on an insulated body and the resulting spark (or electric discharge) caused by this buildup, are both properties of static electricity.
NOTE: Since in this lesson we explore the attractive force between unlike charges and the repulsion between like charges, some students might have questions about the apparent contradiction of the arrangement of the atom. Students may ask why the positively-charged protons do not repel each other and fly out of the nucleus. The protons in the nucleus are held together in spite of their electrical repulsion because of a stronger nuclear force that works at the atomic scale. Students may also wonder how the electrons keep from spinning into the nucleus. The reasons for this are more complicated; it would probably be best to say that it is similar to why the moon is not pulled into the Earth; in fact, the moon is held in orbit by the Earth.
Electronic Paper
Engineers used the principles of static electricity to develop electronic paper. Someday, paper may be obsolete due to the use of electronic paper technology. Newspapers, bestsellers and textbooks may be printed on electronic paper. This technology could greatly reduce paper production and consequently reduce the forest destruction that results from manufacturing large quantities of paper that our society uses. Electronic paper technology is based on the attractive and repulsive forces between charged objects. Engineers developed electronic paper technology and designed the manufacturing processes that produce electronic paper. Electronic paper is already being used for billboards and signs in stores. Electronic "paper" is thin and flexible like traditional paper, but can be erased and reused over and over again like a computer display.
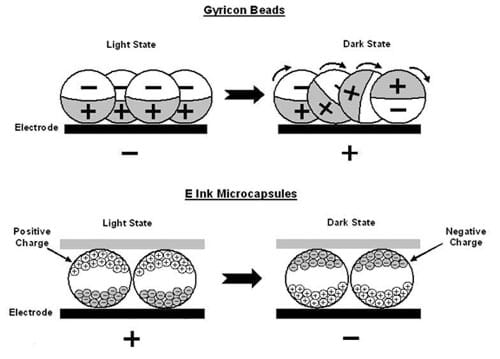
How does electronic paper work? There are two types (see Figure 3). In one product, the "ink" consists of tiny beads that are black on one side (positively charged) and white on the other (negatively charged). In another type of electronic paper, the "ink" is made of microcapsules containing blue (negatively charged) and white (positively charged) dye particles. In both products, the surface under the layer of ink has tiny electrodes that can be either positively- or negatively-charged. When a portion of the surface is charged, the ink experiences an electric force. If the electrode is positively charged, the positive side of a bead or the white dye particles in the microcapsule are repelled. At the same time, the negative side or the blue dye particles in the microcapsule are attracted to the electrode. This produces a different effect for each type of ink. The image stays the same until the charge on the electrodes is changed.
Additional Technologies
Scientists and engineers use their understanding of static electricity to develop industrial air filters to help our environment. These "electrostatic precipitators" use static electricity to remove pollutants without impeding the production efficiency of an industrial plant. Power plants, steel mills and paper plants use electrostatic precipitators to remove the harmful particles generated in the manufacturing process before they can pollute our air.
Electrostatic air cleaners for the home use an electrostatic force to move air molecules and trap small airborne particles (.05 – 30 microns in size, such as pet dander or other allergens) as they circulate past an array of electrically-charged stainless steel blades. One example is the Ionic Breeze by Zenion Effect Technology.
Other technologies that exploit the properties of static electricity may be found in appliances and machines such as copy machines and printers designed by electrical and mechanical engineers.
Associated Activities
- Charge It! All About Electrical Attraction and Repulsion - In this hands-on activity focusing on static electricity and the interactions of charged objects, students charge objects and observe electrical attraction and repulsion.
- Build a Charge Detector - In this hands-on activity focusing on static electricity and the interactions of charged objects, students build an electroscope to develop their understanding of different methods of charging, and the differences between conductors and insulators.
Lesson Closure
Ask the students to explain how an object can become electrically charged. Have the students make a list of conductors and insulators on the board. (Possible answers: Insulators could be wood, plastic, Styrofoam. Conductors could be metals.)
Vocabulary/Definitions
atom: The smallest unit of an element, consisting of a dense, positively-charged nucleus surrounded by electrons.
conductor: A material through which charge moves easily.
electrode: A solid conductor through which a current enters or leaves a medium.
electron: The negatively-charged particle of an atom.
Insulator: A material through which charge does not move easily.
ion: An atom, group of atoms or molecule that has a net electric charge.
molecule: The smallest particle of a substance that retains the chemical and physical properties of the substance, and is composed of two or more atoms.
neutron: The neutral particle in an atom.
nucleus: The positively-charged central region of an atom composed of neutrons and protons.
proton: The positively-charged particle of an atom.
static electricity: The buildup of charge on an object, or the spark that is a result of a buildup of charge on an object.
Assessment
Pre-Lesson Assessment
Discussion Questions: Solicit, integrate and summarize student responses.
- Why do you think that your hair stands up after rubbing it with a balloon? (Answer: Electrons move from your hair to the balloon. Then, each of the hairs has the same positive charge. Since objects with the same charge repel each other, the hairs try to get as far from each other as possible, which is by standing up and away from the other hairs.)
- What are some examples, from your own experiences, of the buildup or discharge of static electricity? (Possible answers: Walking across the carpet [buildup] and touching a door knob [discharge].)
Post-Introduction Assessment
Brainstorming: As a class, have the students engage in open discussion. Remind students that in brainstorming, no idea or suggestion is "silly." All ideas should be respectfully heard. Encourage wild ideas and discourage criticism of ideas. Have them raise their hands to respond. Write ideas on the board. Ask the students:
- Engineers used the principles of static electricity to develop electronic paper. Where could we use electronic paper in this classroom? (Possible ideas: Textbooks, bulletin board, notebook paper, report cards, etc.)
Lesson Summary Assessment
Bingo: Provide each student with a list of the lesson vocabulary terms. Have each student walk around the room and find a student who can define one vocabulary term. Students must find a different student for each word. When a student has all terms completed they shout "Bingo!" Continue until most students have a bingo. Ask the students who shouted "Bingo!" to give definitions of the vocabulary terms.
Homework Worksheet: Assess students' understanding of the lesson by assigning the Take Charge! Homework Worksheet as a take-home quiz. See answers in the Take Charge! Homework Worksheet Answers.
Lesson Extension Activities
Electronic Paper Message Activity: Have students explore electronic paper technology by conducting an activity in which they spell out words using the plus and minus charges on a graph paper grid.
Explore the Van de Graaf generator. Show the students the photograph at the beginning of this lesson, discuss why the person's hair is standing on end. The young person is touching a Van de Graaf generator, which is used to generate static electricity. Developed by an MIT professor, Dr. Robert J. Van de Graaf, they were originally used as research tools in atom-smashing experiments, but now are mainly used to teach people about static electricity and lightning. Note that the person must be standing on an insulator to prevent charge from moving through her body and going into the Earth. Also, they must be very careful not to touch anyone because that would give them a very unpleasant electric shock. Ask students to research how the Van de Graaf generator works. What type of charging occurs in a Van de Graaf generator?
Have students research the history of photocopying. American law student Chester Carlson was a research engineer and a patent attorney who invented the method of xerography — a copy process based on electrostatic energy. Students can create a timeline of the changes in photocopying technology since Chester's time.
Introduce the students to accomplished science researchers such as Marietta Blau (1894-1970). She was an Austrian physicist and pioneer in the study of atomic particles, and the first person to develop methods for identifying both protons and neutrons in experiments, using photographic chemicals. Because she was Jewish, Marietta Blau fled from Austria before the Anschluss. In the U.S., she continued her research in industry and at universities, including Brookhaven National Laboratories, Columbia University and the University of Miami. She was also a professor at the Technical University of Mexico City. Several Nobel Prize-winning scientists nominated Marietta Blau for the Nobel Prize in Physics.
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

This lesson introduces the concept of electricity by asking students to imagine what their life would be like without electricity. Students learn that electrons can move between atoms, leaving atoms in a charged state.

Students explore the static electricity through hands-on activities. They attract O-shaped cereal pieces to charged combs and watch the cereal jump away when it touches the comb. They also observe Styrofoam pellets pulling towards a charged comb, then leaping back to the table.

Students are introduced to the concept of electricity by identifying it as an unseen, but pervasive and important presence in their lives. They compare conductors and insulators based on their capabilities for electron flow. Then water and electrical systems are compared as an analogy to electrical ...

Students are introduced to the fundamental concepts of electricity. They address questions such as "How is electricity generated?" and "How is it used in every-day life?" Illustrative examples of circuit diagrams are used to help explain how electricity flows.
References
Ditlea, Steve. "The Electronic Paper Chase." Scientific American. November 2001 issue.
Electrical Engineering for Pollution Control, Electrostatic Precipitator for Power Plants, ASU Electrical Engineering.
Electronic ink technology, E Ink Corporation.
Galison, Peter L. "Marietta Blau: Between Nazis and Nuclei." Physics Today. Vol. 50. Issue 42 1997.
Hewitt, Paul G. Conceptual Physics. 8th Edition. New York, NY: Addison Publishing Company, 1998.
Patsuris, Penelope. "Marketing Messages Made to Order." Forbes, August 27, 2003.
Copyright
© 2004 by Regents of the University of Colorado.Contributors
Xochitl Zamora Thompson; Sabre Duren; Joe Friedrichsen; Daria Kotys-Schwartz; Malinda Schaefer Zarske; Denise CarlsonSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under a grant from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and National Science Foundation GK-12 grant no. 0338326. However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: June 5, 2026







User Comments & Tips