Quick Look
Grade Level: 5 (3-5)
Time Required: 45 minutes
Lesson Dependency: None
NGSS Performance Expectations:

| 5-ESS3-1 |

Summary
Students are introduced to the concepts of air pollution and technologies that engineers have developed to reduce air pollution. They develop an understanding of visible air pollutants with an incomplete combustion demonstration, a "smog in a jar" demonstration, construction of simple particulate matter collectors and by exploring engineering roles related to air pollution. Next, students develop awareness and understanding of the daily air quality and trends in air quality using the air quality index (AQI) listed in the newspaper or online. Finally, students build and observe a variety of simple models in order to develop an understanding of how engineers use these technologies to clean up and prevent air pollution.Engineering Connection
It is an engineering challenge to find solutions to clean our air, because so many factors contribute to air pollution. Engineers investigate the composition and characteristics of the air in a specific area before they start designing solutions for that region. Some engineering technologies currently used to clean industrial and factory air include: scrubbers, electrostatic precipitators, cyclones and baghouses.
Learning Objectives
After this lesson, student should be able to:
- Identify some of the main causes and sources of air pollution.
- Explain the affects of air pollution on humans and their environment.
- Identify some ways to reduce air pollution.
- Describe how engineers create technology to help industry clean up air pollution
Educational Standards
Each Teach Engineering lesson or activity is correlated to one or more K-12 science,
technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN),
a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics;
within type by subtype, then by grade, etc.
Each Teach Engineering lesson or activity is correlated to one or more K-12 science, technology, engineering or math (STEM) educational standards.
All 100,000+ K-12 STEM standards covered in Teach Engineering are collected, maintained and packaged by the Achievement Standards Network (ASN), a project of D2L (www.achievementstandards.org).
In the ASN, standards are hierarchically structured: first by source; e.g., by state; within source by type; e.g., science or mathematics; within type by subtype, then by grade, etc.
NGSS: Next Generation Science Standards - Science
| NGSS Performance Expectation | ||
|---|---|---|
|
5-ESS3-1. Obtain and combine information about ways individual communities use science ideas to protect the Earth's resources and environment. (Grade 5) Do you agree with this alignment? |
||
| Click to view other curriculum aligned to this Performance Expectation | ||
| This lesson focuses on the following Three Dimensional Learning aspects of NGSS: | ||
| Science & Engineering Practices | Disciplinary Core Ideas | Crosscutting Concepts |
| Obtain and combine information from books and/or other reliable media to explain phenomena or solutions to a design problem. Alignment agreement: | Human activities in agriculture, industry, and everyday life have had major effects on the land, vegetation, streams, ocean, air, and even outer space. But individuals and communities are doing things to help protect Earth's resources and environments. Alignment agreement: | A system can be described in terms of its components and their interactions. Alignment agreement: Science findings are limited to questions that can be answered with empirical evidence.Alignment agreement: |
International Technology and Engineering Educators Association - Technology
-
The use of technology affects the environment in good and bad ways.
(Grades
3 -
5)
More Details
Do you agree with this alignment?
State Standards
Colorado - Science
-
Analyze and interpret a variety of data to understand the origin, utilization, and concerns associated with natural resources
(Grade
5)
More Details
Do you agree with this alignment?
Worksheets and Attachments
Visit [www.teachengineering.org/lessons/view/cub_environ_lesson07] to print or download.Introduction/Motivation
Long ago people were not worried about clean air, because there seemed to be an endless supply of it. Today, many more people live in our world, and we are rapidly polluting this essential resource. We need to find better ways to protect and improve our air.
Most of the time, we hardly notice the air around us; usually, we cannot see it or taste it. Air does not generally smell, but it carries substances that we do smell. However, we can feel air when it moves, and we can see the effects of air on our surroundings. When air moves, it has great power (for example, to push sailboats, drive windmills and move clouds) and when it is compressed (that is, squashed into a small space), it has great strength (for example, air in a tire supports a vehicle and helps a helicopter to rise into the air).
Clean air contains only the gases and water vapor needed to keep the Earth's environment healthy. Pollutants are substances, or even energy, that harm living — and some non-living — things. A high concentration of pollutants in the air is called air pollution.
Air pollution can destroy our environment and can cause humans and other living things to become sick. Sometimes, air pollution can cause rashes, eye/nose irritation, headaches, sleepiness, coughing, sneezing and dizziness. If you breathe in too much air pollution, of a very high concentration, it can cause severe illnesses, such as cancer, asthma, kidney failure, liver damage and even birth defects. Air pollution affects the plants and animals in our environment as well as destroying buildings.
What causes air pollution? Many things. Specific outdoor sources of pollution include car exhaust, residential fireplaces, power plants, and factories. Indoor sources of pollution include cleaning supplies, cigarette smoke, furnaces and outdoor pollutants brought inside.
But can we live without air? No! We must have air to survive. Because of this, engineers are working diligently to try to solve our air pollution problems. There are many things that complicate these solutions, so engineers that work on air pollution problems must have a good understanding of the composition and characteristics of air before beginning to design technology solutions for air pollution. Some of the technologies that engineers have designed to clean up air pollution include a scrubber, an electrostatic precipitator, a cyclone and a baghouse. Today, we are going to look at what air pollution is and how engineers try to clean it up.
Lesson Background and Concepts for Teachers
What are Air Pollutants?
Literally hundreds of pollutants float around at any given time in the air that we breathe. Some of these pollutants come from natural sources, but most come from human activity. Air pollution consists of the gases and particles that affect the quality of the air around us, are harmful to the environment, and/or enters the atmosphere at highly concentrated levels. (For example, oxygen and pollen are often present in the air, but are not considered pollution.) Air pollution generally consists of: visible gases, invisible gases and particulates (such as, soot), as well as fibers, mists, molds and bacteria. These particulates can be found both outdoors and indoors. Most major air pollutants are invisible, but large amounts of them can be seen as smog when they are concentrated in areas such as cities. Because visible pollutants (such as particulate matter and smog) are more obvious, we understand more about them and tend to have more regulations focusing on them than we do on invisible pollutants. Students can expand their understanding of what air pollutants are with the associated activity: What Color is Your Air Today?
Particulate Matter
Particulate matter is very small particles of solid matter and/or droplets of liquid. It includes things that have been burned, ash, smoke, soot, dust, pollen, soil or even chemicals — basically, anything that floats in the air. Particulate matter is harmful to animals that breathe it in and to the surfaces of buildings and other structures that are exposed to it for long periods of time or large concentrations.
Smog
Smog is a type of visible air pollution created from smoke, fog, suspended particles and chemical fumes. The word "smog" comes from the combination of the words "smoke" and "fog." It was first used in the early 1900s to describe the combination of smoke and thick fog that sometimes hung over London, England. Weather conditions, such as lack of wind can cause smog to build up. Smog is especially obvious during a thermal inversion: when an upper layer of warm air traps the pollutants over a lower layer of cool air, preventing normal atmospheric circulation (the smog cannot rise and scatter). Mountain ranges near cities can also trap smog in an area.
Sources of Air Pollution (Outdoor/Indoor)
Human-Made Outdoor Sources
The burning of fossil fuels is one of the main causes of air pollution. Smoke and fumes containing carbon dioxide and sulfur dioxide from power stations and factories are the worst offenders. Car exhaust is another large source of air pollution. It contains both invisible gases (carbon dioxide, carbon monoxide, hydrocarbons, and nitrogen oxides) and particles. Many consumer products (hair spray, paint and cleaners) release high levels of volatile organic compounds (VOCs) into the atmosphere. Other important sources include gas stations, industry (many types), agriculture and forestry.
Natural Outdoor Sources
Volcanic eruptions often spew gases and ash into the atmosphere. Forest fires also release dust and smoke into the air.
Indoor Sources
Indoor air pollution is becoming a large concern of environmental engineers because the majority of the public's time is spent indoors. Indoor air (IA) pollutants can be created within the building or drawn inside from outdoor sources. Indoor sources include: foundations, equipment (malfunctioning HVAC systems, emissions from office equipment/labs), furnishings (new carpet that releases formaldehyde and burnt Teflon pans), dust-producing or water-damaged materials, unsanitary trash emissions, insects and pests (pesticides), food preparation areas, fungi, cleaning materials, pets (dander), and people (smoking). Some of the most common outdoor air pollutants that are brought inside are: smog, bacteria and mold spores, vehicle exhaust, exhaust fumes, pollen and dust, algae (from standing water) and smoke.
Other Sources
Some people also consider sound pollution a type of air pollution, because sound waves travel through the air. Outdoor sources include loud jets, construction equipment, huge trucks and other forms of transportation, etc. Indoor sources include music, TVs, mixers, dishwashers, washing machines, etc. Lights are also considered a source of atmospheric pollution to astronomers.
How Do Air Pollutants Affect Us?
The cumulative effect of air pollution destroys our environment and poses health threats to humans and other living organisms. Figure 1 shows the US Environmental Protection Agency's air quality index, which is used to classify the quality of the air (in terms of level of pollution). Air pollutants may cause a variety of adverse human health effects ranging from more minor effects (rashes, eye/nose irritation, headaches, fatigue, shortness of breath, coughing, sneezing and dizziness) to major effects (cancer, breathing complications, such as asthma, kidney failure, liver damage and birth defects). According to the air quality index, people who are sensitive to polluted air would want to avoid going outdoors on orange, red or purple days (purple days are very rare in most areas).

The environmental effects of air pollution are numerous. For example, sulfur dioxide emissions combine with water particles and fall to Earth in the form of acid rain or snow. This destroys the food and nutrients for fish and other aquatic life in oceans, lakes and streams. It damages trees, other plants and even some buildings. CFCs transform and destroy the ozone layer. This increases the Earth's exposure to ultraviolet radiation and can cause crop failures, skin cancer and other environmental/health disasters.
What are Air Pollutant Solutions?
The water cycle usually cleans air pollutants naturally. Currently, however, two problems exist: the water cycle is unable to clean contaminants as quickly as they are added, and high concentrations of pollutants contribute to acid rain.
Our government is attempting to help control air pollution by passing laws, such as the Clean Air Act. The government also passes strict legislation to control the amount of by-products released by industrial companies and modes of transportation. These air quality standards are designed to protect the health and welfare of people, plants and animals, and to protect buildings, monuments, water resources, etc.
One of the largest problems environmental engineers face is creating new techniques to prevent additional air pollution.
Many changes have occurred as a result of the passing of new environmental laws. For example, many places used to burn coal as a means of heating or running machinery. Now, we know that burning coal (and other fossil fuels) causes air pollution; now many people do not burn coal as a result of public awareness and stricter air emissions laws. Another example is with the engineering of automobiles. Generally, most cars still burn gasoline (a fossil fuel), which, as we know, is a cause of air pollution. New laws have changed how cars are built. In the last few decades, major improvements in motor vehicle technology have led to exhaust emission reductions of up to 96% compared to vehicles from 1960. In the mid-1970s, laws were passed to phase out lead as an additive in gasoline. In the 1980s, regulations were passed to limit gasoline evaporation. Recently, fuel refiners have created reformulated gasoline (RFG), with additives called oxygenates which help to reduce harmful emissions by burning the gasoline more completely. RFG is a cleaner-burning form of gasoline and reduces the amount of pollutants emitted into the air through the tailpipe and via evaporation from the gas tank.
Adjustments can also be made on a more personal level to help improve the air. For example, reducing fuel use by joining a car pool, taking the bus/train, riding a bike or walking, reducing the energy we use, and buying environmentally friendly products, like non-aerosol hair spray and non-toxic cleansers will all help in the efforts to reduce air pollution. Students can learn more about preventing air pollution with the associated activities: Washing Air: Wet Scrubber Pollutant Recovery Method and Cleaning Air with Balloons.
Interesting Air Pollution Facts
- Mexico City is one of the world's most polluted cities. When the air pollution is at its worst, birds have been known to drop dead out of the sky.
- Every day, each of the world's 1.4 billion cattle gives off 0.61 pounds of methane (produced in their gut as they digest food). That means 312 billion pounds (156 million tons) of methane is produced each year! Methane is also produced in large quantities by rotting vegetation in landfills.
- The American Lung Association believes that sulfur-dioxide exposure (one source is the internal combustion engine of autos) is a leading cause of lung disease following smoking. It has also been implicated in the rising occurrence of asthma, bronchitis and emphysema.
- The average person takes about 20,000 breaths a day.
- Plants give us oxygen, and yet people have destroyed almost two-thirds of the original forests on Earth.
- Sometimes Hawaii has "vog" alerts. These are times when volcanically produced smog is at dangerous levels.
Associated Activities
- What Color is Your Air Today? - Students develop awareness and understanding of daily air quality by looking at the Air Quality Index in the newspaper and exploring what engineers can do to help improve poor air quality.
- Cleaning Air with Balloons - Students observe and discuss a simple balloon model of an electrostatic precipitator to better understand how this pollutant recovery method functions in cleaning industrial air pollution.
- Washing Air: Wet Scrubber Pollutant Recovery Method - Students observe and discuss a simple model of a wet scrubber to understand how this pollutant recovery method functions in cleaning industrial air pollution.
Lesson Closure
Imagine what the world will be like if we do not develop some solutions to our air pollution problems. Even today, if people travel to places where there is not as much air (the top of a mountain or underwater) or no air at all (space) they have to take air with them. This could become an everyday experience if we continue to neglect and mistreat this essential natural resource. During this lesson, we have only considered industrial changes and technologies for controlling air pollution. What do you think we can do in our own lives to control/prevent the amount of air pollution that we cause? (Possible answers: Walking more, joining a car pool, taking the bus/train, riding a bike, reducing the amount of energy we use, turning lights off, and buying environmentally friendly products, like non-aerosol hair spray and non-toxic cleansers.)
Vocabulary/Definitions
air: The mixture of invisible odorless tasteless gases (as nitrogen and oxygen) that surrounds the earth.
air pollution: The gases and particles that affect the quality of the air around us, which are harmful to the environment, and/or which enter the atmosphere at highly concentrated levels. Generally consists of: visible gases, invisible gases, and particulates (like soot).
air quality index: A system to describe how clean or dirty the air is based on five main pollutants. Abbreviated as AQU. Also called the pollution standards index.
allergy: A physical reaction (sneezing, coughing, etc.) to something in the air (such as pollen, dust, pollution, etc.).
atmosphere: The layer of air that surrounds Earth like a blanket. It keeps the Earth warm and protects it from harmful radiation.
baghouse: A large filter that works very much like a giant vacuum. Heavy dirt particles fall out and are removed; smaller particulates are trapped on filters (bags) made of cloth, paper or similar materials. Particles are shaken or blown from the filters into a collection hopper. Industries often use baghouses to help decrease (and prevent) air pollution.
carbon dioxide: A colorless, odorless gas formed during breathing, combustion and decaying that adds to the greenhouse effect. Abbreviated as CO2.
carbon monoxide: A colorless, odorless gas made of one carbon atom and one oxygen atom. Abbreviated as CO.
catalytic converter: An emission-control device that changes hydrocarbons and carbon monoxide in automobile exhaust into carbon dioxide and water vapor.
cyclone: A device that provides a low-cost method for controlling particulate pollutants. Dust-laden gas is whirled very rapidly inside a collector shaped like a cylinder. The swirling motion creates inertia that causes the dust particles to leave the circular path of the wind, collect against the wall of the cyclone, and fall into a collection hopper, where they are properly disposed.
dioxin: A highly-toxic family of chemicals used to make poisonous substances, such as weed killers and bleaching products (like for paper). It is poisonous at 1 ppb (part per billion).
electrostatic precipitator: A device for controlling particulate pollutants. A static charge makes dirt particles stick to electrified plates (much the same way that static electricity in clothing attracts small bits of dust and lint). The dirt is knocked loose, collected and removed.
emissions: Gases or particles that are given off when fossil fuels are burned.
fossil fuel: Something that forms naturally, underground from the remains of ancient organisms (plants and animals) from millions of years ago. Coal is an example of a fossil fuel.
global warming: Described as an increase in average temperatures all over the world. Some people think this is caused by an increase in the greenhouse effect due to increased greenhouse gases in the atmosphere.
greenhouse effect: This occurs when gases in the atmosphere, such as carbon dioxide, trap heat in the atmosphere. (Usually just the right amount of heat is trapped to support life on Earth.)
hydrocarbon: An organic compound containing atoms of hydrogen and carbon.
industrial smog: Air that contains sulfur oxide and solid particles that are released when fossil fuels are burned. It appears as a grayish haze and usually forms over heavily industrialized cities.
nitrogen oxide: A gas that forms when nitrogen and oxygen in the atmosphere are burned with fossil fuels at high temperatures.
oxygen: A colorless, tasteless and odorless gaseous element that constitutes 21% of the atmosphere.
ozone: Ground-level ozone is the principal component of smog, created from chemical reactions of pollutants: VOCs, and NOx. Abbreviated as O3.
particulate matter: Very small particles of solid matter and/or droplets of liquid. It includes things that have been burned, ash, smoke, soot, dust, pollen, soil or even chemicals — basically, anything that floats in the air.
photochemical smog: A condition that occurs when strong sunlight reacts with pollution (formaldehyde, ozone and pollutants from cars and power plants). It appears as a brownish-orange haze and usually appears over large cities.
pollutant: A harmful chemical or waste material discharged into the water or atmosphere. Some pollutants are visible and some are invisible.
reformulated gasoline: A cleaner-burning form of gasoline that reduces the amount of pollutants emitted into the air through an automobile's tailpipe and via evaporation from the gas tank. Oxygenates are a principal component. Abbreviated as RFG.
scrubber: A particulate pollution-reduction device in which solid particles and gases are trapped as they pass through a fine water mist. Sometimes the mist is injected with limestone powder to help extract the dirt particles. A drawback of this device is the production of wastewater.
smog: A combination of smoke and fog in the air.
thermal inversion: When an upper layer of warm air traps the pollutants over a lower layer of cool air, preventing normal atmospheric circulation.
visible pollutant: Air pollution that can be seen with the naked eye.
volatile organic compound: An organic chemical containing carbon that escapes into the air easily. Abbreviated as VOC. Many VOCs are also hazardous air pollutants that can cause serious illnesses including cancer. Carbon is the basic chemical element found in living beings; carbon-containing chemicals are called organic. Example VOCs include some hairspray, cleaners, air fresheners and paints.
Assessment
Pre-Lesson Assessment
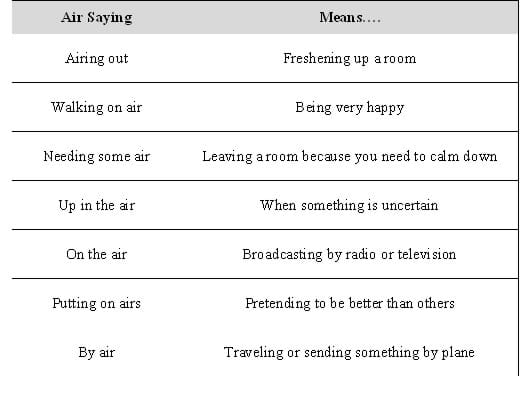
Human Matching: Print out the air sayings and meanings (see Table 1 or the Air Sayings attachment), and cut apart each of the boxes. You may want to add more of your own before doing this. At the end, give a brief explanation of concepts.
There are many ways to conduct this pre-lesson assessment activity:
- As students enter the classroom, hand each one strip (either a saying or a meaning). Tell students that some of them have received strips with sayings that make reference to air, and that other students have received descriptions of what one air saying means. (Note: You may want to use one of them as an example.) Ask students to find their "partner." Each pair should consist of a matching saying and meaning. After students have all found partners, ask them to read their sayings and meanings aloud to the class.
- Give students a handout and have them match the sayings to the meanings.
- Write the "air sayings" on the board or an overhead transparency and ask students to describe what they mean.
- Ask for volunteers from the class to come up to the front of the room. Give each a piece of paper. Have volunteers read what is written on their papers, one at a time. Have students match term to definition by voting. Have student "terms" stand by their "definitions."
Post-Introduction Assessment
Discussion Questions: Ask the students and discuss as a class:
- Does the quality of our air matter? Is clean air important? (Answer: Yes, polluted air can cause serious health problems, such as cancer, lung disease and headaches, among others, for people and is hazardous for animals/plants who inhabit Earth.)
- How does air get dirty? What is air pollution and where does it come from (sources)? (Answer: It is caused by several things that people do, especially burning fossil fuels when driving automobiles or operating power plants.)
- What can happen if you breathe polluted air? (Answer: It can cause serious health effects on humans and other life on Earth, such as itchy eyes, coughing, and change in or destruction of our buildings, etc.)
- How can engineers help control the amount of air pollution in the air? (Answer: Engineers develop technologies to help reduce the emission of and remove pollutants from air.)
Chart Reading: Give each student the Sources of Air Pollution Handout. Briefly discuss the contents. Ask students to keep these for reference during the lesson.
Lesson Summary Assessment
Flashcards: Have students create flashcards of the air pollution vocabulary terms using the Air Pollution Flashcards attachment. (Note: You need one set of flashcards per student. The cards are set up so that you can print two-sided copies on your printer: the odd pages are the terms and the even pages are the definitions. If you cannot print double-sided automatically, print the odd pages first, and then print the even pages on the back side of each odd page. Make sure you put the odd pages back in the printer in the right order and the correct way.) It may help to copy them onto a heavy weight card stock.
- Give each student a set of flashcards.
- Ask them to find pictures or draw pictures to represent each term and place it on the term side of the flashcard.
- Have students cut out their flashcards.
- Have students get in pairs and have them quiz each other. (It is more challenging if they guess using their partner's flashcards.)
Homework
Worksheet: Assign the Clean Air Act Worksheet as homework. Ask students to pretend to be engineers trying to pass a new bill in Congress. They should use specific information that they have learned in this lesson. Review the worksheets during the next class period.
Lesson Extension Activities
Find our about factories in your area. What do they make? What types of waste do they produce? How do they dispose of the waste? Check them out on the EPA website.
Invite an employee from the EPA (or other local person responsible for monitoring air quality) to your classroom. Encourage students to ask questions about the types, causes, and levels of air pollution found in your community.
Read The Wump World (by Bill Peet, Houghton Mifflin, 1981). Discuss the examples and effects of air pollution. Ask students to write their own versions of this book using what they have learned about air pollution.
Have students research the Clean Air Act. What is it? What is the goal? When was it started?
Have students compare the U.S. to other countries in terms of air quality and the regulations for controlling air quality.
Have students research and explore how lichens can indicate levels of air pollution. For an introduction to the topic see 175 Science Experiments to Amuse and Amaze Your Friends (by Brenda Walpole, Random House Books for Young Readers, 1988, p. 85).
Read and discuss the "Automobiles and Air Pollution" Reading Handout.
It's a Problem All over the World! Imagine that you are at a meeting of great leaders from all over the world. They are waiting to hear you speak about what is happening to our fresh air. You want to convince more of them to use the new technologies that you have just studied. What will you tell them? How will you get them to work together? Write down your ideas and make a presentation to the class.
Project A.I.R.E. has developed student-appropriate reading on the topic of air pollution. Have students visit: https://www.epa.gov/students/lesson-plans-teacher-guides-and-online-resources-educators for some information to share with their classmates.
Check out readings and quizzes at "Air Quality Days" at https://www.eekwi.org/engage/outdoor-safety/air-quality-days
Students can investigate how they can improve their school's indoor air quality. The EPA offers a free Indoor Air Quality Tools for Schools Kit at: https://www.epa.gov/iaq-schools/indoor-air-quality-tools-schools-action-kit
Subscribe
Get the inside scoop on all things Teach Engineering such as new site features, curriculum updates, video releases, and more by signing up for our newsletter!More Curriculum Like This

Students are introduced to the concepts of air pollution, air quality, and climate change. The three lesson parts (including the associated activities) focus on the prerequisites for understanding air pollution. First, students use M&M® candies to create pie graphs that express their understanding o...

Students learn what causes air pollution and how to investigate the different pollutants that exist, such as toxic gases and particulate matter. They investigate the technologies developed by engineers to reduce air pollution.

Students learn the basics about the structure of the Earth’s atmosphere, the types of pollutants that are present in the atmosphere (primary, secondary, gas-phase compounds, particulate matter), and the importance of air quality research. They are also introduced to some engineering concepts such as...

Students develop an understanding of visible air pollutants with an incomplete combustion demonstration, a "smog in a jar" demonstration, building simple particulate matter collectors, and exploration of engineering roles related to air pollution. In an associated literacy activity, students learn b...
References
Andromeda, MacMillan Encyclopedia of Science, Revised Edition, Volume 7, The Environment, New York, NY: MacMillan Reference Books, 1997.
Bosak, Susan V. Science is...: A source book of fascinating facts, projects, and activities. Markham, Ontario: Scholastic Canada Ltd., pg. 207, 2000.
DiSpezio, Science Insights: Exploring Matter and Energy, New York, NY: Addison Wesley, 1996.
Kerrod, Robin and Evans, Ted. The Environment (Let's Investigate Science), New York: Benchmark Books, 1993.
Lawrence Hall of Science, University of California, Berkley. FOSS Science Series, Environments, Nashua, NH: Delta Education, 1993.
Markle, Sandra. The Kids' Earth Handbook, New York, NY: Atheneum, 1991.
Peet, Bill. The Wump World, New York, NY: Houghton Mifflin, 1981.
Prentice Hall Science. Ecology Earth's Natural Resources Activity Book, New Jersey: Prentice Hall, Inc., 1993.
Stille, Darlene. The New True Book – Water Pollution, Chicago: Childrens Press, Inc., 1991.
US Environmental Protection Agency's, information on controlling source pollutants: http://www.epa.gov/docs/oar/oaqps/eog/course422/ce6a.html
US Environmental Protection Agency's definition of and information on air pollutants: https://www.epa.gov/environmental-topics/air-topics
Wisconsin Department of Natural Resources, Kid's Page: http://dnr.wi.gov/org/caer/ce/eek/earth/index.htm
AirNow, a source for air quality data: https://www.airnow.gov/
Copyright
© 2005 by Regents of the University of ColoradoContributors
Amy Kolenbrander; Jessica Todd; Malinda Schaefer Zarske; Janet YowellSupporting Program
Integrated Teaching and Learning Program, College of Engineering, University of Colorado BoulderAcknowledgements
The contents of this digital library curriculum were developed under grants from the Fund for the Improvement of Postsecondary Education (FIPSE), U.S. Department of Education and National Science Foundation (GK-12 grant no. 0338326). However, these contents do not necessarily represent the policies of the Department of Education or National Science Foundation, and you should not assume endorsement by the federal government.
Last modified: September 29, 2020







User Comments & Tips